Abstract
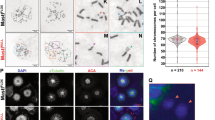
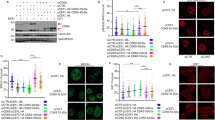
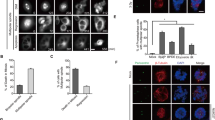
We previously demonstrated that type 2C protein phosphatases (PP2C) Ptc2 and Ptc3 are required for DNA checkpoint inactivation after DNA double-strand break repair or adaptation in Saccharomyces cerevisiae. Here, we show the conservation of this pathway in mammalian cells. In response to DNA damage, ataxia telangiectasia mutated (ATM) phosphorylates the Chk2 tumour suppressor kinase at threonine 68 (Thr68), allowing Chk2 kinase dimerization and activation by autophosphorylations in the T-loop. The oncogenic protein Wip1, a PP2C phosphatase, binds Chk2 and dephosphorylates phospho-Thr68. Consequently, Wip1 opposes Chk2 activation by ATM after ionizing irradiation of cells. In HCT15 colorectal cancer cells corrected for functional Chk2 activity, Wip1 overexpression suppressed the contribution of Chk2 to the G2/M DNA damage checkpoint. These results indicate that Wip1 is one of the phosphatases regulating the activity of Chk2 in response to DNA damage.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ahn J, Prives C . (2002). Checkpoint kinase 2 (Chk2) monomers or dimers phosphorylate Cdc25C after DNA damage regardless of threonine 68 phosphorylation. J Biol Chem 277: 48418–48426.
Ahn J, Urist M, Prives C . (2004). The Chk2 protein kinase. DNA Repair (Amsterdam) 3: 1039–1047.
Ahn JY, Li X, Davis HL, Canman CE . (2002). Phosphorylation of threonine 68 promotes oligomerization and autophosphorylation of the Chk2 protein kinase via the forkhead-associated domain. J Biol Chem 277: 19389–19395.
Bahassi el M, Myer DL, McKenney RJ, Hennigan RF, Stambrook PJ . (2006). Priming phosphorylation of Chk2 by polo-like kinase 3 (Plk3) mediates its full activation by ATM and a downstream checkpoint in response to DNA damage. Mutat Res 596: 166–176.
Bartek J, Lukas J . (2003). Chk1 and Chk2 kinases in checkpoint control and cancer. Cancer Cell 3: 421–429.
Bartkova J, Horejsi Z, Koed K, Kramer A, Tort F, Zieger K et al. (2005). DNA damage response as a candidate anti-cancer barrier in early human tumorigenesis. Nature 434: 864–870.
Bulavin DV, Phillips C, Nannenga B, Timofeev O, Donehower LA, Anderson CW et al. (2004). Inactivation of the Wip1 phosphatase inhibits mammary tumorigenesis through p38 MAPK-mediated activation of the p16(Ink4a)-p19(Arf) pathway. Nat Genet 36: 343–350.
Choi J, Nannenga B, Demidov ON, Bulavin DV, Cooney A, Brayton C et al. (2002). Mice deficient for the wild-type p53-induced phosphatase gene (Wip1) exhibit defects in reproductive organs, immune function, and cell cycle control. Mol Cell Biol 22: 1094–1105.
Craig AL, Hupp TR . (2004). The regulation of CHK2 in human cancer. Oncogene 23: 8411–8418.
Dozier C, Bonyadi M, Baricault L, Tonasso L, Darbon JM . (2004). Regulation of Chk2 phosphorylation by interaction with protein phosphatase 2A via its B′ regulatory subunit. Biol Cell 96: 509–517.
Falck J, Lukas C, Protopopova M, Lukas J, Selivanova G, Bartek J . (2001). Functional impact of concomitant versus alternative defects in the Chk2-p53 tumour suppressor pathway. Oncogene 20: 5503–5510.
Fiscella M, Zhang H, Fan S, Sakaguchi K, Shen S, Mercer WE et al. (1997). Wip1, a novel human protein phosphatase that is induced in response to ionizing radiation in a p53-dependent manner. Proc Natl Acad Sci USA 94: 6048–6053.
Fujimoto H, Onishi N, Kato N, Takekawa M, Xu XZ, Kosugi A et al. (2005). Regulation of the antioncogenic Chk2 kinase by the oncogenic Wip1 phosphatase. Cell Death Differ 13: 1170–1180.
Goodarzi AA, Jonnalagadda JC, Douglas P, Young D, Ye R, Moorhead GB et al. (2004). Autophosphorylation of ataxia-telangiectasia mutated is regulated by protein phosphatase 2A. EMBO J 23: 4451–4461.
Gorgoulis VG, Vassiliou LV, Karakaidos P, Zacharatos P, Kotsinas A, Liloglou T et al. (2005). Activation of the DNA damage checkpoint and genomic instability in human precancerous lesions. Nature 434: 907–913.
Hirao A, Kong YY, Matsuoka S, Wakeham A, Ruland J, Yoshida H et al. (2000). DNA damage-induced activation of p53 by the checkpoint kinase Chk2. Science 287: 1824–1827.
Hirasawa A, Saito-Ohara F, Inoue J, Aoki D, Susumu N, Yokoyama T et al. (2003). Association of 17q21–q24 gain in ovarian clear cell adenocarcinomas with poor prognosis and identification of PPM1D and APPBP2 as likely amplification targets. Clin Cancer Res 9: 1995–2004.
Jazayeri A, Falck J, Lukas C, Bartek J, Smith GC, Lukas J et al. (2006). ATM- and cell cycle-dependent regulation of ATR in response to DNA double-strand breaks. Nat Cell Biol 8: 37–45.
Lee SB, Kim SH, Bell DW, Wahrer DC, Schiripo TA, Jorczak MM et al. (2001). Destabilization of CHK2 by a missense mutation associated with Li-Fraumeni Syndrome. Cancer Res 61: 8062–8067.
Leroy C, Lee SE, Vaze MB, Ochsenbien F, Guerois R, Haber JE et al. (2003). PP2C phosphatases Ptc2 and Ptc3 are required for DNA checkpoint inactivation after a double-strand break. Mol Cell 11: 827–835.
Liang X, Reed E, Yu JJ . (2006). Protein phosphatase 2A interacts with Chk2 and regulates phosphorylation at Thr-68 after cisplatin treatment of human ovarian cancer cells. Int J Mol Med 17: 703–708.
Lu X, Bocangel D, Nannenga B, Yamaguchi H, Appella E, Donehower LA . (2004). The p53-induced oncogenic phosphatase PPM1D interacts with uracil DNA glycosylase and suppresses base excision repair. Mol Cell 15: 621–634.
Lu X, Nannenga B, Donehower LA . (2005). PPM1D dephosphorylates Chk1 and p53 and abrogates cell cycle checkpoints. Genes Dev 19: 1162–1174.
Lukas C, Falck J, Bartkova J, Bartek J, Lukas J . (2003). Distinct spatiotemporal dynamics of mammalian checkpoint regulators induced by DNA damage. Nat Cell Biol 5: 255–260.
Matsuoka S, Rotman G, Ogawa A, Shiloh Y, Tamai K, Elledge SJ . (2000). Ataxia telangiectasia-mutated phosphorylates Chk2 in vivo and in vitro. Proc Natl Acad Sci USA 97: 10389–10394.
Melchionna R, Chen XB, Blasina A, McGowan CH . (2000). Threonine 68 is required for radiation-induced phosphorylation and activation of Cds1. Nat Cell Biol 2: 762–765.
Nannenga B, Lu X, Dumble M, Van Maanen M, Nguyen TA, Sutton R et al. (2006). Augmented cancer resistance and DNA damage response phenotypes in PPM1D null mice. Mol Carcinog (in press).
Pommier Y, Weinstein JN, Aladjem MI, Kohn KW . (2006). Chk2 molecular interaction map and rationale for Chk2 inhibitors. Clin Cancer Res 12: 2657–2661.
Saito-Ohara F, Imoto I, Inoue J, Hosoi H, Nakagawara A, Sugimoto T et al. (2003). PPM1D is a potential target for 17q gain in neuroblastoma. Cancer Res 63: 1876–1883.
Schwarz JK, Lovly CM, Piwnica-Worms H . (2003). Regulation of the Chk2 protein kinase by oligomerization-mediated cis- and trans-phosphorylation. Mol Cancer Res 1: 598–609.
Schweighofer A, Hirt H, Meskiene I . (2004). Plant PP2C phosphatases: emerging functions in stress signaling. Trends Plant Sci 9: 236–243.
Sinclair CS, Rowley M, Naderi A, Couch FJ . (2003). The 17q23 amplicon and breast cancer. Breast Cancer Res Treat 78: 313–322.
Takekawa M, Adachi M, Nakahata A, Nakayama I, Itoh F, Tsukuda H et al. (2000). p53-inducible wip1 phosphatase mediates a negative feedback regulation of p38 MAPK-p53 signaling in response to UV radiation. EMBO J 19: 6517–6526.
Wu X, Chen J . (2003). Autophosphorylation of checkpoint kinase 2 at serine 516 is required for radiation-induced apoptosis. J Biol Chem 278: 36163–36168.
Wu X, Webster SR, Chen J . (2001). Characterization of tumor-associated Chk2 mutations. J Biol Chem 276: 2971–2974.
Xiao Z, Chen Z, Gunasekera AH, Sowin TJ, Rosenberg SH, Fesik S et al. (2003). Chk1 mediates S and G2 arrests through Cdc25A degradation in response to DNA-damaging agents. J Biol Chem 278: 21767–21773.
Xu X, Tsvetkov LM, Stern DF . (2002). Chk2 activation and phosphorylation-dependent oligomerization. Mol Cell Biol 22: 4419–4432.
Yamaguchi H, Minopoli G, Demidov ON, Chatterjee DK, Anderson CW, Durell SR et al. (2005). Substrate specificity of the human protein phosphatase 2Cdelta, Wip1. Biochemistry 44: 5285–5294.
Zhao H, Watkins JL, Piwnica-Worms H . (2002). Disruption of the checkpoint kinase 1/cell division cycle 25A pathway abrogates ionizing radiation-induced S and G2 checkpoints. Proc Natl Acad Sci USA 99: 14795–14800.
Zhou BB, Elledge SJ . (2000). The DNA damage response: putting checkpoints in perspective. Nature 408: 433–439.
Acknowledgements
We warmly thank Anne Peyroche for her valuable critical reviewing of this manuscript. This work was partly financed by the Association pour la Recherche sur le Cancer (Project #3592 and #4470).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Oliva-Trastoy, M., Berthonaud, V., Chevalier, A. et al. The Wip1 phosphatase (PPM1D) antagonizes activation of the Chk2 tumour suppressor kinase. Oncogene 26, 1449–1458 (2007). https://doi.org/10.1038/sj.onc.1209927
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209927
Keywords
This article is cited by
-
Sustained CHK2 activity, but not ATM activity, is critical to maintain a G1 arrest after DNA damage in untransformed cells
BMC Biology (2021)
-
ATR kinase regulates its attenuation via PPM1D phosphatase recruitment to chromatin during recovery from DNA replication stress signalling
Journal of Biosciences (2018)
-
PPM1D phosphatase, a target of p53 and RBM38 RNA-binding protein, inhibits p53 mRNA translation via dephosphorylation of RBM38
Oncogene (2015)
-
Truncating mutations of PPM1D are found in blood DNA samples of lung cancer patients
British Journal of Cancer (2015)
-
Wip1 phosphatase in breast cancer
Oncogene (2015)