Abstract
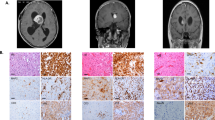
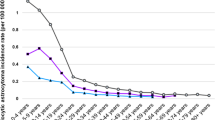
In astrocytic neoplasms, the number of cells expressing glial fibrillary acidic protein (GFAP) is inversely proportional to the extent of anaplasia. The loss of GFAP expression, the principal marker of astroglial cells, in these tumors has been proposed to constitute a step in their development and progression. To test this hypothesis, we crossed p53-negative (p53−/−) mice, which frequently develop astrocytomas after intrauterine exposure to ethylnitrosourea, with GFAP-negative (GFAP−/−) mice or GFAP+/+ controls. Brain tumors of glial origin were found in 12 of 35 GFAP+/+ p53−/− mice (34%) and in 11 of 27 GFAP−/− p53−/− mice (41%). The two groups did not differ in the age at which tumors were detected or in tumor histology or progression. Thus, the loss of GFAP expression frequently seen in high-grade astrocytomas does not constitute a step in tumor development. Rather, it may represent the undifferentiated state of these cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bigner SH, McLendon RE, Al-dosari N and Rasheed A . (1998). The Genetic Basis of Human Cancer. Vogelstein B and Kinsler KW (eds). McGraw-Hill: New York, pp. 661–670.
Chen WJ and Liem RK . (1994). J. Cell Biol., 127, 813–823.
Dalton S . (1992). EMBO J., 11, 1797–1804.
Deck JH, Eng LF, Bigbee J and Woodcock SM . (1978). Acta Neuropathol. (Berl.), 42, 183–190.
Eliasson C, Sahlgren C, Berthold CH, Stakeberg J, Celis JE, Betsholtz C, Eriksson JE and Pekny M . (1999). J. Biol. Chem., 274, 3996–4006.
Eng LF, Vanderhaeghen JJ, Bignami A and Gerstl G . (1971). Brain Res., 28, 351–354.
Eng LF and Rubinstein LJ . (1978). J. Histochem. Cytochem., 26, 513–522.
Eng LF, Ghirnikar RS and Lee YL . (2000). Neurochem. Res., 25, 1439–1451.
Engebraaten O, Hjortland GO, Hirschberg H and Fodstad O . (1999). J. Neurosurg., 90, 125–132.
Hara A, Sakai N, Yamada H, Niikawa S, Ohno T, Tanaka T and Mori H . (1991). Surg. Neurol., 36, 190–194.
Inagaki M, Gonda Y, Nishizawa K, Kitamura S, Sato C, Ando S, Tanabe K, Kikuchi K, Tsuiki S and Nishi Y . (1990). J. Biol. Chem., 265, 4722–4729.
Jacque CM, Vinner C, Kujas M, Raoul M, Racadot J and Baumann NA . (1978). J. Neurol. Sci., 35, 147–155.
Jacque CM, Kujas M, Poreau A, Raoul M, Collier P, Racadot J and Baumann N . (1979). J. Natl. Cancer Inst., 62, 479–483.
Kajiwara K, Orita T, Nishizaki T, Kamiryo T, Nakayama H and Ito H . (1992). Brain Res., 572, 314–318.
Ku NO, Liao J, Chou CF and Omary MB . (1996). Cancer Metastasis Rev., 15, 429–444.
Leonard JR, D'Sa C, Klocke BJ and Roth KA . (2001). Oncogene, 20, 8281–8286.
Livingstone LR, White A, Sprouse J, Livanos E, Jacks T and Tlsty TD . (1992). Cell, 70, 923–935.
Oda H, Zhang S, Tsurutani N, Shimizu S, Nakatsuru Y, Aizawa S and Ishikawa T . (1997). Cancer Res., 57, 646–650.
Pekny M, Levéen P, Pekna M, Eliasson C, Berthold C-H, Westermark B and Betsholtz C . (1995). EMBO J., 14, 1590–1598.
Pekny M, Eliasson C, Chien CL, Kindblom LG, Liem R, Hamberger A and Betsholtz C . (1998). Exp. Cell Res., 239, 332–343.
Pekny M, Johansson CB, Eliasson C, Stakeberg J, Wallen A, Perlmann T, Lendahl U, Betsholtz C, Berthold CH and Frisen J . (1999). J. Cell Biol., 145, 503–514.
Pekny M . (2001). Prog. Brain Res., 132, 23–30.
Rutka JT and Smith SL . (1993). Cancer Res., 53, 3624–3631.
Rutka JT, Hubbard SL, Fukuyama K, Matsuzawa K, Dirks PB and Becker LE . (1994). Cancer Res., 54, 3267–3272.
Sidransky D, Mikkelsen T, Schwechheimer K, Rosenblum ML, Cavanee W and Vogelstein B . (1992). Nature, 355, 846–847.
Steck PA, Pershouse MA, Jasser SA, Yung WK, Lin H, Ligon AH, Langford LA, Baumgard ML, Hattier T, Davis T, Frye C, Hu R, Swedlund B, Teng DH and Tavtigian SV . (1997). Nat. Genet., 15, 356–362.
Tascos NA, Parr J and Gonatas NK . (1982). Hum. Pathol., 13, 454–458.
Toda M, Miura M, Asou H, Toya S and Uyemura K . (1994). J. Neurochem., 63, 1975–1978.
Toda M, Miura M, Asou H, Sugiyama I, Kawase T and Uyemura K . (1999). Neurochem. Res., 24, 339–343.
Tsujimura K, Tanaka J, Ando S, Matsuoka Y, Kusubata M, Sugiura H, Yamauchi T and Inagaki M . (1994). J. Biochem. (Tokyo), 116, 426–434.
van der Meulen JD, Houthoff HJ and Ebels EJ . (1978). Neuropathol. Appl. Neurobiol., 4, 177–190.
Velasco ME, Dahl D, Roessmann U and Gambetti P . (1980). Cancer, 45, 484–494.
Wechsler W, Rice JM and Vesselinovitch SD . (1979). Natl. Cancer Inst. Monogr., 51, 219–226.
Weinstein DE, Shelanski ML and Liem RK . (1991). J. Cell. Biol., 112, 1205–1213.
Weiss WA . (2000). Curr. Opin. Pediatr., 6, 543–548.
Westermark B . (1973). Int. J. Cancer, 12, 438–451.
Yahanda AM, Bruner JM, Donehower LA and Morrison RS . (1995). Mol. Cell. Biol., 15, 4249–4259.
Yong VW . (1992). J. Neurol. Sci., 111, 92–103.
Zhu Y and Parada LF . (2002). Nat. Rev. Cancer, 8, 616–626.
Acknowledgements
This article is dedicated to the memory of Professor Jan Pontén. We thank Dr Ricardo Feinstein (State Veterinary Institute, Uppsala, Sweden) for help with characterization of the tumors, Professor Christer Betsholtz for valuable discussions, and Dr Marcela Pekna for critical reading of the manuscript. This study was supported by grants from the Swedish Cancer Foundation (project no. 3622), the Swedish Medical Research Council (project no. 11548), the Swedish Society for Medicine, the Swedish Society for Medical Research, the King Gustaf V Foundation, Volvo Assar Gabrielsson Fond, and the Swedish Stroke Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wilhelmsson, U., Eliasson, C., Bjerkvig, R. et al. Loss of GFAP expression in high-grade astrocytomas does not contribute to tumor development or progression. Oncogene 22, 3407–3411 (2003). https://doi.org/10.1038/sj.onc.1206372
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206372
Keywords
This article is cited by
-
Molecular features of glioblastomas in long-term survivors compared to short-term survivors—a matched-pair analysis
Radiation Oncology (2022)
-
Rat Adipose-Derived Stromal Cells (ADSCs) Increases the Glioblastoma Growth and Decreases the Animal Survival
Stem Cell Reviews and Reports (2022)
-
Virus vector-mediated genetic modification of brain tumor stromal cells after intravenous delivery
Journal of Neuro-Oncology (2018)
-
Concurrent Chemotherapy of Malignant Glioma in Rats by Using Multidrug-Loaded Biodegradable Nanofibrous Membranes
Scientific Reports (2016)
-
Prospective evaluation of serum glial fibrillary acidic protein (GFAP) as a diagnostic marker for glioblastoma
Journal of Neuro-Oncology (2016)