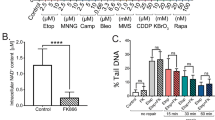
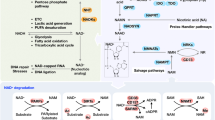
Abstract
IN the oxidation–reduction metabolism of a cell the pyridine nucleotide NAD is used catalytically; any NAD that is reduced is reoxidised. But NAD is consumed in certain metabolic reactions in which it serves as a substrate. In eukaryotic cells, the most intriguing of these reactions is the cleavage of NAD to form nicotinamide and a unique polymer, poly adenosine diphosphoribose (poly ADPR) (Fig. 1); the reaction is catalysed by the enzyme poly ADPR synthetase1–4. Other well studied reactions involving the destruction of NAD include the cleavage of NAD to form AMP and NMN by bacterial DNA ligases5,6 and the breakdown of NAD (with the concomitant inactivation of protein synthesis) by diphtheria toxin7,8.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Chambon, P., Weill, J., and Mandel, P., Biochem. biophys. Res. Commun., 11, 39–43 (1963).
Chambon, P., Weill, J., Doly, J., Strosser, M., and Mandel, P., Biochem. biophys Res. Commun., 25, 634–643 (1966).
Sugimura, T., Prog. Nucleic Acid Res. molec. Biol., 13, 127–151 (1973).
Honjo, T., and Hayaishi, O., Curr. Top. cell. Reg., 7, 87–127 (1973).
Olivera, B., and Lehman, I., Proc. natn. Acad. Sci. U.S.A., 57, 1426–1433 (1967).
Zimmerman, S., Little, B., Oshinsky, J., and Gellert, C., Proc. natn. Acad. Sci. U.S.A., 57, 1841–1848 (1967).
Collier, R., and Pappenheimer, A. M., J. exp. Med., 120, 1019–1039 (1964).
Collier, R., J. molec. Biol., 25, 83–98 (1967).
Matsuya, Y., and Green, H., Science, 163, 697–698 (1969).
Rechsteiner, M., Hillyard, D., and Olivera, B., J. cell. Physiol., (in the press).
Hillyard, D., Rechsteiner, M., and Olivera, B., J. cell. Physiol., 82, 165–179 (1973).
Rechsteiner, M., and Catanzarite, V., J. cell. Physiol., 84, 409–422 (1974).
Prescott, D., Myerson, D., and Wallace, J., Expl Cell. Res., 71, 480–485 (1971).
Rechsteiner, M., Lund, K., Hillyard, D., and Olivera, B., J. cell. Physiol., 83, 389–400 (1974).
Hogeboom, G., and Schneider, W., J. Biol. Chem., 197, 611–620 (1952).
Siebert, G., and Humphrey, G., Adv. Enzymol., 21, 239–288 (1965).
Ueda, K., Reeder, R., Honjo, T., Nishizuka, Y., and Hayaishi, O., Biochem. biophys. Res. Commun., 31, 379–385 (1968).
Smith, J., and Stocken, L., Biochem. biophys. Res. Commun., 54, 297–300 (1973).
Shall, S., Brightwell, M., O'Farrell, M., Stone, P., and Whish, W., Z. physiol. Chem., 353, 846–847 (1972).
Futai, M., Mizuno, D., and Sugimara, T., J. biol. Chem., 243, 6325–6329 (1968).
Miwa, M., and Sugimura, T., J. biol. Chem., 246, 6362–6364 (1971).
Gholson, R. K., Nature, 212, 933–935 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
RECHSTEINER, M., HILLYARD, D. & OLIVERA, B. Magnitude and significance of NAD turnover in human cell line D98/AH2. Nature 259, 695–696 (1976). https://doi.org/10.1038/259695a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/259695a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.