Abstract
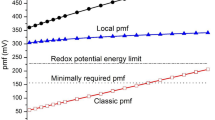
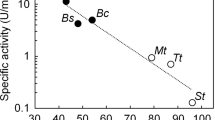
HOMOLOGOUS enzymes of species with different cell temperatures usually display temperature-adaptive differences in their catalytic properties1–7. Thus enzymes of low cell temperature species characteristically exhibit higher specific activities and lower activation free energies and enthalpies than the homologous enzymes of high cell temperature species2–7. For lactate dehydrogenase (LDH, E.G. 1.1.1.27; lactate: NAD+ oxidoreductase), a single M4 (muscle type) LDH molecule from a cold-water fish can catalyse the conversion of pyruvate to lactate three times as rapidly at 5 °C as an M4-LDH from a mammal5. In this paper we present evidence that these temperature-compensating differences in catalytic efficiency may derive in part from different amounts of exergonic hydration of the enzymes during the activation step in catalysis. Lowering the activation free energy (ΔG‡) ‘barriers’ to enzymic reactions by way of the reversible hydration of amino acid side chains and/or polar peptide linkages permits the adjustment of catalytic efficiencies without necessitating the modification of amino acid sequences in the active sites of the enzymes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hochachka, P. W. & Somero, G. N. Strategies of Biochemical Adaptation Saunders, Philadelphia, 1973).
Somero, G. N. & Low, P. S. in Biochemical Adaptation to Environmental Change, Biochemical Society Symposium No. 41, (eds Smellie, R. M. S. & Pennock, J. F.) 33–42 (Biochemical Society, London, 1976).
Somero, G. N. J. exp. Zool. 194, 175–188 (1975).
Low, P. S. & Somero, G. N. J. exp. Zool. 198, 1–12 (1976).
Low, P. S., Bada, J. L. & Somero, G. N. Proc. natn. Acad. Sci. U.S.A. 70, 430–432 (1973).
Johnston, I. A. & Goldspink, G. Nature 257, 620–623 (1975).
Borgmann, U. & Moon, T. W. Can. J. Biochem. 53, 998–1004 (1975).
Taylor, S. S. & Oxley, S. S. Archs Biochem. Biophys. 175, 373–383 (1976).
Perham, R. N. Biochem. J. 111, 17–21 (1969).
Ting, S., Lai, C. Y. & Horecker, B. L. Archs Biochem. Biophys. 144, 476–484 (1971).
Low, P. S. & Somero, G. N. Proc. natn. Acad. Sci. U.S.A. 72, 3305–3309 (1975).
Koshland, D. E. & Neet, K. E. A. Rev. Biochem. 37, 359–410 (1968).
Low, P. S. & Somero, G. N. Proc. natn. Acad. Sci. U.S.A. 72, 3014–3018 (1975).
Johnson, F. H., Eyring, H. & Polissar, M. J. in Kinetic Basis of Molecular Biology, 1–41, (Wiley, New York, 1954).
Von Hippel, P. H. & Schleich, T. in Structure and Stability of Biological Macromolecules (eds Timesheff, S. N. & Fasman, G. D.) 417–574 (Dekker, New York, 1969).
Somero, G. N. & Low, P. S. Am. Nat. (in press).
Banerjee, S. K. & Rupley, J. A. J. biol. Chem. 248, 2117–2124 (1973).
Krishnan, C. V. & Friedman J. phys. Chem. 73, 1572–1580 (1969); 75, 3598–3606 (1971).
Pesce, A., Fondy, T. P., Stolzenbach, F., Castillo, F. & Kaplan, N. O. J. biol. Chem. 242, 2151–2167 (1967).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SOMERO, G., LOW, P. Enzyme hydration may explain catalytic efficiency differences among lactate dehydrogenase homologues. Nature 266, 276–278 (1977). https://doi.org/10.1038/266276a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/266276a0
This article is cited by
-
Evolutionary temperature adaptation and the calcium regulation of fish actomyosin ATPases
Journal of Comparative Physiology ? B (1979)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.