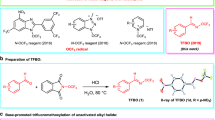
Abstract
REACTIONS of organic halides in solution which involve substitution by a nucleophilic reagent at a saturated carbon atom have been extensively studied by Hughes, Ingold and co-workers1. These authors conclude that reactions of this type may occur by two possible mechanisms, a unimolecular mechanism or a bimolecular mechanism . The increase in the unimolecular reaction-rate of the halide R – X as R varies along the series methyl, ethyl, sec.-propyl, tert.butyl, is attributed by these authors to the increase in electron accession to the reaction centre2. The decrease in the bimolecular reaction-rate along this series3 is also attributed to the increase in electron accession to the reaction centre ; this electron accession is assumed to inhibit the approach of the nucleophilic reagent.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ingold and Rothstein, J. Chem. Soc., 1217 (1928). Hughes, Ingold and Patel, J. Chem. Soc., 526 (1933). For further references see "General Discussion on Mechanism of Organic Reactions in Liquid Systems", Trans. Farad. Soc., 37, 601 (1941).
Bateman and Hughes, J. Chem. Soc., 945 (1940).
Bateman, Cooper, Hughes and Ingold, J. Chem. Soc., 925 (1940).
Ogg and Polanyi, Trans. Farad. Soc., 31, 604 (1935).
Baughan, Evans, M. G., and Polanyi, Trans. Farad. Soc., 37, 377
Hippie and Stevenson, Phys. Rev., 63, 121 (1943).
Hippie and Stevenson, J. Amer. Chem. Soc., 64 1590, 2766, 2769 (1942).
Meer and Polanyi, Z. phys. Chem., B, 19, 164 (1932).
Evans, A. G., and Polanyi, Nature, 149, 608 (1942).
Hughes, Trans. Farad. Soc., 37, 603 (1941).
Young and Andrews, J. Amer. Chem. Soc., 66, 421 (1944).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
EVANS, A. Reactions of Organic Halides in Solution. Nature 157, 438–439 (1946). https://doi.org/10.1038/157438c0
Issue Date:
DOI: https://doi.org/10.1038/157438c0
This article is cited by
-
Carbene-catalysed reductive coupling of nitrobenzyl bromides and activated ketones or imines via single-electron-transfer process
Nature Communications (2016)
-
Reactions of Organic Halides in Solution
Nature (1946)
-
Reactions of Organic Halides in Solution
Nature (1946)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.