Abstract
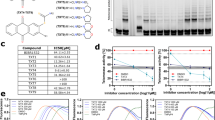
The vast majority of human cancers express telomerase activity, while most human somatic cells do not have detectable telomerase activity. Since telomerase plays a critical role in cell immortality, it is an attractive target for a selective cancer therapy. Oligonucleotides complementary to the RNA template region of human telomerase (hTR) have been shown to be effective inhibitors of telomerase and, subsequently, cancer cell growth in vitro. We show here that a lipid-modified N3′ → P5′ thio-phosphoramidate oligonucleotide (GRN163L) inhibits telomerase more potently than its parental nonconjugated thio-phosphoramidate sequence (GRN163). Cells were treated with both the first- (GRN163) and second-generation (GRN163L) oligonucleotides, including a mismatch control, with or without a transfection enhancer reagent. GRN163L inhibited telomerase activity effectively in a dose-dependent manner, even without the use of a transfection reagent. The IC50 values for GRN163 in various cell lines were on average sevenfold higher than for GRN163L. GRN163L inhibition of telomerase activity resulted in a more rapid loss of telomeres and cell growth than GRN163. This report is the first to show that lipid modification enhanced the potency of the novel GRN163 telomerase inhibitor. These results suggest that the lipid-conjugated thio-phosphoramidates could be important for improved pharmacodynamics of telomerase inhibitors in cancer therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Agrawal S, Goodchild J, Civeira MP, Thornton AH, Sarin PS and Zamecnik PC . (1988). Proc. Natl. Acad. Sci. USA, 85, 7079–7083.
Akiyama M, Hideshima T, Shammas MA, Hayashi T, Hamasaki M, Tai YT, Richardson P, Gryaznov S, Munshi NC and Anderson KC . (2003). Cancer Res., 63, 6187–6194.
Asai A, Oshima Y, Yamamoto Y, Uochi TA, Kusaka H, Akinaga S, Yamashita Y, Pongracz K, Pruzan R, Wunder E, Piatyszek M, Li S, Chin AC, Harley CB and Gryaznov S . (2003). Cancer Res., 63, 3931–3939.
Boutorin AS, Gus'kova LV, Ivanova EM, Kobetz ND, Zarytova VF, Ryte AS, Yurchenko LV and Vlassov VV . (1989). FEBS Lett., 254, 129–132.
Cong YS, Wright WE and Shay JW . (2002). Microbiol. Mol. Biol. Rev., 66, 407–425.
de Lange T . (2002). Oncogene, 21, 532–540.
Farooqui F, Sarin PS, Sun D and Letsinger RL . (1991). Bioconjug. Chem., 2, 422–426.
Hahn WC, Stewart SA, Brooks MW, York SG, Eaton E, Kurachi A, Beijersbergen RL, Knoll JH, Meyerson M and Weinberg RA . (1999). Nat. Med., 5, 1164–1170.
Herbert B, Pitts AE, Baker SI, Hamilton SE, Wright WE, Shay JW and Corey DR . (1999). Proc. Natl. Acad. Sci. USA, 96, 14276–14281.
Herbert BS, Pongracz K, Shay JW and Gryaznov SM . (2002). Oncogene, 21, 638–642.
Holt SE, Norton JC, Wright WE and Shay JW . (1996). Methods Cell Sci., 18, 237–248.
Kim NW and Wu F . (1997). Nucleic Acids Res., 25, 2595–2597.
Kim NW, Piatyszek MA, Prowse KR, Harley CB, West MD, Ho PL, Coviello GM, Wright WE, Weinrich SL and Shay JW . (1994). Science, 266, 2011–2015.
Letsinger RL, Zhang GR, Sun DK, Ikeuchi T and Sarin PS . (1989). Proc. Natl. Acad. Sci. USA, 86, 6553–6556.
MacKellar C, Graham D, Will DW, Burgess S and Brown T . (1992). Nucleic Acids Res., 20, 3411–3417.
Mishra RK, Moreau C, Ramazeilles C, Moreau S, Bonnet J and Toulme JJ . (1995). Biochim. Biophys. Acta, 1264, 229–237.
Morales CP, Holt SE, Ouellette M, Kaur KJ, Yan Y, Wilson KS, White MA, Wright WE and Shay JW . (1999). Nat. Genet., 21, 115–118.
Oberhauser B and Wagner E . (1992). Nucleic Acids Res., 20, 533–538.
Ouellette MM, Aisner DL, Savre-Train I, Wright WE and Shay JW . (1999). Biochem. Biophys. Res. Commun., 254, 795–803.
Ozawa T, Gryaznov SM, Hu LJ, Pongracz K, Santos RA, Bollen AW, Lamborn KR and Deen DF . (2004). Neuro-oncology, 6, 218–226.
Piatyszek MA, Kim NW, Weinrich SL, Hiyama K, Hiyama E, Wright WE and Shay JW . (1995). Methods Cell Sci., 17, 1–15.
Polushin NN and Cohen JS . (1994). Nucleic Acids Res., 22, 5492–5496.
Reed MW, Adams AD, Nelson JS and Meyer Jr RB . (1991). Bioconjug. Chem., 2, 217–225.
Shay JW, Tomlinson G, Piatyszek MA and Gollahon LS . (1995). Mol. Cell. Biol., 15, 425–432.
Shea RG, Marsters JC and Bischofberger N . (1990). Nucleic Acids Res., 18, 3777–3783.
Stein CA, Pal R, DeVico AL, Hoke G, Mumbauer S, Kinstler O, Sarngadharan MG and Letsinger RL . (1991). Biochemistry, 30, 2439–2444.
Wang ES, Wu K, Chin AC, Chen-Kiang S, Pongracz K, Gryaznov S and Moore MA . (2004). Blood, 103, 258–266.
White LK, Wright WE and Shay JW . (2001). Trends Biotechnol., 19, 114–120.
Wright WE, Shay JW and Piatyszek MA . (1995). Nucleic Acids Res., 23, 3794–3795.
Yi X, White DM, Aisner DL, Baur JA, Wright WE and Shay JW . (2000). Neoplasia, 2, 433–440.
Zhang X, Mar V, Zhou W, Harrington L and Robinson MO . (1999). Genes Dev., 13, 2388–2399.
Acknowledgements
We thank M White for providing the H-RasV12 viral supernatant, D LaRue and M Granger for technical assistance, Sherry Hunt for veterinary assistance, Barbara Norman for assistance with tissue sectioning, and the Indiana University School of Medicine Office of Visual Media for assistance with the figures. The work at IUSM was supported by the Indiana Genomics Initiative (INGEN). INGEN of Indiana University is supported in part by Lilly Endowment Inc. Work carried out at UTSW was supported by the Geron Corporation (Menlo Park, CA), NCI CA85363 (JWS), NCI CA70907 (JWS), and DAMD17-00-1-0438 (to B-SH while a postdoctoral fellow at UTSW).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Herbert, BS., Gellert, G., Hochreiter, A. et al. Lipid modification of GRN163, an N3′ → P5′ thio-phosphoramidate oligonucleotide, enhances the potency of telomerase inhibition. Oncogene 24, 5262–5268 (2005). https://doi.org/10.1038/sj.onc.1208760
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208760
Keywords
This article is cited by
-
Telomerase gene therapy: a remission toward cancer
Medical Oncology (2022)
-
Pancreatic adenocarcinoma: molecular drivers and the role of targeted therapy
Cancer and Metastasis Reviews (2021)
-
A method for measuring the distribution of the shortest telomeres in cells and tissues
Nature Communications (2017)
-
Novel Therapies for Myelofibrosis
Current Hematologic Malignancy Reports (2017)
-
Roles of telomeres and telomerase in cancer, and advances in telomerase-targeted therapies
Genome Medicine (2016)