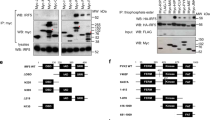
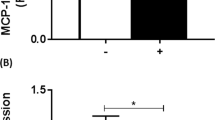
Abstract
NF-κB comprises a family of cellular transcription factors that are involved in the inducible expression of a variety of cellular genes that regulate the inflammatory response1,2. NF-κB is sequestered in the cytoplasm by inhibitory proteins, IκB, which are phosphorylated by a cellular kinase complex known as IKK. IKK is made up of two kinases, IKK-α and IKK-β, which phosphorylate IκB, leading to its degradation and translocation of NF-κB to the nucleus3,4,5,6,7,8,9. IKK kinase activity is stimulated when cells are exposed to the cytokine TNF-α or by overexpression of the cellular kinases MEKK1 and NIK10,11. Here we demonstrate that the anti-inflammatory agents aspirin and sodium salicylate specifically inhibit IKK-β activity in vitro and in vivo. The mechanism of aspirin and sodium salicylate inhibition is due to binding of these agents to IKK-β to reduce ATP binding. Our results indicate that the anti-inflammatory properties of aspirin and salicylate are mediated in part by their specific inhibition of IKK-β, thereby preventing activation by NF-κB of genes involved in the pathogenesis of the inflammatory response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Baeuerle, P. A. & Baltimore, D. NF-κB: Ten years after. Cell 87, 13–20 (1996).
Baldwin, J. A. S. The NF-κB and IκB proteins: new discoveries and insights. Annu. Rev. Immunol. 14, 649–681 (1996).
Brown, K., Gerstberger, S., Carlson, L., Franzoso, G. & Siebenlist, U. Control of IκBα proteolysis by site specific signal induced phosphorylation. Science 267, 1485–1488 (1995).
Chen, Z.et al. Signal-induced site-specific phosphorylation targets IκBα to the ubiquitin–proteasome pathway. Genes Dev. 9, 1586–1597 (1995).
DiDonato, J. A., Hayakawa, M., Rothwarf, D. M., Zandi, E. & Karin, M. Acytokine-responsive IκB kinase that activates the transcription factor NF-κB. Nature 388, 548–554 (1997).
Régnier, C. H.et al. Identification and characterization of an IκB kinase. Cell 90, 373–383 (1997).
Mercurio, F.et al. IKK-1 and IKK-2: cyotkine-activated IκB kinases essential for NF-κB activation. Science 278, 860–866 (1997).
Zandi, E., Rothwarf, D. M., Delhase, M., Hayakawa, M. & Karin, M. The IκB kinase complex (IKK) contains two kinase subunits, IKKα and IKKβ, necessary for IκB phosphorylation and NF-κB activation. Cell 91, 243–252 (1997).
Woronicz, J. D., Gao, X., Cao, Z., Rothe, M. & Goeddel, D. V. IκB kinase-β: NF-κB activation and complex formation with IκB kinase-α and NIK. Science 278, 866–869 (1997).
Malinin, N. L., Boldin, M. P., Kovalenko, A. V. & Wallach, D. MAP3K-related kinase involved in NF-κB induction by TNF, CD95 and IL-1. Nature 385, 540–544 (1997).
Yin, M.-J.et al. HTLV-I Tax protein binds to MEKK1 to stimulate IκB kinase activity and NF-κB activation. Cell 93, 875–884 (1998).
Vane, J. R. Inhibition of prostaglandin synthesis as a mechanism of action of the aspirin-like drugs. Nature New Biol. 231, 232–235 (1971).
Vane, J. Towards a better aspirin. Nature 367, 215–216 (1994).
Kopp, E. & Ghosh, S. Inhibition of NF-κB by sodium salicylate and aspirin. Science 265, 956–959 (1994).
Grilli, M., Pizzi, M., Memo, M. & Spano, P. Neuroprotection by aspirin and sodium salicylate through blockade of NF-κB activation. Science 274, 1383–1385 (1996).
Rothe, M., Sarma, V., Dixit, V. M. & Goeddel, D. V. TRAF2-mediated activation of NF-κB by TNF receptor 2 and CD40. Science 269, 1424–1427 (1995).
Insel, P. The Pharmacological Basis of Therapeutics(eds Harman, J. G., Molinoff, P. B., Rudden, R. W. & Gilman, A. G.) (McGraw-Hill, New York, (1996)).
Auphan, N., DiDonato, J. A., Rosette, C., Helmberg, A. & Karin, M. Immunosuppression by glucocorticoids: Inhibition of NF-κB activity through induction of IκB synthesis. Science 270, 286–290 (1995).
Scheinman, R. I., Cogswell, P. C., Lofquist, A. K. & Baldwin, A. S. Role of transcriptional activation of IκBα in mediation of immunosuppression by glucocorticoids. Science 270, 283–286 (1995).
Tan, T. H., Jia, R. & Roeder, R. G. Utilization of signal transduction pathway by the human T-cell leukemia virus type I transcriptional activator tax. J. Virol. 63, 3761–3768 (1989).
Boulton, T. G.et al. ERKs: a family of protein-serine/threonine kinases that are activated and tyrosine phosphorylated in response to insulin and NGF. Cell 65, 663–675 (1991).
Khokhlatchev, A.et al. Reconstitution of mitogen-activated protein kinase phosphorylation cascades in bacteria. Efficient synthesis of active protein kinases. J. Biol. Chem. 272, 11057–11062 (1997).
Xu, S. & Cobb, M. H. MEKK1 binds directly to the c-Jun N-terminal kinases/stress-activated protein kinases. J. Biol. Chem. 272, 32056–32060 (1997).
Kyriakis, J. M.et al. The stress-activated protein kinase subfamily of c-Jun kinases. Nature 369, 156–160 (1994).
Mitchell, J. A., Akarasereenont, P., Thiermermann, C., Flower, R. J. & Vane, J. R. Selectivity of nonsteroidal antiinflammatory drugs as inhibitors of constitutive and inducible cyclooxygenase. Proc. Natl Acad. Sci. USA 90, 11693–11697 (1994).
Kalgutkar, A. S.et al. Aspirin-like molecule that covalently inactivates cyclooxygenase-2. Science 280, 1268–1270 (1998).
Acknowledgements
We thank F. Mercurio for IKK-α and IKK-β cDNAs, M. Cobb and E. Ross for discussion, and S. Johnson and E. Pasternack respectively for preparing the manuscript and figures. This work was supported by grants from the NIH, the Council for Tobacco Research and the Robert Welch Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yin, MJ., Yamamoto, Y. & Gaynor, R. The anti-inflammatory agents aspirin and salicylate inhibit the activity of IκB kinase-β. Nature 396, 77–80 (1998). https://doi.org/10.1038/23948
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/23948
This article is cited by
-
Large-scale computational modelling of the M1 and M2 synovial macrophages in rheumatoid arthritis
npj Systems Biology and Applications (2024)
-
The role of non-steroidal anti-inflammatory drugs as adjuncts to periodontal treatment and in periodontal regeneration
Journal of Translational Medicine (2023)
-
Costunolide alleviates atherosclerosis in high-fat diet-fed ApoE−/− mice through covalently binding to IKKβ and inhibiting NF-κB-mediated inflammation
Acta Pharmacologica Sinica (2023)
-
Osteoarthritis: pathogenic signaling pathways and therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
New insights into the properties, functions, and aging of skeletal stem cells
Osteoporosis International (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.