Abstract
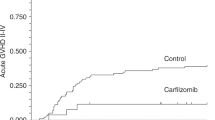
We report the results of a phase III trial comparing tacrolimus (FK506) with cyclosporine for GVHD prophylaxis after allogeneic BMT. From February 1995 to July 1996, 136 patients were enrolled and followed up to September 1997. During the first 100 days post-transplant the incidence of grade II–IV acute GVHD (the primary end-point) was lower in the tacrolimus group (17.5%) compared with the cyclosporine group (48.0%, P < 0.0001). A significant difference was observed between the tacrolimus and cyclosporine groups when subset analyses were performed based on recipients from HLA-matched siblings (13.3% vs 41.3%, P = 0.015) or donors other than HLA-matched siblings (21.4% vs 53.8%, P = 0.0029). The incidence of chronic GVHD (47.3% and 47.8%) and Kaplan–Meier estimate of overall survival (62.9% and 65.2%) were similar between the tacrolimus and cyclosporine groups, respectively. The overall leukemia relapse rate was not significantly different between the tacrolimus and cyclosporine groups (19.6% and 11.4%, respectively). However, the relapse rate among recipients from HLA-matched siblings was significantly higher in the tacrolimus group (30.9%) compared with the cyclosporine group (3.6%, P = 0.013). These results suggest the merit of tacrolimus for the prophylaxis of acute GVHD, but a lack of merit for a graft-versus-leukemia effect among recipients from HLA-matched sibling donors. Bone Marrow Transplantation (2001) 28, 181–185.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kino T, Hatanaka H, Miyata S et al. FK506: a novel immunosuppressant isolated from a streptomyces: I. Fermentation, isolation, and physicochemical and biological characteristics J Antibiot 1987 40: 1249–1255
Kino T, Hatanaka H, Miyata S et al. FK506: a novel immunosuppressant isolated from a streptomyces: II. Immunosuppressive effect of FK506 in vitro J Antibiot 1987 40: 1256–1265
Starzl TE, Todo S, Fung J et al. FK506 for liver, kidney and pancreas transplantation Lancet 1989 2: 1000–1004
Blazar BR, Taylor PA, Fitzsimmons WE, Vallera DE . FK506 inhibits graft-versus-host disease and bone marrow graft rejection in murine recipients of MHC disparate donor grafts by interfering with mature peripheral T cell expansion post-transplantation J Immunol 1994 153: 1836–1846
Markus PM, Cai X, Ming W et al. Prevention of graft-versus-host disease following allogeneic bone marrow transplantation in rats using FK506 Transplantation 1991 52: 590–594
Yu C, Storb R, Deeg HJ et al. Tacrolimus (FK506) and methotrexate regimens to prevent graft-versus-host disease after unrelated dog leukocyte antigen (DLA) nonidentical marrow transplantation Bone Marrow Transplant 1996 17: 649–653
Storb R, Raff RF, Appelbaum FR et al. FK-506 and methotrexate prevent graft-versus-host disease in dogs given 9.2 Gy total body irradiation and marrow grafts from unrelated dog leukocyte antigen-nonidentical donors Transplantation 1993 56: 800–807
Kanamaru A, Takemoto Y, Kakishita E et al. for the Japanese FK506 BMT Study Group FK506 treatment of graft-versus-host disease developing or exacerbating during prophylaxis and therapy with cyclosporin and/or other immunosuppressants Bone Marrow Transplant 1995 15: 885–889
Masaoka T, Shibata H, Kakishita E et al. Phase II study of FK506 for allogeneic bone marrow transplantation Transplant Proc 1991 23: 3228–3231
Hiraoka A, Takemoto Y, Kanamaru A et al . for the Japanese FK506 BMT Study Group Use of FK506 for prophylaxis of acute graft-versus-host disease after allogeneic bone marrow transplantation: an early phase II study Transplant Now 1997 10: 593–604
Kanamaru A, Okamoto S, Moriyama Y et al . for the Japanese FK506 BMT Study Group FK506 therapy for the prevention of acute graft-versus-host disease after allogeneic bone marrow transplantation: a late phase II study Transplant Now 1998 11: 367–380
Thomas ED, Storb R, Clift RA et al. Bone-marrow transplantation New Engl J Med 1975 292: 895–902
Shulman HM, Sullivan KM, Weiden PL et al. Chronic graft-versus-host syndrome in man. A long-term clinicopathologic study of 20 Seattle patients Am J Med 1980 69: 204–217
Ratanatharathorn V, Nash RA, Przepiorka D et al. Phase III study comparing methotrexate and tacrolimus (Prograf, FK506) with methotrexate and cyclosporine for graft-versus-host disease prophylaxis after HLA-identical sibling bone marrow transplantation Blood 1998 92: 2303–2314
Nash RA, Antin JH, Karanes C et al. Phase 3 study comparing methotrexate and tacrolimus with methotrexate and cyclosporine for prophylaxis of acute graft-versus-host disease after marrow transplantation from unrelated donors Blood 2000 96: 2062–2068
Morishima Y . Results of unrelated bone marrow transplantation through Japan marrow donor program Hematol Oncol 1997 35: 197–203
Nagoya Bone Marrow Transplantation Study Group . Bone marrow transplantation in Nagoya Bone Marrow Transplantation Study Group In: Morishita Y, Horibe K, Morishima Y (eds) Manual of Blood and Marrow Transplantation Nihon Igakukan: Tokyo 1995 423–435
Porter DL, Antin JH . The graft-versus-leukemia effects of allogeneic cell therapy Ann Rev Med 1999 50: 369–386
Acknowledgements
The authors thank the investigators of centers participating in the Japanese FK506 BMT Study Group for their collaboration: Dr M Asaka (Hokkaido University), Dr M Kasai (Sapporo Hokuyu Hospital), Dr N Maseki (Saitama Cancer Center), Dr Y Saitoh (Chiba University), Drs S Takamoto, T Sasaki and Y Onozawa (Tokyo Metro Komagome Hospital), Dr Y Ikeda (Keio University), Dr Y Ichikawa (Tokai University), Dr H Kigasawa (Kanagawa Children's Medical Center), Dr Y Aisawa (Niigata University), Dr N Hirabayashi (Nagoya Daini Red Cross Hospital), Dr H Sao (Meitetsu Hospital), Dr T Matsuda (Kanazawa University), Dr T Yoshida (Toyama Pref. Central Hospital), Dr K Kawa-Ha (Osaka Medical Center and Res Inst for Maternal and Child Health), Dr A Horiuchi (Kinki University), Dr E Kakishita (Hyogo College of Medicine), Dr Y Niho (Kyushu Univesity), Dr S Hayashi (Sanshinkai Hara Hospital), and Dr M Tomonaga (Nagasaki University).
Author information
Authors and Affiliations
Additional information
for the Japanese FK506 BMT Study Group
Rights and permissions
About this article
Cite this article
Hiraoka, A., Ohashi, Y., Okamoto, S. et al. Phase III study comparing tacrolimus (FK506) with cyclosporine for graft-versus-host disease prophylaxis after allogeneic bone marrow transplantation. Bone Marrow Transplant 28, 181–185 (2001). https://doi.org/10.1038/sj.bmt.1703097
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703097
Keywords
This article is cited by
-
Risk factors for adverse outcomes following haploidentical hematopoietic cell transplantation with posttransplant cyclophosphamide: a two-center analysis
Annals of Hematology (2022)
-
Cyclosporine/methotrexate versus tacrolimus/methotrexate with or without anti-thymocyte globulin as GVHD prophylaxis in adult patients with aplastic anemia
Annals of Hematology (2021)
-
Effects of red blood cell concentrate transfusion on blood tacrolimus concentration
International Journal of Clinical Pharmacy (2020)
-
Low mean post-transplantation tacrolimus levels in weeks 2–3 correlate with acute graft-versus-host disease in allogeneic hematopoietic stem cell transplantation from related and unrelated donors
Bone Marrow Transplantation (2019)
-
Analysis of the variable factors influencing tacrolimus blood concentration during the switch from continuous intravenous infusion to oral administration after allogeneic hematopoietic stem cell transplantation
International Journal of Hematology (2017)