Abstract
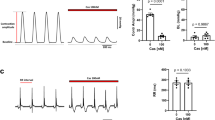
Transmembrane influx of extracellular calcium through specific calcium channels is now accepted to have an important role in the excitation–contraction coupling of cardiac and smooth muscle1–3. The importance of such slow calcium channels has been underlined by the development of specific calcium channel blocking agents, the ‘calcium antagonists’, typified by verapamil, nifedipine and diltiazem4. These drugs have been used to investigate the properties of slow calcium channels in a variety of tissues. We have found that small modifications to the nifedipine molecule produce other dihydropyridine derivatives (see Fig. 1) with effects diametrically opposite to those of the calcium antagonists: cardiac contractility is stimulated and smooth muscle is contracted. These effects are competitively antagonized by nifedipine. Apparently, nifedipine and the novel compounds bind to the same specific dihydropyridine binding sites in or near the calcium channel. In contrast to nifedipine, however, the new compounds promote—instead of inhibiting—the influx of Ca2+ ions. We report here the properties of BAY K 8644 (methyl 1,4-dihydro-2,6-dimethyl-3-nitro-4-(2-trifluoromethylphenyl)-pyridine-5-carboxylate), one of the most potent of these novel compounds.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kohlhardt, M. Basic Res. Cardiol. 76, 589–601 (1981).
Bolton, T. B. Physiol. Rev. 59, 607–718 (1979).
Reuter, H. Nature 301, 569–574 (1983).
Fleckenstein, A. A. Rev. Pharmac. Tox. 17, 149–166 (1977).
Loev, B., Ehrreich, S. J. & Tedeschi, R. E. J. Pharm. Pharmac. 24, 917–918 (1972).
Loev, B., Goodman, M. M., Snader, K. M., Tedeschi, R. & Macko, E. J. med. Chem. 17, 956–965 (1974).
Rodenkirchen, R. et al. Naunyn-Schmiedebergs Archs Pharmak. 310, 69–78 (1979).
Triggle, D. J. & Swany, V. C. Circulation Res. (in the press).
Germ. Pat. Applic. DOS 3130041 (1981).
Lineweaver, H. & Burke, D. J. Am. chem. Soc. 56, 658–666 (1934).
Rosenberger, L. B., Ticku, M. K. & Triggle, D. J. Can. J. Physiol. Pharmac. 57, 333–347 (1979).
Bellemann, P., Ferry, D., Lübbecke, F. & Glossmann, H. Arzneimittel-Forsch. 31, 2064–2067 (1981).
Bolger, G. T. et al. Biochem. biophys. Res. Commun. 104, 1604–1609 (1982).
Murphy, K. M. M. & Snyder, S. H. Eur. J. Pharmac. 77, 201–202 (1982).
Holck, M., Thorens, S. & Haeusler, G. Eur. J. Pharmac. 85, 305–315 (1982).
Vater, W. et al. Arzneimittel-Forsch. 22, 1–14 (1972).
Opie, L. H. J. Physiol., Lond. 180, 529–541 (1965).
Furchgott, R. F. & Bhadrakom, S. J. Pharmac. exp. Ther. 108, 129–143 (1953).
Towart, R. J. Cardiovasc. Pharmac. 4, 895–902 (1982).
Bellemann, P., Schade, A. & Towart, R. Proc. natn. Acad. Sci. U.S.A. 80, 2356–2360 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schramm, M., Thomas, G., Towart, R. et al. Novel dihydropyridines with positive inotropic action through activation of Ca2+ channels. Nature 303, 535–537 (1983). https://doi.org/10.1038/303535a0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/303535a0
This article is cited by
-
Design and exploration of caffeine-based Brönsted acidic ionic liquid (CaffBAIL) for the synthesis of DHPMs, xanthenediones, and acridinediones
Research on Chemical Intermediates (2022)
-
An Efficient One-Pot Four-Component Synthesis of 9-Aryl-Hexahydroacridine-1,8-Dione Derivatives in the Presence of a Molecular Sieves Supported Iron Catalyst
Catalysis Letters (2019)
-
Simvastatin reduces adrenal catecholamine secretion evoked by stimulation of cholinergic nicotinic and angiotensinergic AT1 receptors
Archives of Pharmacal Research (2018)
-
Dihydropyridines’ metabolites-induced early apoptosis after myocardial infarction in rats; new outlook on preclinical study with M-2 and M-3
Apoptosis (2016)
-
A one-pot four-component reaction providing quinoline-based 1,4-dihydropyridines
Journal of the Iranian Chemical Society (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.