Abstract
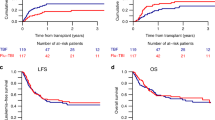
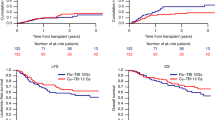
We compared fractionated total body irradiation (12 Gy)/cyclophosphamide (120 mg/kg) with busulfan (16 mg/kg)/cyclophosphamide (120 mg/kg) as preparative therapy in unrelated donor stem cell transplantation of CML patients. Fifty patients with CML (1.CP = 46; aP = 4) and a median age of 36 years (range 16–52) were enrolled in this sequential trial between 1994 and 1999. In both groups patients were well balanced with respect to age, disease status, stem cell source and CMV status. All patients received standard doses of cyclosporin A, methotrexate and anti-thymocyte globulin (ATG) as GVHD prophylaxis. No graft failures occurred in either group. The median day of leukocyte engraftment was earlier in the Bu/Cy than in the TBI/Cy group (day 15 vs 17; P = 0.006). The incidence of grade II–IV GVHD was 40% in the TBI/Cy and 36% in the Bu/Cy group, whereas severe grade III/IV GVHD was only observed in 12% of patients in both groups. The incidence of chronic GVHD (limited and extensive) at 1 year was higher in the Bu/Cy arm (65% vs 30%; P = 0.02). More toxicity grade I/II of the liver (88% vs 44%; P = 0.002) and more hemorrhagic cystitis (32% vs 8%; P = 0.02) were observed in the Bu/Cy regimen. Seven relapses in the TBI and no relapse in the Bu/Cy group were observed after a median follow-up of 44 and 15 months, respectively. The estimated 3 year OS and DFS was 72% (95% CI: 55–98%) and 58% (95% CI: 39–77%) in the TBI and 70% (95% CI: 51–89%) for DFS and OS in the Bu/Cy group. We conclude that the anti-leukemic effect of the Bu/Cy regimen seems to be at least as effective as the TBI/Cy combination in unrelated stem cell transplantation of CML patients, with no graft failures, but that it correlates with a higher incidence of liver toxicity, hemorrhagic cystitis and chronic GVHD. Longer follow-up is necessary to determine the late relapse rate and late toxicity. Bone Marrow Transplantation (2001) 27, 349–354.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Marks D, Cullis J, Ward K et al. Allogeneic bone marrow transplantation for chronic myeloid leukemia using sibling and volunteer unrelated donors: a comparison of complications in the first 2 years Ann Intern Med 1993 19: 207–214
McGlave P, Bartsch G, Anasetti C et al. Unrelated donor marrow transplantation for chronic myelogenous leukemia: Initial experience of the National Marrow Donor Program Blood 1993 81: 543–550
Kernan NA, Bartsch G, Ash RC et al. Analysis of 462 transplantations from unrelated donors facilitated by the national marrow donor program New Engl J Med 1993 328: 593–602
Hansen JA, Gooley TA, Martin PJ et al. Bone marrow transplants from unrelated donors for patients with chronic myeloid leukemia New Engl J Med 1998 338: 962–968
Spencer A, Szydia RM, Brookes PA et al. Bone marrow transplantation for chronic myeloid leukemia with volunteer unrelated donors using ex vivo or in vivo T-cell depletion: major prognostic impact of HLA class I identity between donor and recipient Blood 1995 86: 3590–3597
Devergie A, Blaise D, Attal M et al. Allogeneic bone marrow transplantation for chronic myeloid leukemia in first chronic phase: a randomized trial of busulfan-cytoxan versus cytoxan-Total-body irradiation as preparative regimen: a report from the French Society of Bone Marrow Graft Blood 1995 85: 2263–2268
Clift RA, Buckner CD, Thomas ED et al. Marrow transplantation for chronic myeloid leukemia: a randomized study comparing cyclophosphamide and total body irradiation with busulfan and cyclophosphamide Blood 1994 84: 2036–2043
Mehta J, Powles RL, Mitchell P et al. Graft failure after bone marrow transplantation from unrelated donors using busulfan and cyclophosphamide for conditioning Bone Marrow Transplant 1994 13: 583–587
Klein JL, Avalos BR, Belt P et al. Bone marrow engraftment following unrelated donor transplantation utilizing busulfan and cyclophosphamide preparatory chemotherapy Bone Marrow Transplant 1996 17: 479–483
Topolsky D, Crilley P, Styler MJ et al. Unrelated donor bone marrow transplantation without T cell depletion using a chemotherapy only conditioning regimen. Low incidence of failed engraftment and severe acute GVHD Bone Marrow Transplant 1996 17: 549–554
Bertz H, Potthoff K, Mertelsmann R, Finke J . Busulfan/cyclophosphamide in volunteers unrelated donor (VUD) BMT: excellent feasibility and low incidence of treatment-related toxicity Bone Marrow Transplant 1997 19: 1169–1173
Przepiorka KM, Weisdorf D, Martin P . 1994 Consensus Conference on acute GVHD-grading Bone Marrow Transplant 1995 15: 825–828
Bearman SI, Appelbaum FR, Buckner CD et al. Regimen related toxicity in patients undergoing bone marrow transplantation J Clin Oncol 1988 6: 1562–1568
McDonald GB, Hinds MS, Fisher LD et al. Veno-occlusive disease of the liver and multiorgan failure after bone marrow transplantation: a cohort study of 355 patients Ann Intern Med 1993 118: 255–267
Beatty PG, Ash R, Hows JM, McGlave PB . The use of unrelated bone marrow donors in the treatment of chronic myelogenous leukemia: experience of four centers Bone Marrow Transplant 1989 4: 287–290
Santos GW, Tutschka Pj, Brookmeyer R et al. Marrow transplantation for acute non-lymphomic leukemia after treatment with busulfan and cyclophosphamide New Engl J Med 1983 309: 1347–1353
Tutschka PJ, Copelan EA, Klein, JA et al. Bone marrow transplantation for leukemia following new busulfan and cyclophosphamide regime Blood 1987 70: 1382–1388
Sahebi F, Copelan E, Crilley P et al. Unrelated allogeneic bone marrow transplantation using high-dose busulfan and cyclophosphamide for preparative regimen Bone Marrow Transplant 1996 17: 329–333
Ringden O, Ruutu T, Remberger M et al . for the Nordic Bone Marrow Transplantation Group A randomized trial comparing busulfan with total body irradiation as conditioning in allogeneic marrow transplant recipients with leukemia: a report from the Nordic Bone Marrow Transplantion Group Blood 1994 83: 2723–2730
Andersson BS, Giralt S, Tran H et al. I.V. busulfan, cytoxan and allogeneic hematopoietic stem cell transplantation for advanced hematological malignancies Bone Marrow Transplantat 1999 23: (Suppl. 1) 223
Gratwohl A, Hermans J, Niederwieser D et al . for the Chronic Leukemia Working Party of the EBMT Bone marrow transplantation for chronic myeloid leukemia: long terms results Bone Marrow Transplant 1993 12: 509–516
Devergie A, Apperley JF, Labopin M et al for the Chronic Leukemia Working Party of the European Group of Blood and Marrow Transplantation . European results of matched unrelated bone marrow transplantation for chronic myeloid leukemia. Impact of HLA class II matching Bone Marrow Transplant 1997 20: 11–19
Sierra J, Storer B, Hansen J et al. Transplantation of marrow cells from unrelated donors for treatment of high risk acute leukemia: the effect of leukemia burden, donor HLA-matching and marrow cell dose Blood 1997 89: 4226–4235
Kröger N, Zabelina T, Krüger W et al. Anti-human T-lymphocyte globulin (ATG) as part of the conditioning regimen in unrelated bone marrow transplantation of patients with CML in chronic or accelerated phase Blood 1998 92: (Suppl. 1) 578
Zander AR, Zabelina T, Löliger C et al. Bone marrow transplantation from mismatched unrelated donors Bone Marrow Transplant 2000 25: (Suppl. 1) 36
Zander AR, Zabelina T, Kröger N et al. Use of a five-agent GVHD prevention regimen in recipients of unrelated donor marrow Bone Marrow Transplant 1999 23: 889–893
Acknowledgements
We thank the staff of the BMT unit for providing excellent care of our patients and the medical technicians for their excellent work in the BMT laboratory.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kröger, N., Zabelina, T., Krüger, W. et al. Comparison of total body irradiation vs busulfan in combination with cyclophosphamide as conditioning for unrelated stem cell transplantation in CML patients. Bone Marrow Transplant 27, 349–354 (2001). https://doi.org/10.1038/sj.bmt.1702802
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1702802
Keywords
This article is cited by
-
Impact of extended infusional mesna prophylaxis on the incidence of BK viruria and hemorrhagic cystitis following post-transplantation cyclophosphamide and CTLA4Ig-based haploidentical transplantation
Annals of Hematology (2020)
-
Reduced incidence of interstitial pneumonitis after allogeneic hematopoietic stem cell transplantation using a modified technique of total body irradiation
Scientific Reports (2016)
-
Combination of a haploidentical SCT with an unrelated cord blood unit: a single-arm prospective study
Bone Marrow Transplantation (2014)
-
Cardiopulmonary exercise testing prior to myeloablative allo-SCT: a feasibility study
Bone Marrow Transplantation (2014)
-
The risk of polyomavirus BK-associated hemorrhagic cystitis after allogeneic hematopoietic SCT is associated with myeloablative conditioning, CMV viremia and severe acute GVHD
Bone Marrow Transplantation (2014)