Abstract
Objective:
The regulatory processes that modulate adiponectin production and the mechanisms involved in nuclear factor kB (NF-kB) transcriptional activity in human adipocytes are not yet fully known. The aim of our study was to evaluate the inter-relationships between body fat, fat distribution, systemic inflammation, insulin resistance, leptin and the serum and subcutaneous adipose tissue gene expression levels of tumor necrosis factor-alpha (TNF-α), adiponectin and the inhibitor kappa B-alpha (IkB-α), in subjects with a wide range of body mass index (BMI). We also wanted to determine which of these variables was most closely related to adiponectin gene expression and adipocyte NF-kB transcriptional power.
Methods:
A total of 27 women aged between 50 and 80 years, with BMI ranging from 22.1 to 53.3 kg/m2, were studied. In all subjects BMI, waist circumference, body composition by dual X-ray absorptometry, triglycerides, cholesterol, high-density lipoprotein cholesterol (HDL-Ch), glucose, insulin, homeostasis model assessment of insulin resistance (HOMA), high-sensitive C-reactive protein (hs-CRP), serum adiponectin, leptin and TNF-α were evaluated. Subcutaneous adipose tissue biopsies were taken from the abdomen of all subjects and the mRNA levels of adiponectin, TNF-α and IkB-α were determined.
Results:
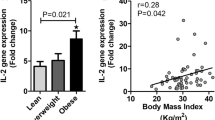
BMI and waist circumference were associated positively with leptin, HOMA, and hs-CRP, and negatively with HDL-Ch; waist was also associated with adiponectin and IkB-α mRNA. HOMA was negatively associated with serum adiponectin and adiponectin mRNA. Hs-CRP was negatively associated with IkB-α mRNA, and was positively associated with HOMA. Step-down multiple regression analysis was performed to determine the joint effects of BMI, waist circumference, triglycerides, HDL-Ch, HOMA, hs-CRP, leptin, serum and TNF-α mRNA on adiponectin gene expression: waist circumference and leptin were both included in the best fitting regression equation for predicting adiponectin gene expression (R2=0.403, P=0.006). Stepwise multiple regression analysis was performed, considering IkB-α mRNA as a dependent variable and BMI, waist, HDL-Ch, HOMA, hs-CRP and adiponectin mRNA as independent variables. Adiponectin mRNA was the only variable to enter the regression (R2=0.406, P<0.001).
Conclusion:
Our results suggest that abdominal adiposity and leptin are independent predictors of adiponectin gene expression and that in human adipocytes, adiponectin gene expression is strongly related to IkB-α mRNA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Visser M, Bouter LM, McQuillan GM, Wener MH, Harris TB . Elevated C-reactive protein levels in overweight and obese adults. JAMA 1999; 282: 2131–2135.
Greenberg AS, Obin MS . Obesity and the role of adipose tissue in inflammation and metabolism. Am J Clin Nutr 2006; 83: S461–S465.
McLaughlin T, Abbasi F, Lamendola C, Liang L, Reaven G, Schaaf P et al. Differentiation between obesity and insulin resistance in the association with C-reactive protein. Circulation 2002; 106: 2908–2912.
Arner P . The adipocyte in insulin resistance: key molecules and the impact of the thiazolidinediones. Trends Endocrinol Metab 2003; 14: 137–145.
Lihn AS, Pedersen SB, Richelsen B . Adiponectin: action, regulation and association to insulin sensitivity. Obes Rev 2005; 6: 13–21.
Sharma AM, Tarnopolsky MA . Regulating adiponectin: of flax and flux. Diabetologia 2005; 48: 1035–1037.
Ouchi N, Kihara S, Arita Y, Okamoto Y, Maeda K, Kuriyama H et al. Adiponectin, an adipocyte-derived plasma protein, inhibits endothelial NF-kB signalling through a c-AMPdependent pathway. Circulation 2000; 102: 1296–1301.
Arita Y, Kihara S, Ouchi N, Takahashi M, Maeda K, Miyagawa J et al. Paradoxical decrease of an adipose-specific protein, adiponectin, in obesity. Biochem Biophys Res Commun 1999; 257: 79–83.
Weyer C, Funahashi T, Tanaka S, Hotta K, Matsuzawa Y, Pratley RE et al. Hypoadiponectinemia in obesity and type 2 diabetes: close association with insulin resistance and hyperinsulinemia. J Clin Endocrinol Metab 2001; 86: 1930–1935.
Matsubara M, Namioka K, Katayose S . Decreased plasma adiponectin concentrations in women with low-grade C-reactive protein elevation. Eur J Endocrinol 2003; 148: 657–662.
Krakoff J, Funahashi T, Stehouwer CDA, Schalkwijk CG, Tanaka S, Matsuzawa Y et al. Inflammatory markers, adiponectin, and risk of type 2 diabetes in the Pima Indian. Diabetes Care 2003; 26: 1745–1751.
Ajuwon KM, Spurlock ME . Adiponectin inhibits LPS-induced NF-kB activation and IL-6 production and increases PPARγ2 expression in adipocytes. Am J Physiol Regul Integr Comp Physiol 2005; 288: R1220–R1225.
Barnes PJ, Karin M . Nuclear factor-k-B- A pivotal transcription factor in chronic inflammatory diseases. N Engl J Med 1997; 15: 1066–1071.
Karin M, Yamamoto Y, Wang QM . The IKK NF-kB system: a treasure trove for drug development. Nat Rev Drug Discov 2004; 3: 17–26.
Aljada A, Ghanim H, Assian E, Mohanty P, Hamouda W, Garg R et al. Increased IkB expression and diminished nuclear NF-kB in human mononuclear cells following hydrocortisone injection. J Clin Endocr Metab 1999; 84: 3386–3389.
Dandona P, Aljada A, Mohanty P, Ghanim H, Hamouda W, Assian E et al. Insulin inhibits intranuclear nuclear factor kB and stimulates IkB in mononuclear cells in obese subjects: evidence for an anti-inflammatory effect? J Clin Endocr Metab 2001; 86: 3257–3265.
The Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Report of the Expert Committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 2003; 26: S5–S20.
Mazess RB, Barden HS, Bisek JP, Hanson J . Dual-energy X-ray Absorptiometry for total-body and regional bone-mineral and soft-tissue composition. Am J Clin Nutr 1990; 51: 1106–1112.
Bonora E, Targher G, Alberiche M, Bonadonna RC, Saggiani F, Zenere MB et al. Homeostasis model assessment closely mirrors the glucose clamp technique in the assessment of insulin sensitivity. Diabetes Care 2000; 23: 57–63.
Corporate SPSS Inc. 2nd edn. McGraw-Hill: New York, 1986.
You T, Yang R, Lyles MF, Gong D, Nicklas BJ . Abdominal adipose tissue cytokine gene expression: relationship to obesity and metabolic risk factors. Am J Physiol Endocrinol Metab 2005; 288: E741–E747.
Bosello O, Zamboni M . Visceral obesity and metabolic syndrome. Obesity Rev 2000; 1: 47–56.
Garg A . Regional adiposity and insulin resistance. J Clin Endocrinol Metab 2004; 89: 4206–4210.
Lemieux I, Pascot A, Prud'homme D, Alméras N, Bogaty P, Nadeau A et al. Elevated C-reactive protein: another component of the atherothrombotic profile of abdominal obesity. Arterioscler Thromb Vasc Biol 2001; 21: 961–967.
Fasshauer M, Klein J, Neumann S, Eszlinger M, Paschke R . Hormonal regulation of adiponectin gene expression in 3T3-L1 adipocytes. Biochem Biophys Res Commun 2002; 290: 1084–1089.
Lihn AS, Østergård T, Nyholm B, Pedersen SB, Richelsen B, Schmitz O . Adiponectin expression in adipose tissue is reduced in first-degree relatives of type 2 diabetic patients. Am J Physiol Endocrinol Metab 2003; 284: E443–E448.
Ueno N, Dube MG, Inui A, Kalra PS, Kalra SP . Leptin modulates orexigenic effects of ghrelin and attenuates adiponectin and insulin levels and selectively the dark-phase feeding as revealed by central leptin gene therapy. Endocrinology 2004; 145: 4176–4184.
Chen J, Tan B, Karteris E, Zervou S, Digby J, Hillhouse EW et al. Secretion of adiponectin by human placenta: differential modulation of adiponectin and its receptors by cytokines. Diabetologia 2006; 49: 1292–1302.
Bottero V, Imbert V, Frelin C, Formento JL, Peyron JF . Monitoring NF-kappa B transactivation potential via real-time PCR quantification of I kappa B-alpha gene expression. Mol Diagn 2003; 7: 187–194.
Acknowledgements
This study was supported by grants from MIUR project COFIN no. 2005063885_005.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zamboni, M., Di Francesco, V., Garbin, U. et al. Adiponectin gene expression and adipocyte NF-κB transcriptional activity in elderly overweight and obese women: inter-relationships with fat distribution, hs-CRP, leptin and insulin resistance. Int J Obes 31, 1104–1109 (2007). https://doi.org/10.1038/sj.ijo.0803563
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0803563
Keywords
This article is cited by
-
Protectin DX ameliorates palmitate- or high-fat diet-induced insulin resistance and inflammation through an AMPK-PPARα-dependent pathway in mice
Scientific Reports (2017)
-
Interactions of exercise training and high-fat diet on adiponectin forms and muscle receptors in mice
Nutrition & Metabolism (2016)
-
Acerola (Malpighia emarginata DC.) juice intake protects against alterations to proteins involved in inflammatory and lipolysis pathways in the adipose tissue of obese mice fed a cafeteria diet
Lipids in Health and Disease (2014)
-
Favorable modulation of benign breast tissue and serum risk biomarkers is associated with >10 % weight loss in postmenopausal women
Breast Cancer Research and Treatment (2013)
-
The use of LeptiCore® in reducing fat gain and managing weight loss in patients with metabolic syndrome
Lipids in Health and Disease (2010)