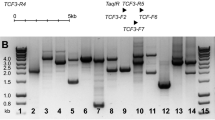
Abstract
Several studies have shown that quantitative detection of minimal residual disease (MRD) predicts clinical outcome in childhood acute lymphoblastic leukemia (ALL). In this report we investigated the applicablility of T cell receptor gamma (TCRG) gene rearrangements as targets for MRD detection by real-time quantitative PCR analysis. Seventeen children with precursor-B-ALL and 15 children with T-ALL were included in this study. Using an allele-specific (ASO) forward primer in combination with germline Jγ reverse primers and Jγ TaqMan probes, a reproducible sensitivity of ⩽10−4 (defined by strict criteria) was obtained in only four out of 19 (21%) TCRG gene rearrangements in precursor-B-ALL patients and in 10 out of 15 (67%) TCRG gene rearrangements in T-ALL patients. The main reason for not obtaining a reproducible sensitivity of ⩽10−4 in approximately 60% of cases was the non-specific amplification of TCRG gene rearrangements in normal T-lymphocytes. A maximal sensitivity of ⩽10−4 (defined by less strict criteria) was obtained in 42% of TCRG gene rearrangements in precursor-B-ALL patients. The number of inserted nucleotides was significantly higher in T-ALL (mean: 8.5) as compared to precursor-B-ALL (mean: 6.8) and appeared to be the most important predictor for reaching a reproducible sensitivity ⩽10−4. The usage of a touchdown PCR or the usage of an ASO reverse primer in combination with Vγ member forward primers and TaqMan probes did not clearly improve the overall results. Nevertheless, RQ-PCR analysis of TCRG gene rearrangements in follow-up samples obtained from 12 ALL patients showed the applicability of this method for MRD detection. We conclude that RQ-PCR analysis of TCRG gene rearrangements can be used for the detection of MRD, but that sensitivities might be limited due to non-specific amplification. This method is applicable in the majority of T-ALL patients and in almost half of precursor-B-ALL patients, particularly when used as second-choice target for confirmation of the MRD results obtained via the first-choice target.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Szczepanski T, Orfao A, van der Velden VHJ, San Miguel JF, van Dongen JJM . Minimal residual disease in leukaemia patients Lancet Oncology 2001 2: 409–417
Cave H, van der Werff ten Bosch J, Suciu S, Guidal C, Waterkeyn C, Otten J, Bakkus M, Thielemans K, Grandchamp B, Vilmer E . Clinical significance of minimal residual disease in childhood acute lymphoblastic leukemia. European Organization for Research and Treatment of Cancer – Childhood Leukemia Cooperative Group N Engl J Med 1998 339: 591–598
Coustan-Smith E, Behm FG, Sanchez J, Boyett JM, Hancock ML, Raimondi SC, Rubnitz JE, Rivera GK, Sandlund JT, Pui CH, Campana D . Immunological detection of minimal residual disease in children with acute lymphoblastic leukaemia Lancet 1998 351: 550–554
van Dongen JJM, Seriu T, Panzer-Grümayer ER, Biondi A, Pongers-Willemse MJ, Corral L, Stolz F, Schrappe M, Masera G, Kamps WA, Gadner H, van Wering ER, Ludwig WD, Basso G, de Bruijn MA, Cazzaniga G, Hettinger K, van der Does-van den Berg A, Hop WC, Riehm H, Bartram CR . Prognostic value of minimal residual disease in acute lymphoblastic leukaemia in childhood Lancet 1998 352: 1731–1738
Knechtli CJ, Goulden NJ, Hancock JP, Grandage VL, Harris EL, Garland RJ, Jones CG, Rowbottom AW, Hunt LP, Green AF, Clarke E, Lankester AW, Cornish JM, Pamphilon DH, Steward CG, Oakhill A . Minimal residual disease status before allogeneic bonemarrow transplantation is an important determinant of successfuloutcome for children and adolescents with acute lymphoblasticleukemia Blood 1998 92: 4072–4079
van der Velden VHJ, Joosten SA, Willemse MJ, van Wering ER, Lankester A, van Dongen JJM, Hoogerbrugge PM . Real-time quantitative PCR for detection of minimal residual disease before allogeneic stem cell transplantation predicts outcome in children with acute lymphoblastic leukemia Leukemia 2001 15: 1485–1487
Davis MM, Bjorkman PJ . T-cell antigen receptor genes and T-cell recognition Nature 1988 334: 395–402
Szczepanski T, Langerak AW, Willemse MJ, Wolvers-Tettero IL, van Wering ER, van Dongen JJM . T cell receptor gamma (TCRG) gene rearrangements in T cell acute lymphoblastic leukemia reflect ‘end-stage’ recombinations: implications for minimal residual disease monitoring Leukemia 2000 14: 1208–1214
Szczepanski T, Beishuizen A, Pongers-Willemse MJ, Hahlen K, Van Wering ER, Wijkhuijs AJ, Tibbe GJ, De Bruijn MA, Van Dongen JJM . Cross-lineage T cell receptor gene rearrangements occur in more than ninety percent of childhood precursor-B acute lymphoblastic leukemias: alternative PCR targets for detection of minimal residual disease Leukemia 1999 13: 196–205
Pongers-Willemse MJ, Verhagen OJ, Tibbe GJ, Wijkhuijs AJ, de Haas V, Roovers E, van der Schoot CE, van Dongen JJM . Real-time quantitative PCR for the detection of minimal residual disease in acute lymphoblastic leukemia using junctional region specific TaqMan probes Leukemia 1998 12: 2006–2014
Verhagen O, Willemse M, Breunis W, Wijkhuis A, Jacobs D, Joosten S, van Wering E, van Dongen JJM, van der Schoot C . Application of germline IGH probes in real-time quantitative PCR for the detection of minimal residual disease in acute lymphoblastic leukemia Leukemia 2000 14: 1426–1435
Brüggemann M, Droese J, Bolz I, Luth P, Pott C, von Neuhoff N, Scheuering U, Kneba M . Improved assessment of minimal residual disease in B cell malignancies using fluorogenic consensus probes for real-time quantitative PCR Leukemia 2000 14: 1419–1425
Donovan JW, Ladetto M, Zou G, Neuberg D, Poor C, Bowers D, Gribben JG . Immunoglobulin heavy-chain consensus probes for real-time PCR quantification of residual disease in acute lymphoblastic leukemia Blood 2000 95: 2651–2658
Nakao M, Janssen JW, Flohr T, Bartram CR . Rapid and reliable quantification of minimal residual disease in acute lymphoblastic leukemia using rearranged immunoglobulin and T-cell receptor loci by LightCycler technology Cancer Res 2000 60: 3281–3289
Kwan E, Norris MD, Zhu L, Ferrara D, Marshall GM, Haber M . Simultaneous detection and quantification of minimal residual disease in childhood acute lymphoblastic leukaemia using real-time polymerase chain reaction Br J Haematol 2000 109: 430–434
van der Velden VHJ, Willemse MJ, van der Schoot CE, van Wering ER, van Dongen JJM . Immunoglobulin kappa deleting element rearrangements in precursor-B-acute lymphoblastic leukemia are stable targets for detection of minimal residual disease by real-time quantitative PCR Leukemia 2002 (in press)
Macintyre EA, d'Auriol L, Duparc N, Leverger G, Galibert F, Sigaux F . Use of oligonucleotide probes directed against T cell antigen receptor gamma delta variable-(diversity)-joining junctional sequences as a general method for detecting minimal residual disease in acute lymphoblastic leukemias J Clin Invest 1990 86: 2125–2135
Breit TM, Wolvers-Tettero IL, Hahlen K, van Wering ER, van Dongen JJM . Extensive junctional diversity of gamma delta T-cell receptors expressed by T-cell acute lymphoblastic leukemias: implications for the detection of minimal residual disease Leukemia 1991 5: 1076–1086
Landman-Parker J, Aubin J, Delabesse E, Tabone MD, Adam M, Millien C, Leboeuf D, Buzyn-Veil A, Dollfus C, Leverger G, Macintyre EA . Simplified strategies for minimal residual disease detection in B-cell precursor acute lymphoblastic leukaemia Br J Haematol 1996 95: 281–290
Xue W, Luo S, Adler WH, Schulze DH, Berman JE . Immunoglobulin heavy chain junctional diversity in young and aged humans Hum Immunol 1997 57: 80–92
Beishuizen A, de Bruijn MA, Pongers-Willemse MJ, Verhoeven MA, van Wering ER, Hählen K, Breit TM, de Bruin-Versteeg S, Hooijkaas H, van Dongen JJM . Heterogeneity in junctional regions of immunoglobulin kappa deleting element rearrangements in B cell leukemias: a new molecular target for detection of minimal residual disease Leukemia 1997 11: 2200–2207
van Wering ER, van der Linden-Schrever BE, van der Velden VHJ, Szczepanski T, van Dongen JJM . T-lymphocytes in bone marrow samples of children with acute lymphoblastic leukemia during and after chemotherapy might hamper PCR-based minimal residual disease studies Leukemia 2001 15: 1301–1303
Verhagen OJ, Wijkhuijs AJ, van der Sluijs-Gelling AJ, Szczepanski T, van der Linden-Schrever BE, Pongers-Willemse MJ, van Wering ER, van Dongen JJM, van der Schoot CE . Suitable DNA isolation method for the detection of minimal residual disease by PCR techniques Leukemia 1999 13: 1298–1299
Pongers-Willemse MJ, Seriu T, Stolz F, d'Aniello E, Gameiro P, Pisa P, Gonzalez M, Bartram CR, Panzer-Grümayer ER, Biondi A, San Miguel JF, van Dongen JJM . Primers and protocols for standardized detection of minimal residual disease in acute lymphoblastic leukemia using immunoglobulin and T cell receptor gene rearrangements and TAL1 deletions as PCR targets: report of the BIOMED-1 Concerted Action: investigation of minimal residual disease in acute leukemia Leukemia 1999 13: 110–118
Langerak AW, Szczepanski T, van der Burg M, Wolvers-Tettero IL, van Dongen JJM . Heteroduplex PCR analysis of rearranged T cell receptor genes for clonality assessment in suspect T cell proliferations Leukemia 1997 11: 2192–2199
van Dongen JJM, Wolvers-Tettero IL . Analysis of immunoglobulin and T cell receptor genes. Part I: Basic and technical aspects Clin Chim Acta 1991 198: 1–91
Delabesse E, Burtin ML, Millien C, Madonik A, Arnulf B, Beldjord K, Valensi F, Macintyre EA . Rapid, multifluorescent TCRG Vgamma and Jgamma typing: application to T cell acute lymphoblastic leukemia and to the detection of minor clonal populations Leukemia 2000 14: 1143–1152
Nyvold C, Madsen HO, Ryder LP, Seyfarth J, Engel CA, Svejgaard A, Wesenberg F, Schmiegelow K . Competitive PCR for quantification of minimal residual disease in acute lymphoblastic leukaemia J Immunol Meth 2000 233: 107–118
Breit TM, Wolvers-Tettero IL, Beishuizen A, Verhoeven MA, van Wering ER, van Dongen JJM . Southern blot patterns, frequencies, and junctional diversity of T-cell receptor-delta gene rearrangements in acute lymphoblastic leukemia Blood 1993 82: 3063–3074
Beishuizen A, Hählen K, Hagemeijer A, Verhoeven MA, Hooijkaas H, Adriaansen HJ, Wolvers-Tettero IL, van Wering ER, van Dongen JJM . Multiple rearranged immunoglobulin genes in childhood acute lymphoblastic leukemia of precursor B-cell origin Leukemia 1991 5: 657–667
Beishuizen A, Verhoeven MA, Mol EJ, van Dongen JJM . Detection of immunoglobulin kappa light-chain gene rearrangement patterns by Southern blot analysis Leukemia 1994 8: 2228–2236
Acknowledgements
We gratefully acknowledge Annella Boon for secretarial assistance, Jacqueline van Valen and Phary Hart for technical assistance, Professor Rob Benner for continuous support, and Marieke Comans-Bitter for preparation of the figures. We thank the pediatric oncologists of the Department of Pediatrics, Sophia Children's Hospital, Rotterdam, for the collection of samples at diagnosis and during follow-up. We acknowledge the Dutch Childhood Leukemia Study Group for kindly providing additional ALL cell samples. Board members of the DCLSG are PJ van Dijken, K Hählen, WA Kamps, ETh Korthof, FAE Nabben, A Postma, JA Rammeloo, GAM de Vaan, AJP Veerman and RS Weening. This study was supported by the Dutch Cancer Society/Koningin Wilhelmina Fonds (grants: SNWLK 97–1567 and SNWLK 2000–2268).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
van der Velden, V., Wijkhuijs, J., Jacobs, D. et al. T cell receptor gamma gene rearrangements as targets for detection of minimal residual disease in acute lymphoblastic leukemia by real-time quantitative PCR analysis. Leukemia 16, 1372–1380 (2002). https://doi.org/10.1038/sj.leu.2402515
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402515
Keywords
This article is cited by
-
GvL effects in T-prolymphocytic leukemia: evidence from MRD kinetics and TCR repertoire analyses
Bone Marrow Transplantation (2017)
-
Next-generation sequencing indicates false-positive MRD results and better predicts prognosis after SCT in patients with childhood ALL
Bone Marrow Transplantation (2017)
-
Prognostic value and clinical significance of TCR rearrangements for MRD monitoring in ALL patients
Comparative Clinical Pathology (2017)
-
Minimal residual disease in acute lymphoblastic leukemia: optimal methods and clinical relevance, pitfalls and recent approaches
Medical Oncology (2014)
-
Improved flow cytometric detection of minimal residual disease in childhood acute lymphoblastic leukemia
Leukemia (2013)