Abstract
Study design:
Case report.
Objective:
We describe a patient who developed transverse myelitis (TM) following a nerve root injection of steroids and anesthetic at L2 for radicular pain.
Setting:
Baltimore, MD, USA.
Clinical presentation:
A 42-year-old woman developed progressive lower extremity weakness and paresthesias, a T12 sensory level and urinary urgency 8 h following the injection of Marcaine and Celestone into the left L2 nerve root. Magnetic resonance imaging showed T2 signal abnormality with gadolinium enhancement from T12 to the conus medullaris and there was no evidence of traumatic injury to the spinal cord. The patient had undiagnosed Behcet's disease (BD) and had experienced multiple episodes of pathergy: hyper-responsiveness of the skin to local trauma, resulting in inflammation and edema. Intravenous steroids were initiated and the patient experienced a near total clinical resolution and a complete radiologic resolution.
Conclusion:
Since the spinal cord inflammation developed after and immediately adjacent to local spinal trauma, we suggest that the TM in this patient was related to BD and was a pathergy response in the spinal cord.
Similar content being viewed by others
Introduction
Behcet's disease (BD) is a chronic, multisystem vasculitic disorder of uncertain etiology. It is characterized clinically by recurrent oral and genital ulcers, livedo reticularis, erythema nodosum, uveitis or retinal vasculitis and nondeforming oligo-arthritis. There is no serologic or radiologic test that defines (or is even supportive of) BD, although there is an increased incidence in patients with HLA-51 haplotype.1 The pathogenesis of BD is unknown but may involve activation of the innate immune system by heat–shock proteins2 and the subsequent elaboration of TH1-type cytokines.3
A pathergy reaction (hyper-responsiveness to skin needle prick resulting in erthyema or swelling at the site) is often associated with BD and is one of the main diagnostic criterion.4 It is uncommon in BD patients living in the US or in Britain.5, 6 The pathergy response likely reflects endothelial dysfunction within small blood vessels and indeed a recent study has shown enhanced immunohistochemical staining of cell adhesion molecules and endothelial growth factor markers within skin of BD patients with a pathergy response.7 Neurologic BD (nBD) most commonly involves the cerebral or brainstem parenchyma although spinal cord involvement is seen in 14% of patients.8
Transverse myelitis (TM) is a clinical syndrome in which an immune-mediated process causes neural injury to the spinal cord, resulting in varying degrees of weakness, sensory alterations and autonomic dysfunction. TM may exist as part of a multifocal CNS disease (eg multiple sclerosis), multisystemic disease (eg systemic lupus erythematosus) or as an isolated entity. TM is a rare syndrome with an incidence of between one and eight new cases per million people per year.9 There is often a clearly defined rostral border of sensory dysfunction and evidence of acute inflammation demonstrated by a spinal MRI and lumbar puncture. When the maximal level of deficit is reached, approximately 50% of patients have lost all movements of their legs, virtually all patients have some degree of bladder dysfunction and 80–94% of patients have numbness, paresthesias or band-like dysesthesias.9, 10, 11, 12, 13, 14 Autonomic symptoms consist variably of increased urinary urgency, bowel or bladder incontinence, difficulty or inability to void, incomplete evacuation or bowel constipation and sexual dysfunction.15 Recently, we proposed a diagnostic and classification scheme which has defined TM as either idiopathic or associated with a known inflammatory disease (ie multiple sclerosis, systemic lupus erythematosus, Sjogren's syndrome or neurosarcoidosis).16 Patients with TM should be offered immunomodulatory treatment such as steroids and plasmapheresis, although there is no consensus as to the most appropriate strategy yet.
Case report
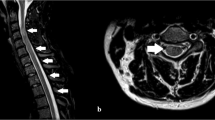
A 42-year-old female with undiagnosed Behcet's disease (BD) underwent a CT-guided nerve root injection to alleviate L2 radicular pain. She had a history of recurrent oral and genital ulcers, erythema nodosum and oligo-arthritis. She had noted frequent development of pustules and swelling at the site of venipuncture, consistent with a pathergy response. A 22-gauge needle was placed adjacent to the L2 nerve root and 1 cc of 0.5% Marcaine containing 6 mg of Celestone was injected. Within 8 h, the patient began to experience bilateral lower extremity weakness, urinary urgency and paresthesias, extending rostrally to a T12 level. Spinal cord MRI the following day (see Figure 1) revealed edematous changes with gadolinium enhancement from the conus medullaris rostrally to T9. Brain and cervical spine MRI studies were normal. Intravenous dexamethasone therapy was instituted and the patient made a significant, albeit incomplete recovery. The MRI abnormalities resolved completely.
(a) Sagittal T2 MRI of the thoracolumbar spinal column revealed abnormally increased T2 signal within the spinal cord from T9 to the conus (arrow indicates the lesion in the spinal cord and the asterisks denote the T11 and T10 vertebral bodies. (b) Sagittal T1 MRI after administration of gadolinium reveals intramedullary enhancement from T11 to the conus. Arrow indicates the lesion in the spinal cord and the asterisks denote the T11 and T10 vertebral bodies. (c) Coronal T1 MRI after administration of gadolinium also reveals inflammation within the spinal cord from T11 to the conus
Discussion
The temporal and spatial relationship of the injection to TM suggests a causal relationship. We suggest that just as the skin is hypersensitive to a needle prick in patients with BD (pathergy), the spinal cord may be hypersensitive to a local needle prick. The TM in this patient likely represents the consequence of this hypersensitivity. Other studies have suggested that heightened inflammatory responses in BD patients may lead to frequent postoperative complications.17, 18, 19 However, this is the first report of nBD presenting after local spinal needle puncture and, if confirmed in larger series, may suggest caution in recommending such procedures to patients with untreated BD.
An alternative explanation is that the clinical manifestations were instead due to vascular or traumatic myelopathy. Indeed, a recent report presented three cases of paraplegia or paraparesis following nerve root injection.20 However, in those cases, the neurologic impairment occurred immediately after injection, suggesting a vascular etiology. In our case, neurologic symptoms did not begin until many hours after the procedure making a vascular cause less likely. Further, while the patients presented by Houten and Errico20 were left with persistent motor deficits, our patient had complete motor recovery, again suggesting a distinct etiology. Finally, in our case, there was no evidence of intraparenchymal blood, so direct needle trauma to the spinal cord was unlikely.
We suggest that physicians use caution in offering spinal anesthetic treatments for patients with BD as such treatment may induce ‘pathergy of the spinal cord’.
References
Lee LA . Behcet disease. Semin Cutan Med Surg 2001; 20: 53–57.
Direskeneli H, Saruhan-Direskeneli G . The role of heat shock proteins in Behcet's disease. Clin Exp Rheumatol 2003; 21: S44–S48.
Ben Ahmed M, Houman H, Miled M, Dellagi K, Louzir H . Involvement of chemokines and Th1 cytokines in the pathogenesis of mucocutaneous lesions of Behcet's disease. Arthritis Rheum 2004; 50: 2291–2295.
International Study Group for Behcet's Disease. Criteria for diagnosis of Behcet's disease. Lancet 1990; 335: 1078–1080.
O'duffy JD . Summary of International Symposium on Behcet's Disease. Istanbul, September 29–30, 1977. J Rheumatol 1978; 5: 229–233.
Yazici H, Chamberlain MA, Tuzun Y, Yurdakul S, Muftuoglu A . A comparative study of the pathergy reaction among Turkish and British patients with Behcet's disease. Ann Rheum Dis 1984; 43: 74–75.
Inaloz HS, Evereklioglu C, Unal B, Kirtak N, Eralp A, Inaloz SS . The significance of immunohistochemistry in the skin pathergy reaction of patients with Behcet's syndrome. J Eur Acad Dermatol Venereol 2004; 18: 56–61.
Akman-Demir G, Serdaroglu P, Tasci B . Clinical patterns of neurological involvement in Behcet's disease: evaluation of 200 patients. The Neuro-Behcet Study Group. Brain 1999; 122 (Part 11): 2171–2182.
Berman M, Feldman S, Alter M, Zilber N, Kahana E . Acute transverse myelitis: incidence and etiologic considerations. Neurology 1981; 31: 966–971.
Jeffery DR, Mandler RN, Davis LE . Transverse myelitis. Retrospective analysis of 33 cases, with differentiation of cases associated with multiple sclerosis and parainfectious events. Arch Neurol 1993; 50: 532–535.
Christensen PB, Wermuth L, Hinge HH, Bomers K . Clinical course and long-term prognosis of acute transverse myelopathy. Acta Neurol Scand 1990; 81: 431–435.
Altrocchi PH . Acute transverse myelopathy. Arch Neurol 1963; 9: 21–29.
Misra UK, Kalita J, Kumar S . A clinical, MRI and neurophysiological study of acute transverse myelitis. J Neurol Sci 1996; 138: 150–156.
Lipton HL, Teasdall RD . Acute transverse myelopathy in adults. A follow-up study. Arch Neurol 1973; 28: 252–257.
Sakakibara R, Hattori T, Yasuda K, Yamanishi T . Micturition disturbance in acute transverse myelitis. Spinal Cord 1996; 34: 481–485.
Transverse Myelitis Consortium Working Group. Proposed diagnostic criteria and nosology of acute transverse myelitis. Neurology 2002; 59: 499–505.
Yazici H, Yurdakul S, Hamuryudan V . Behcet disease. Curr Opin Rheumatol 2001; 13: 18–22.
Ando M, Okita Y, Sasako Y, Kobayashi J, Tagusari O, Kitamura S . Surgery for aortic regurgitation caused by Behcet's disease: a clinical study of 11 patients. J Card Surg 1999; 14: 116–121.
Ando M, Kosakai Y, Okita Y, Nakano K, Kitamura S . Surgical treatment of Behcet's disease involving aortic regurgitation. Ann Thorac Surg 1999; 68: 2136–2140.
Houten JK, Errico TJ . Paraplegia after lumbosacral nerve root block: report of three cases. Spine J 2002; 2: 70–75.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Deshpande, D., Krishnan, C. & Kerr, D. Transverse myelitis after lumbar steroid injection in a patient with Behcet's disease. Spinal Cord 43, 735–737 (2005). https://doi.org/10.1038/sj.sc.3101779
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101779