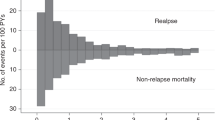
Abstract
Allogeneic blood and marrow transplantation (BMT) is curative for many patients with high-risk and relapsed acute lymphoblastic leukemia (ALL). However, relapse is an important cause of post-transplantation failure, and there are no reliable markers to predict relapse. A retrospective review of patients with ALL who underwent matched related allogeneic BMT was carried out to examine whether the rate of lymphocyte recovery after transplantation had any prognostic value in ALL. The absolute lymphocyte count (ALC) at days 21 and 30 after transplantation was obtained for 43 patients who received transplants during an 18-year period. Patients with an ALC of 175 × 106/l or less on day 21 were more likely to relapse than those with ALC greater than 175 × 106/l (relative risk, 4; 95% confidence interval, 1.5–11.2). Patients with slower lymphocyte recovery had significantly lower relapse-free survival than those with faster recovery (P=0.0028). There was also a trend toward poorer overall survival among those with a slow lymphocyte recovery (log-rank test; P=0.028). The rate of lymphocyte recovery is prognostic in patients with ALL undergoing allogeneic BMT, and this should be integrated with other predictors to identify patients at high risk of relapse. Such patients could be considered for interventions aimed at prevention of relapse, including rapid withdrawal of immunosuppressive medication or donor lymphocyte infusion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Cortes JE, Kantarjian HM . Acute lymphoblastic leukemia. A comprehensive review with emphasis on biology and therapy. Cancer 1995; 76: 2393–2417.
Butturini A, Gale RP . Chemotherapy versus transplantation in acute leukaemia. Br J Haematol 1989; 72: 1–8.
Horowitz MM, Messerer D, Hoelzer D, Gale RP, Neiss A, Atkinson K et al. Chemotherapy compared with bone marrow transplantation for adults with acute lymphoblastic leukemia in first remission. Ann Intern Med 1991; 115: 13–18.
Sebban C, Lepage E, Vernant JP, Gluckman E, Attal M, Reiffers J et al for the French Group of Therapy of Adult Acute Lymphoblastic Leukemia. Allogeneic bone marrow transplantation in adult acute lymphoblastic leukemia in first complete remission: a comparative study. J Clin Oncol 1994; 12: 2580–2587.
Barrett AJ, Horowitz MM, Pollock BH, Zhang MJ, Bortin MM, Buchanan GR et al. Bone marrow transplants from HLA-identical siblings as compared with chemotherapy for children with acute lymphoblastic leukemia in a second remission. N Engl J Med 1994; 331: 1253–1258.
De Witte T, Awwad B, Boezeman J, Schattenberg A, Muus P, Raemaekers J et al. Role of allogenic bone marrow transplantation in adolescent or adult patients with acute lymphoblastic leukaemia or lymphoblastic lymphoma in first remission. Bone Marrow Transplant 1994; 14: 767–774.
Saarinen UM, Mellander L, Nysom K, Ringden O, Schroeder H, Glomstein A et al. Allogeneic bone marrow transplantation in first remission for children with very high-risk acute lymphoblastic leukemia: a retrospective case-control study in the Nordic countries. Nordic Society for Pediatric Hematology and Oncology (NOPHO). Bone Marrow Transplant 1996; 17: 357–363.
Dopfer R, Henze G, Bender-Gotze C, Ebell W, Ehninger G, Friedrich W et al. Allogeneic bone marrow transplantation for childhood acute lymphoblastic leukemia in second remission after intensive primary and relapse therapy according to the BFM- and CoALL-protocols: results of the German Cooperative Study. Blood 1991; 78: 2780–2784.
Hoogerbrugge PM, Gerritsen EJ, vd Does-van den Berg A, vd Berg H, Zwinderman AH, Hermans J et al. Case-control analysis of allogeneic bone marrow transplantation versus maintenance chemotherapy for relapsed ALL in children. Bone Marrow Transplant 1995; 15: 255–259.
Uderzo C, Valsecchi MG, Bacigalupo A, Meloni G, Messina C, Polchi P et al. Treatment of childhood acute lymphoblastic leukemia in second remission with allogeneic bone marrow transplantation and chemotherapy: ten-year experience of the Italian Bone Marrow Transplantation Group and the Italian Pediatric Hematology Oncology Association. J Clin Oncol 1995; 13: 352–358.
Doney K, Fisher LD, Appelbaum FR, Buckner CD, Storb R, Singer J et al. Treatment of adult acute lymphoblastic leukemia with allogeneic bone marrow transplantation. Multivariate analysis of factors affecting acute graft-versus-host disease, relapse, and relapse-free survival. Bone Marrow Transplant 1991; 7: 453–459.
Ringden O, Horowitz MM . Graft-versus-leukemia reactions in humans. The Advisory Committee of the International Bone Marrow Transplant Registry. Transplant Proc 1989; 21: 2989–2992.
Powles R, Singhal S, Treleaven J, Kulkarni S, Horton C, Mehta J . Identification of patients who may benefit from prophylactic immunotherapy after bone marrow transplantation for acute myeloid leukemia on the basis of lymphocyte recovery early after transplantation. Blood 1998; 91: 3481–3486.
Kumar S, Chen MG, Gastineau DA, Gertz MA, Inwards DJ, Lacy MQ et al. Effect of slow lymphocyte recovery and type of graft-versus-host disease prophylaxis on relapse after allogeneic bone marrow transplantation for acute myelogenous leukemia. Bone Marrow Transplant 2001; 28: 951–956.
Bostrom B, Woods WG, Nesbit ME, Krivit W, Kersey J, Weisdorf D et al. Successful reinduction of patients with acute lymphoblastic leukemia who relapse following bone marrow transplantation. J Clin Oncol 1987; 5: 376–381.
Mehta J . The first clinical use of donor leukocyte infusions for the treatment of leukemia relapsing after allogeneic bone marrow transplantation. Blood 1996; 87: 4010.
Singhal S, Powles R, Kulkarni S, Treleaven J, Saso R, Mehta J . Long-term follow-up of relapsed acute leukemia treated with immunotherapy after allogeneic transplantation: the inseparability of graft-versus-host disease and graft-versus-leukemia, and the problem of extramedullary relapse. Leukemia Lymphoma 1999; 32: 505–512.
Collins Jr RH, Shpilberg O, Drobyski WR, Porter DL, Giralt S, Champlin R et al. Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol 1997; 15: 433–444.
Kolb HJ, Schattenberg A, Goldman JM, Hertenstein B, Jacobsen N, Arcese W et al for the European Group for Blood and Marrow Transplantation Working Party Chronic Leukemia. Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. Blood 1995; 86: 2041–2050.
Collins Jr RH, Goldstein S, Giralt S, Levine J, Porter D, Drobyski W et al. Donor leukocyte infusions in acute lymphocytic leukemia. Bone Marrow Transplant 2000; 26: 511–516.
Yazaki M, Andoh M, Ito T, Ohno T, Wada Y . Successful prevention of hematological relapse for a patient with Philadelphia chromosome-positive acute lymphoblastic leukemia after allogeneic bone marrow transplantation by donor leukocyte infusion. Bone Marrow Transplant 1997; 19: 393–394.
Bader P, Klingebiel T, Schaudt A, Theurer-Mainka U, Handgretinger R, Lang P et al. Prevention of relapse in pediatric patients with acute leukemias and MDS after allogeneic SCT by early immunotherapy initiated on the basis of increasing mixed chimerism: a single center experience of 12 children. Leukemia 1999; 13: 2079–2086.
Pavletic ZS, Joshi SS, Pirruccello SJ, Tarantolo SR, Kollath J, Reed EC et al. Lymphocyte reconstitution after allogeneic blood stem cell transplantation for hematologic malignancies. Bone Marrow Transplant 1998; 21: 33–41.
Weiden PL, Flournoy N, Thomas ED, Prentice R, Fefer A, Buckner CD et al. Antileukemic effect of graft-versus-host disease in human recipients of allogeneic-marrow grafts. N Engl J Med 1979; 300: 1068–1073.
Horowitz MM, Gale RP, Sondel PM, Goldman JM, Kersey J, Kolb HJ et al. Graft-versus-leukemia reactions after bone marrow transplantation. Blood 1990; 75: 555–562.
Weisdorf DJ, Nesbit ME, Ramsay NK, Woods WG, Goldman AI, Kim TH et al. Allogeneic bone marrow transplantation for acute lymphoblastic leukemia in remission: prolonged survival associated with acute graft-versus-host disease. J Clin Oncol 1987; 5: 1348–1355.
Passweg JR, Tiberghien P, Cahn JY, Vowels MR, Camitta BM, Gale RP et al. Graft-versus-leukemia effects in T lineage and B lineage acute lymphoblastic leukemia. Bone Marrow Transplant 1998; 21: 153–158.
Martin PJ, Hansen JA, Buckner CD, Sanders JE, Deeg HJ, Stewart P et al. Effects of in vitro depletion of T cells in HLA-identical allogeneic marrow grafts. Blood 1985; 66: 664–672.
Hoffmann T, Theobald M, Bunjes D, Weiss M, Heimpel H, Heit W . Frequency of bone marrow T cells responding to HLA-identical non-leukemic and leukemic stimulator cells. Bone Marrow Transplant 1993; 12: 1–8.
Jiang YZ, Kanfer EJ, Macdonald D, Cullis JO, Goldman JM, Barrett AJ . Graft-versus-leukaemia following allogeneic bone marrow transplantation: emergence of cytotoxic T lymphocytes reacting to host leukaemia cells. Bone Marrow Transplant 1991; 8: 253–258.
Faber LM, van Luxemburg-Heijs SA, Willemze R, Falkenburg JH . Generation of leukemia-reactive cytotoxic T lymphocyte clones from the HLA-identical bone marrow donor of a patient with leukemia. J Exp Med 1992; 176: 1283–1289.
Truitt RL, Atasoylu AA . Contribution of CD4+ and CD8+ T cells to graft-versus-host disease and graft-versus-leukemia reactivity after transplantation of MHC-compatible bone marrow. Bone Marrow Transplant 1991; 8: 51–58.
Glass B, Uharek L, Gassmann W, Focks B, Bolouri H, Loeffler H et al. Graft-versus-leukemia activity after bone marrow transplantation does not require graft-versus-host disease. Ann Hematol 1992; 64: 255–259.
Jiang YZ, Barrett AJ, Goldman JM, Mavroudis DA . Association of natural killer cell immune recovery with a graft-versus-leukemia effect independent of graft-versus-host disease following allogeneic bone marrow transplantation. Ann Hematol 1997; 74: 1–6.
Schroff RW, Gale RP, Fahey JL . Regeneration of T cell subpopulations after bone marrow transplantation: cytomegalovirus infection and lymphoid subset imbalance. J Immunol 1982; 129: 1926–1930.
Azogui O, Kalil J, Andersen E, Guillet J, Fellous M, Gluckman E . Peripheral blood T lymphocytes bearing DR antigen after bone marrow transplantation. Transplantation 1983; 35: 513–515.
Hercend T, Takvorian T, Nowill A, Tantravahi R, Moingeon P, Anderson KC et al. Characterization of natural killer cells with antileukemia activity following allogeneic bone marrow transplantation. Blood 1986; 67: 722–728.
Keever CA, Klein J, Leong N, Copelan EA, Avalos BR, Kapoor N et al. Effect of GVHD on the recovery of NK cell activity and LAK precursors following BMT. Bone Marrow Transplant 1993; 12: 289–295.
Mackinnon S, Hows JM, Goldman JM . Induction of in vitro graft-versus-leukemia activity following bone marrow transplantation for chronic myeloid leukemia. Blood 1990; 76: 2037–2045.
Elfenbein GJ, Anderson PN, Humphrey RL, Mullins GM, Sensenbrenner LL, Wands JR et al. Immune system reconstitution following allogeneic bone marrow transplantation in man: a multiparameter analysis. Transplant Proc 1976; 8: 641–646.
Noel DR, Witherspoon RP, Storb R, Atkinson K, Doney K, Mickelson EM et al. Does graft-versus-host disease influence the tempo of immunologic recovery after allogeneic human marrow transplantation? An observation on 56 long-term survivors. Blood 1978; 51: 1087–1105.
Atkinson K, Hansen JA, Storb R, Goehle S, Goldstein G, Thomas ED . T-cell subpopulations identified by monoclonal antibodies after human marrow transplantation. I. Helper-inducer and cytotoxic-suppressor subsets. Blood 1982; 59: 1292–1298.
Ault KA, Antin JH, Ginsburg D, Orkin SH, Rappeport JM, Keohan ML et al. Phenotype of recovering lymphoid cell populations after marrow transplantation. J Exp Med 1985; 161: 1483–1502.
Niederwieser D, Gastl G, Rumpold H, Marth C, Kraft D, Huber C . Rapid reappearance of large granular lymphocytes (LGL) with concomitant reconstitution of natural killer (NK) activity after human bone marrow transplantation (BMT). Br J Haematol 1987; 65: 301–305.
Herberman RB, Reynolds CW, Ortaldo JR . Mechanism of cytotoxicity by natural killer (NK) cells. Annu Rev Immunol 1986; 4: 651–680.
Porrata LF, Gertz MA, Inwards DJ, Litzow MR, Lacy MQ, Tefferi A et al. Early lymphocyte recovery predicts superior survival after autologous hematopoietic stem cell transplantation in multiple myeloma or non-Hodgkin lymphoma. Blood 2001; 98: 579–585.
Porrata LF, Inwards DJ, Lacy MQ, Markovic SN . Immunomodulation of early engrafted natural killer cells with interleukin-2 and interferon-alpha in autologous stem cell transplantation. Bone Marrow Transplant 2001; 28: 673–680.
Adler A, Chervenick PA, Whiteside TL, Lotzova E, Herberman RB . Interleukin 2 induction of lymphokine-activated killer (LAK) activity in the peripheral blood and bone marrow of acute leukemia patients. I. Feasibility of LAK generation in adult patients with active disease and in remission. Blood 1988; 71: 709–716.
Nakano E, Tada Y, Ichikawa Y, Fujioka H, Matsuda M, Takaha M et al. Lysis of autologous tumor cells by peripheral blood lymphocytes treated with interleukin 2 in patients with renal cell carcinoma. J Urol 1987; 137: 641–648.
Soiffer RJ, Murray C, Gonin R, Ritz J . Effect of low-dose interleukin-2 on disease relapse after T-cell-depleted allogeneic bone marrow transplantation. Blood 1994; 84: 964–971.
Meyers JD, Flournoy N, Sanders JE, McGuffin RW, Newton BA, Fisher LD et al. Prophylactic use of human leukocyte interferon after allogeneic marrow transplantation. Ann Intern Med 1987; 107: 809–816.
Einsele H, Ehninger G, Steidle M, Fischer I, Bihler S, Gerneth F et al. Lymphocytopenia as an unfavorable prognostic factor in patients with cytomegalovirus infection after bone marrow transplantation. Blood 1993; 82: 1672–1678.
Fries BC, Khaira D, Pepe MS, Torok-Storb B . Declining lymphocyte counts following cytomegalovirus (CMV) infection are associated with fatal CMV disease in bone marrow transplant patients. Exp Hematol 1993; 21: 1387–1392.
Acknowledgements
We thank Ms Norine E Huneke for her expert assistance with BMT database management.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kumar, S., Chen, M., Gastineau, D. et al. Lymphocyte recovery after allogeneic bone marrow transplantation predicts risk of relapse in acute lymphoblastic leukemia. Leukemia 17, 1865–1870 (2003). https://doi.org/10.1038/sj.leu.2403055
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2403055
Keywords
This article is cited by
-
Increased bone marrow CD56bright natural killer cells at 30 days after allogeneic stem cell transplantation associated with adverse patient outcome
Bone Marrow Transplantation (2019)
-
Early lymphocyte recovery predicts clinical outcome after HSCT with mycophenolate mofetil prophylaxis in the Japanese population
International Journal of Hematology (2018)
-
Low blood lymphocyte count at 30 days post transplant predicts worse acute GVHD and survival but not relapse in a large retrospective cohort
Bone Marrow Transplantation (2015)
-
Impact of lymphocyte and monocyte recovery on the outcomes of allogeneic hematopoietic SCT with fludarabine and melphalan conditioning
Bone Marrow Transplantation (2013)
-
Characteristics and influencing factors of CD19+ B cell reconstitution in patients following haploidentical/mismatched hematopoietic stem cell transplantation
International Journal of Hematology (2012)