Abstract
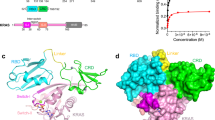
The crystal structure of human H-Ras complexed with the Ras guanine-nucleotide-exchange-factor region of the Son of sevenless (Sos) protein has been determined at 2.8 Å resolution. The normally tight interaction of nucleotides with Ras is disrupted by Sos in two ways. First, the insertion into Ras of an α-helix from Sos results in the displacement of the Switch 1 region of Ras, opening up the nucleotide-binding site. Second, side chains presented by this helix and by a distorted conformation of the Switch 2 region of Ras alter the chemical environment of the binding site for the phosphate groups of the nucleotide and the associated magnesium ion, so that their binding is no longer favoured. Sos does not impede the binding sites for the base and the ribose of GTP or GDP, so the Ras–Sos complex adopts a structure that allows nucleotide release and rebinding.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bourne, H. R., Sanders, D. A. & McCormick, F. The GTPase superfamily: conserved structure and molecular mechanism. Nature 349, 117–127 (1991).
Boguski, M. S. & McCormick, F. Proteins regulating Ras and its relatives. Nature 366, 643–654 (1993).
Medema, R. H., de Vries-Smits, A. M., van der Zon, G. C. M., Maassen, J. A. & Bos, J. L. Ras activation by insulin and epidermal growth factor through enhanced exchange of guanine nucleotides on p21ras. Mol. Cell. Biol. 13, 155–162 (1993).
Buday, L. & Downward, J. Epidermal growth factor regulates p21ras through the formation of a complex of receptor, Grb2 adapter protein, and Sos nucleotide exchange factor. Cell 73, 611–620 (1993).
Gale, N. W., Kaplan, S., Lowenstein, E. J., Schlessinger, J. & Bar-Sagi, D. Grb2 mediates the EGF-dependent activation of guanine nucleotide exchange on Ras. Nature 363, 88–92 (1993).
Bar-Sagi, D. The Sos (Son of sevenless) protein. Trends Endocrin. Metab. 5, 165–169 (1994).
Schlessinger, J. How receptor tyrosine kinases activate Ras. Trends Biochem. Sci. 18, 273–275 (1994).
Yu, H. & Schreiber, S. L. Structure of guanine-nucleotide-exchange factor human Mss4 and identification of its Rab-interacting surface. Nature 376, 788–791 (1995).
Mossessova, E., Gulbis, J. M. & Goldberg, J. Structure of the guanine nucleotide exchange factor Sec7 domain of human Arno and analysis of the interaction with ARF GTPase. Cell 92, 415–423 (1998).
Cherfils, J. et al. Structure of the Sec7 domain of the Arf exchange factor ARNO. Nature 392, 101–105 (1998).
Renault, L. et al. The 1.7 Å crystal structure of the regulator of chromosome condensation (RCC1) reveals a seven-bladed propeller. Nature 392, 97–101 (1998).
Wang, Y., Jiang, Y., Meyering-Voss, M., Sprinzl, M. & Sigler, P. B. Crystal structure of the EF-Tu·EF-Ts complex from Thermus thermophilus. Nat. Struct. Biol. 4, 650–656 (1997).
Kawashima, T., Berthet-Colominas, C., Wulff, M., Cusack, S. & Leberman, R. The structure of the Escherichia coli EF-Tu-EF-Ts complex at 2.5 Å resolution. Nature 379, 511–518 (1996).
Holm, L. & Sander, C. Protein structure comparison by alignment of distance matrices. J. Mol. Biol. 233, 123–138 (1993).
Chardin, P. et al. Human Sos 1: A gunaine nucleotide exchange factor for Ras that binds to GRB2. Science 260, 1338–1343 (1993).
Lenzen, C., Cool, R. H., Prinz, H., Kuhlmann, J. & Wittinghofer, A. Kinetic analysis by fluorescence of the interaction between Ras and the catalytic domain of the guanine nucleotide exchange factor Cdc25Mm Biochemistry 37, 7420–7430 (1998).
Lai, C.-C., Boguski, M., Broek, D. & Powers, S. Influence of guanine nucleotides on complex formation between Ras and Cdc25 proteins. Mol. Cell. Biol. 13, 1345–1352 (1993).
Mistou, M. Y. et al. Mutations of H-Ras p21 that define important regions for the molecular mechanism of the SDC25 C-domain, a guanine nucleotide dissociation stimulator. EMBO J. 11, 2391–2397 (1992).
Milburn, M. V. et al. Molecular switch for signal transduction: structural differences between active and inactive forms of protooncogenic ras proteins. Science 247, 939–945 (1990).
Jurnak, F. Structure of the GDP domainof EF-Tu and location of the amino acids homologous to ras oncogene proteins. Science 230, 32–36 (1985).
1. Powers, S., O'Neill, K. & Wigler, M. Dominant yeast and mammalian Ras mutants that interfere with CDC25-dependent activation of wild-type Ras in Saccharomyces cerevisiae. Mol. Cell. Biol. 9, 390–395 (1989).
Haney, S. A. & Broach, J. R. Cdc25p, the guanine nucleotide exchange factor for the Ras proteins of Saccharomyces cerevisiae, promotes exchange by stabilizing ras in a nucleotide free state. J. Biol. Chem. 269, 16541–16548 (1994).
Klebe, C., Prinz, H., Wittinghofer, A. & Goody, R. S. The kinetic mechanism of Ran-nucleotide exchange catalyzed by RCC1. Biochemistry 34, 12543–12552 (1995).
Verrotti, A. C. et al. Ras residues that are distant from the GDP binding site play a critical role in dissociation factor-stimulated release of GDP. EMBO J. 11, 2855–2862 (1992).
Segal, M., Willumsen, B. M. & Levitzki, A. Residues crucial for Ras interaction with GDP-GTP exchangers. Proc. Natl Acad. Sci. USA 90, 5564–5568 (1993).
Mosteller, R. D., Han, J. & Broek, D. Identification of residues of the H-Ras protein critical for functional interaction with guanine nucleotide exchange factors. Mol. Cell. Biol. 14, 1104–1112 (1994).
Segal, M., Marbach, I., Willumsen, B. M. & Levitzki, A. Two distinct regions of Ras participate in functional interaction with GDP-GTP exchangers. Eur. J. Biochem. 228, 96–101 (1995).
Leonardsen, L., DeClue, J. E., Lybaek, H., Lowy, D. R. & Willumsen, B. M. Rasp21 sequences opposite the nucleotide binding pocket are required for GRF-mediated nucleotide release. Oncogene 13, 2177–2187 (1996).
Crechet, J.-B., Bernardi, A. & Parmeggiani, A. Distal switch II region of Ras2p is required for interaction with guanine nucleotide exchange factor. J. Biol. Chem. 271, 17234–17240 (1996).
Quilliam, L. A. et al. Involvement of the switch 2 domain of Ras in its interaction with guanine nucleotide exchange factors. J. Biol. Chem. 271, 11076–11082 (1996).
1. Feig, L. A. & Cooper, G. M. Inhibition of NIH 3T3 cell proliferation by a mutant Ras protein with preferential affinity for GDP. Mol. Cell. Biol. 8, 3235–3243 (1988).
2. Chen, S.-Y., Huff, S. Y., Lai, C.-C., Der, C. J. & Powers, S. Ras-15A protein shares highly similar dominant-negative biological properties with Ras-17N and forms a stable, guanine-nucleotide resistant complex with CDC25 exchange factor. Oncogene 9, 2691–2698 (1994).
Powers, S., Gonzales, E., Christensen, T., Cubert, J. & Broek, D. Functional cloning of Bud5, a Cdc25-related gene from S. cerevisiae that can suppress a dominant-negative Ras2 mutant. Cell 65, 1225–1231 (1991).
Westbrook, E. M. & Naday, I. Charge-coupled device-based area detectors. Methods Enzymol. 276, 244–268 (1997).
Otwinowski, Z. & Minor, W. Processing of x-ray diffraction data collected in oscillation model Mehtods Enzymol. 276, 307–326 (1997).
CCP4 Suite: Programs for protein crystallography. Acta Crystallogr. D 50, 760–763 (1994).
Cowtan, K. Joint CCP4 and ESF-EACBM Newslett. Protein Crystallogr. 31, 34–38 (1994).
Jones, T. A., Zou, J.-Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for building protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991).
Pai, E. F. et al. Refined crystal structure of the triphosphate conformation of H-ras p21 at 1.35 Å resolution: implications for the mechanism of GTP hydrolysis. EMBO J. 9, 2351–2359 (1990).
Bernstein, F. C. et al. The protein data bank: a computer-based archival file for macromolecular structures. Arch. Biochem. Biophys. 185, 584–591 (1978).
1. Brünger, A. T. et al. Crystallography and NMR system (CNS): A new software suite for macromolecular structure determination. Acta Crystallogr. D (in the press).
2. Read, R. J. Improved Fourier coefficients for maps using phases from partial structures with errors. Acta Crystallogr. A 42, 140–149 (1986).
3. Brünger, A. T., Adams, P. D. & Rice, L. M. New applications of simulated annealing in X-ray crystallography and solution NMR. Structure 5, 325–336 (1997).
4. Carson, M. Ribbons 2.0. J. Appl. Crystallogr. 24, 958–961 (1991).
Nicholls, A., Sharp, K. A. & Honig, B. Protein folding and association: insights from the interfacial and thermodynamic properties of hydrocarbons. Proteins Struct. Funct. Genet. 11, 281–296 (1991).
Pai, E. F. et al. Structure of the guanine-nucleotide-binding domain of Ha-ras oncogene product p21 in the triphosphate conformation. Nature 341, 209–214 (1989).
Acknowledgements
We thank L. Leighton and M. Uy for technical assistance; J. Bonanno, S. K. Burley, R.H. Chen, X. Chen, E. Conti, S. Corbalan-Garcia, D. Galron, B. Hall, M. Huse, S. Jacques, D. Jeruzalmi, C.-H. Lee, S. Soisson, H. Yamaguchi, S.-S. Yang and Y. Zhang for advice and help; L. Berman, R. Sweet, the staff of NSLS and CHESS for help with synchrotron data collection; and A. Wittinghofer and R. H. Cool for sharing a manuscript prior to publication. P.A.B.-S. is supported by grants for the Cystic Fibrosis Foundation and the NIH. D.B.-S. acknowledges grant support from the NIH.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boriack-Sjodin, P., Margarit, S., Bar-Sagi, D. et al. The structural basis of the activation of Ras by Sos. Nature 394, 337–343 (1998). https://doi.org/10.1038/28548
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/28548
This article is cited by
-
The emerging roles of MAPK-AMPK in ferroptosis regulatory network
Cell Communication and Signaling (2023)
-
EIF4A3-mediated circ_0042881 activates the RAS pathway via miR-217/SOS1 axis to facilitate breast cancer progression
Cell Death & Disease (2023)
-
Designing of disruptor molecules to restrain the protein–protein interaction network of VANG1/SCRIB/NOS1AP using fragment-based drug discovery techniques
Molecular Diversity (2023)
-
Drugging KRAS: current perspectives and state-of-art review
Journal of Hematology & Oncology (2022)
-
The path to the clinic: a comprehensive review on direct KRASG12C inhibitors
Journal of Experimental & Clinical Cancer Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.