Abstract
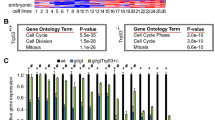
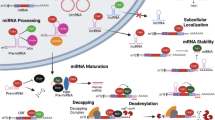
Genetic instabilities are believed to be one of the major causes of developing a cancer phenotype in humans. During the progression of cancer, aberrant expression of proteins, either owing to genetic (amplification, mutation and deletion) or epigenetic modifications (DNA methylation and histone deacetylation), contributes in different ways to the development of cancer. By differential screening analysis, an amplification of the 19q13 locus containing a novel pancreatic differentiation 2 (PD2) gene was identified. PD2 is the human homolog of the yeast RNA polymerase II-associated factor 1 (yPaf1) and is part of the human RNA polymerase II-associated factor (hPAF) complex. hPAF is comprised of five subunits that include PD2/hPaf1, parafibromin, hLeo1, hCtr9 and hSki8. This multifaceted complex was first identified in yeast (yPAF) and subsequently in Drosophila and human. Recent advances in the study on PAF have revealed various functions of the complex in human, which are similar to yPAF, including efficient transcription elongation, mRNA quality control and cell-cycle regulation. Although the precise function of this complex in cancer is not clearly known, some of its subunits have been linked to a malignant phenotype. Its core subunit, PD2/hPaf1, is amplified and overexpressed in many cancers. Further, an overexpression of PD2/hPaf1 results in the induction of a transformed phenotype, suggesting its possible involvement in tumorigenesis. The parafibromin subunit of the hPAF complex is a product of the HRPT-2 (hereditary hyperparathyroidism type 2) tumor suppressor gene, which is mutated in the germ line of hyperparathyroidism-jaw tumor patients. This review focuses on the functions of the PAF complex and its individual subunits, the interaction of the subunits with each other and/or with other molecules, and dysregulation of the complex, providing an insight into its potential involvement in the development of cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Armengol G, Knuutila S, Lluis F, Capella G, Miro R, Caballin MR . (2000). DNA copy number changes and evaluation of MYC, IGF1R, and FES amplification in xenografts of pancreatic adenocarcinoma. Cancer Genet Cytogenet 116: 133–141.
Bashyam MD, Bair R, Kim YH, Wang P, Hernandez-Boussard T, Karikari CA et al. (2005). Array-based comparative genomic hybridization identifies localized DNA amplifications and homozygous deletions in pancreatic cancer. Neoplasia 7: 556–562.
Batra SK, Metzgar RS, Hollingsworth MA . (1991). Molecular cloning and sequence analysis of the human ribosomal protein S16. J Biol Chem 266: 6830–6833.
Batra SK, Rasheed BK, Bigner SH, Bigner DD . (1994). Oncogenes and anti-oncogenes in human central nervous system tumors. Lab Invest 71: 621–637.
Birkenkamp-Demtroder K, Christensen LL, Olesen SH, Frederiksen CM, Laiho P, Aaltonen LA et al. (2002). Gene expression in colorectal cancer. Cancer Res 62: 4352–4363.
Bradley KJ, Bowl MR, Williams SE, Ahmad BN, Partridge CJ, Patmanidi AL et al. (2007). Parafibromin is a nuclear protein with a functional monopartite nuclear localization signal. Oncogene 26: 1213–1221.
Camps J, Armengol G, del Rey J, Lozano JJ, Vauhkonen H, Prat E et al. (2006). Genome-wide differences between microsatellite stable and unstable colorectal tumors. Carcinogenesis 27: 419–428.
Cavalli LR, Urban CA, Dai D, de AS, Tavares DC, Rone JD et al. (2003). Genetic and epigenetic alterations in sentinel lymph nodes metastatic lesions compared to their corresponding primary breast tumors. Cancer Genet Cytogenet 146: 33–40.
Chang MC, Chang YT, Tien YW, Sun CT, Wu MS, Lin JT . (2005). Distinct chromosomal aberrations of ampulla of Vater and pancreatic head cancers detected by laser capture microdissection and comparative genomic hybridization. Oncol Rep 14: 867–872.
Cheng JQ, Ruggeri B, Klein WM, Sonoda G, Altomare DA, Watson DK et al. (1996). Amplification of AKT2 in human pancreatic cells and inhibition of AKT2 expression and tumorigenicity by antisense RNA. Proc Natl Acad Sci USA 93: 3636–3641.
Curtis LJ, Li Y, Gerbault-Seureau M, Kuick R, Dutrillaux AM, Goubin G et al. (1998). Amplification of DNA sequences from chromosome 19q13.1 in human pancreatic cell lines. Genomics 53: 42–55.
De PK, Speleman F, Combaret V, Lunec J, Board J, Pearson A et al. (2005). No evidence for correlation of DDX1 gene amplification with improved survival probability in patients with MYCN-amplified neuroblastomas. J Clin Oncol 23: 3167–3168.
Grandori C, Robinson KL, Galloway DA, Swisshelm K . (2004). Functional link between Myc and the Werner gene in tumorigenesis. Cell Cycle 3: 22–25.
Hahn MA, Marsh DJ . (2005). Identification of a functional bipartite nuclear localization signal in the tumor suppressor parafibromin. Oncogene 24: 6241–6248.
Heidenblad M, Lindgren D, Veltman JA, Jonson T, Mahlamaki EH, Gorunova L et al. (2005). Microarray analyses reveal strong influence of DNA copy number alterations on the transcriptional patterns in pancreatic cancer: implications for the interpretation of genomic amplifications. Oncogene 24: 1794–1801.
Honore B, Baandrup U, Nielsen S, Vorum H . (2002). Endonuclein is a cell cycle regulated WD-repeat protein that is up-regulated in adenocarcinoma of the pancreas. Oncogene 21: 1123–1129.
Huntsman DG, Chin SF, Muleris M, Batley SJ, Collins VP, Wiedemann LM et al. (1999). MLL2, the second human homolog of the Drosophila trithorax gene, maps to 19q13.1 and is amplified in solid tumor cell lines. Oncogene 18: 7975–7984.
Knudsen KE, Diehl JA, Haiman CA, Knudsen ES . (2006). Cyclin D1: polymorphism, aberrant splicing and cancer risk. Oncogene 25: 1620–1628.
Koch C, Wollmann P, Dahl M, Lottspeich F . (1999). A role for Ctr9p and Paf1p in the regulation G1 cyclin expression in yeast. Nucleic Acids Res 27: 2126–2134.
Korshunov A, Sycheva R, Gorelyshev S, Golanov A . (2005). Clinical utility of fluorescence in situ hybridization (FISH) in nonbrainstem glioblastomas of childhood. Mod Pathol 18: 1258–1263.
Krogan NJ, Kim M, Ahn SH, Zhong G, Kobor MS, Cagney G et al. (2002). RNA polymerase II elongation factors of Saccharomyces cerevisiae: a targeted proteomics approach. Mol Cell Biol 22: 6979–6992.
Mahlamaki EH, Kauraniemi P, Monni O, Wolf M, Hautaniemi S, Kallioniemi A . (2004). High-resolution genomic and expression profiling reveals 105 putative amplification target genes in pancreatic cancer. Neoplasia 6: 432–439.
Mao X, Lillington D, Child F, Russell-Jones R, Young B, Whittaker S . (2002). Comparative genomic hybridization analysis of primary cutaneous B-cell lymphomas: identification of common genomic alterations in disease pathogenesis. Genes Chromosomes Cancer 35: 144–155.
Moniaux N, Nemos C, Schmied BM, Chauhan SC, Deb S, Morikane K et al. (2006). The human homologue of the RNA polymerase II-associated factor 1 (hPaf1), localized on the 19q13 amplicon, is associated with tumorigenesis. Oncogene 25: 3247–3257.
Mosimann C, Hausmann G, Basler K . (2006). Parafibromin/Hyrax activates Wnt/Wg target gene transcription by direct association with beta-catenin/Armadillo. Cell 125: 327–341.
Mueller CL, Porter SE, Hoffman MG, Jaehning JA . (2004). The Paf1 complex has functions independent of actively transcribing RNA polymerase II. Mol Cell 14: 447–456.
Nathrath MH, Kuosaite V, Rosemann M, Kremer M, Poremba C, Wakana S et al. (2002). Two novel tumor suppressor gene loci on chromosome 6q and 15q in human osteosarcoma identified through comparative study of allelic imbalances in mouse and man. Oncogene 21: 5975–5980.
Parada LA, Hallen M, Tranberg KG, Hagerstrand I, Bondeson L, Mitelman F et al. (1998). Frequent rearrangements of chromosomes 1, 7, and 8 in primary liver cancer. Genes Chromosomes Cancer 23: 26–35.
Pavri R, Zhu B, Li G, Trojer P, Mandal S, Shilatifard A et al. (2006). Histone H2B monoubiquitination functions cooperatively with FACT to regulate elongation by RNA polymerase II. Cell 125: 703–717.
Redeker E, Alders M, Hoovers JM, Richard III CW, Westerveld A, Mannens M . (1995). Physical mapping of 3 candidate tumor suppressor genes relative to Beckwith–Wiedemann syndrome associated chromosomal breakpoints at 11p15.3. Cytogenet Cell Genet 68: 222–225.
Rozenblatt-Rosen O, Hughes CM, Nannepaga SJ, Shanmugam KS, Copeland TD, Guszczynski T et al. (2005). The parafibromin tumor suppressor protein is part of a human Paf1 complex. Mol Cell Biol 25: 612–620.
Rybakin V, Clemen CS . (2005). Coronin proteins as multifunctional regulators of the cytoskeleton and membrane trafficking. BioEssays 27: 625–632.
Schleger C, Arens N, Zentgraf H, Bleyl U, Verbeke C . (2000). Identification of frequent chromosomal aberrations in ductal adenocarcinoma of the pancreas by comparative genomic hybridization (CGH). J Pathol 191: 27–32.
Schwienbacher C, Gramantieri L, Scelfo R, Veronese A, Calin GA, Bolondi L et al (2000). Gain of imprinting at chromosome 11p15: A pathogenetic mechanism identified in human hepatocarcinomas. Proc Natl Acad Sci USA 97: 5445–5449.
Stange DE, Radlwimmer B, Schubert F, Traub F, Pich A, Toedt G et al. (2006). High-resolution genomic profiling reveals association of chromosomal aberrations on 1q and 16p with histologic and genetic subgroups of invasive breast cancer. Clin Cancer Res 12: 345–352.
Tang TC, Sham JS, Xie D, Fang Y, Huo KK, Wu QL et al. (2002). Identification of candidate oncogene SEI-1 within a minimal amplified region at 19q13.1 in ovarian cancer cell lines. Cancer Res 62: 7157–7161.
Tarkkanen M, Larramendy ML, Bohling T, Serra M, Hattinger CM, Kivioja A et al. (2006). Malignant fibrous histiocytoma of bone: analysis of genomic imbalances by comparative genomic hybridisation and C-MYC expression by immunohistochemistry. Eur J Cancer 42: 1172–1180.
Thiagalingam S . (2006). A cascade of modules of a network defines cancer progression. Cancer Res 66: 7379–7385.
Vogelstein B, Kinzler KW . (2004). Cancer genes and the pathways they control. Nat Med 10: 789–799.
Woodard GE, Lin L, Zhang JH, Agarwal SK, Marx SJ, Simonds WF . (2005). Parafibromin, product of the hyperparathyroidism-jaw tumor syndrome gene HRPT2, regulates cyclin D1/PRAD1 expression. Oncogene 24: 1272–1276.
Zhang C, Kong D, Tan MH, Pappas Jr DL, Wang PF, Chen J et al. (2006). Parafibromin inhibits cancer cell growth and causes G1 phase arrest. Biochem Biophys Res Commun 350: 17–24.
Zhu B, Mandal SS, Pham AD, Zheng Y, Erdjument-Bromage H, Batra SK et al. (2005). The human PAF complex coordinates transcription with events downstream of RNA synthesis. Genes Dev 19: 1668–1673.
Acknowledgements
The authors are supported by grants from the National Institutes of Health (CA78590, CA111294), the US Department of Defense (PC040493, PC04502, OC04110) and the Peter Kiewit Foundation. We thank Drs Ajay P Singh and Subhankar Chakraborty for critical reading and Ms Kristi L Berger for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Chaudhary, K., Deb, S., Moniaux, N. et al. Human RNA polymerase II-associated factor complex: dysregulation in cancer. Oncogene 26, 7499–7507 (2007). https://doi.org/10.1038/sj.onc.1210582
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210582
Keywords
This article is cited by
-
UBE2S promotes cell chemoresistance through PTEN-AKT signaling in hepatocellular carcinoma
Cell Death Discovery (2021)
-
A KDM4A-PAF1-mediated epigenomic network is essential for acute myeloid leukemia cell self-renewal and survival
Cell Death & Disease (2021)
-
Downregulation of SELENBP1 enhances oral squamous cell carcinoma chemoresistance through KEAP1–NRF2 signaling
Cancer Chemotherapy and Pharmacology (2021)
-
Genomics and Epigenomics in Parathyroid Neoplasia: from Bench to Surgical Pathology Practice
Endocrine Pathology (2021)
-
Paf1 and Ctr9 subcomplex formation is essential for Paf1 complex assembly and functional regulation
Nature Communications (2018)