Abstract
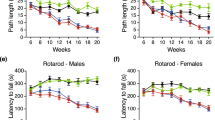
Huntington's disease is an autosomal-dominant progressive neurodegenerative disorder resulting in specific neuronal loss and dysfunction in the striatum and cortex1. The disease is universally fatal, with a mean survival following onset of 15–20 years and, at present, there is no effective treatment. The mutation in patients with Huntington's disease is an expanded CAG/polyglutamine repeat in huntingtin, a protein of unknown function with a relative molecular mass of 350,000 (M r 350K)2. The length of the CAG/polyglutamine repeat is inversely correlated with the age of disease onset. The molecular pathways mediating the neuropathology of Huntington's disease are poorly understood. Transgenic mice expressing exon 1 of the human huntingtin gene with an expanded CAG/polyglutamine repeat develop a progressive syndrome with many of the characteristics of human Huntington's disease3. Here we demonstrate evidence of caspase-1 activation in the brains of mice and humans with the disease. In this transgenic mouse model of Huntington's disease, expression of a dominant-negative caspase-1 mutant extends survival and delays the appearance of neuronal inclusions, neurotransmitter receptor alterations and onset of symptoms, indicating that caspase-1 is important in the pathogenesis of the disease. In addition, we demonstrate that intracerebroventricular administration of a caspase inhibitor delays disease progression and mortality in the mouse model of Huntington's disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Harper, P. S. Huntington's Disease (Saunders, London, (1991).
Huntington's Disease Collaborative Research Group. Anovel gene containing a trinucleotide repeat that is unstable on Huntington's disease chromosomes. Cell 72, 971–983 (1993).
Mangiarini, L. et al. Exon 1 of the HD gene with an expanded CAG repeat is sufficient to cause a progressive neurological phenotype in transgenic mice. Cell 87, 493–506 ( 1996).
Davies, S. W. et al. Formation of neuronal intranuclear inclusions underlies the neurological dysfunction in mice transgenic for the HD mutation. Cell 90, 537–548 ( 1997).
Cha, J. H. et al. Altered brain neurotransmitter receptors in transgenic mice expressing a portion of an abnormal human huntington disease gene. Proc. Natl Acad. Sci. USA 95, 6480–6485 (1998).
DiFiglia, M. et al. Aggregation of huntingtin in neuronal intranuclear inclusions and dystrophic neurites in brain. Science 277, 1990–1993 (1997).
Alnemri, E. S. et al. Human ICE/CED-3 protease nomenclature. Cell 87, 171 (1996).
Friedlander, R. M. et al. Expression of a dominant negative mutant of interleukin-1 beta converting enzyme in transgenic mice prevents neuronal cell death induced by trophic factor withdrawal and ischemic brain injury. J. Exp. Med. 185, 933–940 ( 1997).
Forss-Petter, S. et al. Transgenic mice expressing beta-galactosidase in mature neurons under neuron-specific enolase promoter control. Neuron 5, 187–197 (1996).
Burne, J. F., Staple, J. K. & Raff, M. C. Glial cells are increased proportionally in transgenic optic nerves with increased numbers of axons. J. Neurosc. 16, 2064–2073 (1996).
Hara, H. et al. Attenuation of transient focal cerebral ischemic injury in transgenic mice expressing a mutant ICE inhibitory protein. J. Cereb. Blood Flow Metab. 17, 370–375 (1997).
Friedlander, R. M., Brown, R. H., Gagliardini, V., Wang, J. & Yuan, J. Inhibition of ICE slows ALS in mice. Nature 388, 31 (1997 ).
Li, P. et al. Mice deficient in IL-1 beta-converting enzyme are defective in production of mature IL-1 beta and resistant to endotoxic shock. Cell 80, 401–411 ( 1995).
Saudou, F., Finkbeiner, S., Devys, D. & Greenberg, M. E. Huntingtin acts in the nucleus to induce apoptosis but death does not correlate with the formation of intranuclear inclusions. Cell 95, 55–66 (1998).
Wellington, C. L. et al. Caspase cleavage of gene products associated with triplet expansion disorders generates truncated fragments containing the polyglutamine tract. J. Biol. Chem. 273, 9158– 9167 (1998).
Persichetti, F. et al. Differential expression of normal and mutant Huntington's disease gene alleles. Neurobiol. Dis. 3, 183–190 (1996).
Kosinski, C. M. et al. Huntingtin immunoreactivity in the rat neostriatum: differential accumulation in the projection and interneurons. Exp. Neurol. 144, 239–247 (1997).
Kim, M. et al. Mutant huntingtin expression in clonal striatal cells: dissociation of inclusion formation and neuronal survival by caspase inhibition. J. Neurosci. 19, 964–973 (1999).
Portera-Cailliau, C., Hedreen, J. C., Price, D. L. & Koliatsos, V. E. Evidence for apoptotic cell death in Huntington disease and excitotoxic animal models. J. Neurosci. 15, 3775– 3787 (1995).
Lunkes, A. & Mandel, J. L. Acellular model that recapitulates major pathogenic steps of Huntington's disease. Hum. Mol. Genet. 7, 1355–1361 ( 1998).
Reddy, P. H. et al. Behavioural abnormalities and selective neuronal loss in HD transgenic mice expressing mutated full-length HD cDNA. Nature Genet. 20, 198–202 ( 1998).
Hurlbert, M. S. et al. Mice transgenic for an expanded CAG repeat in Huntington's disease gene develop diabetes. Diabetes 8, 649–651 (1999).
Hara, H. et al. Inhibition of interleukin 1beta converting enzyme family proteases reduces ischemic and excitotoxic neuronal damage. Proc. Natl Acad. Sci. USA 94, 2007–2012 ( 1997).
Goldberg, Y. P. et al. Cleavage of huntingtin by apopain, a proapoptotic cysteine protease, is modulated by the polyglutamine tract. Nature Genet. 13, 442–449 ( 1996).
Tewari, M. et al. Yama/CPP32 beta, a mammalian homolog of CED-3, is a CrmA-inhibitable protease that cleaves the death substrate poly(ADP-ribose) polymerase. Cell 81, 801–809 ( 1995).
Thornberry, N. A. et al. Anovel heterodimeric cysteine protease is required for interleukin-1 beta processing in monocytes. Nature 356, 768–774 (1992).
Troy, C. M., Stefanis, L., Prochiantz, A., Greene, L. A. & Shelanski, M. L. The contrasting roles of ICE family proteases and interleukin-1beta in apoptosis induced by trophic factor withdrawal and by copper/zinc superoxide dismutase down-regulation. Proc. Natl Acad. Sci. USA 93, 5635– 5640 (1996).
Friedlander, R. M., Gagliardini, V., Rotello, R. J. & Yuan, J. Functional role of interleukin 1 beta (IL-1 beta) in IL-1 beta-converting enzyme-mediated apoptosis. J. Exp. Med. 184, 717–724 (1996).
Lee, S. C., Dickson, D. W. & Brosnan, C. F. Interleukin-1, nitric oxide and reactive astrocytes. Brain Behav. Immun. 9, 345– 354 (1995).
Klivenyi, P., Andreassen, O., Friedlander, R. M. & Beal, M. F. Transgenic mice expressing a dominant negative mutant interleukin-1b converting enzyme shows resistance to MPTP neurotoxicity. NeuroReport 10, 635–638 (1999).
Acknowledgements
We thank E. Signer for advice and support; C. Swap, A. Dunah and W. Hobbs for expert technical assistance; M.MacDonald for providing Huntingtin antibodies; and E. Balodimas for editorial assistance. This work was supported by a grant from the Hereditary Disease Foundation (to R.M.F. and J.H.J.C.), the Huntington's Disease Society of America (to J.-H.J.C.), the Glendorn Foundation (to A.B.Y.) and by grants from the NIH (to J.-H.J.C., A.B.Y. and J.Y.). This paper is dedicated to J.B.P., who died during the review of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ona, V., Li, M., Vonsattel, J. et al. Inhibition of caspase-1 slows disease progression in a mouse model of Huntington's disease. Nature 399, 263–267 (1999). https://doi.org/10.1038/20446
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/20446
This article is cited by
-
Longitudinal modeling of human neuronal aging reveals the contribution of the RCAN1–TFEB pathway to Huntington’s disease neurodegeneration
Nature Aging (2023)
-
A selective inhibitor of the NLRP3 inflammasome as a potential therapeutic approach for neuroprotection in a transgenic mouse model of Huntington’s disease
Journal of Neuroinflammation (2022)
-
Age-related Huntington’s disease progression modeled in directly reprogrammed patient-derived striatal neurons highlights impaired autophagy
Nature Neuroscience (2022)
-
Inflammasome Signaling in the Aging Brain and Age-Related Neurodegenerative Diseases
Molecular Neurobiology (2022)
-
A soluble truncated tau species related to cognitive dysfunction and caspase-2 is elevated in the brain of Huntington’s disease patients
Acta Neuropathologica Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.