Abstract
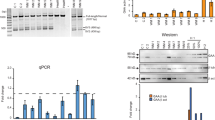
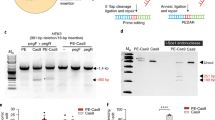
Deficiency in acid α-D-glucosidase results in Pompe's disease. Modified single-stranded oligonucleotide (ODN) was designed to correct the acid α-D-glucosidase gene with a C1935 → A (Asp → Glu) point mutation which causes a complete loss of enzymatic activity for glycogen digestion in the lysosome. The ODN vectors contained a stretch of normal oligonucleotide flanked by phosphorothioated sequences. The 25mer and 35mer ODNs were homologous to the target sequence, except for a mismatched base in the middle. The ODNs caused permanent and inheritable restoration of acid α-D-glucosidase activity in skin fibroblast cells carrying this mutation derived from a Pompe's disease patient. Gene correction was confirmed by amplification refractory mutation system-PCR (ARMS-PCR), restriction fragment length polymorphism (RFLP) and direct DNA cloning and sequencing. The increased acid α-D-glucosidase activity was detected using 4-MUG as the artificial substrate. The correction efficiency, ranging from 0.5 to 4%, was dependent on the length and polarity of the MSSOV used, the optimal design being a sense-strand 35mer ODNs. Repeated treatment of the mutant fibroblast cells with the ODNs substantially increased correction. We also constructed ODN vectors to trigger specific and in vivo nonsense mutation in the mouse acid α-D-glucosidase gene. The ODNs were in complex with YEEE-K18, an asialoglycoprotein-receptor ligand tagged with polylysine and targeted to hepatocytes and renal cells in vivo through intravenous injection. The mutated genotype was detected in the liver and the kidney by ARMS-PCR and glycogen accumulation in the lysosome of the liver cells. The studies demonstrate the utility of single-stranded ODN to direct targeted gene correction or mutation in a human hereditary disease and in an animal model. Our data open the possibility of developing ODN vector as a therapeutic approach for treatment of human hereditary diseases caused by point mutation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Pompe JC . Over idiopatische hypertrophic van het hart. Ned Tijdschr Geneeskd 1932; 76: 304–311.
Hers HG . Alpha-glucosidase deficiency in generalized glycogen storage disease. Biochem J 1963; 86: 11–16.
Loonen MC et al. Identification of heterozyotes for glycogenosis 2. Clin Genet 1981; 19: 55–63.
Gillette PC, Nihill MR, Singer DB . Electrophysiological mechanism of the short PR interval in Pompe disease. Am J Dis Child 1974; 12: 622–626.
Metzl JD, Elias ER, Berul CI . An interesting case of infant sudden death: severe hypertrophic cardiomyopathy in Pompe's disease. Pacing Clin Electrophysiol 1999; 22: 821–822.
Lin CY . Prenatal diagnosis of glycogen storage disease type II. The 100th Scientific Meeting of the Pediatric Association of the Republic of China 1984.
Engel AG . Acid maltase deficiency. In: Engel AG, Banker BQ (eds) Basic and Clinical Myology. McGraw-Hill: New York, 1986, pp 1629–1650.
Nicolino MP et al. Adenovirus-mediated transfer of the acid alpha-glucosidase gene into fibroblasts, myoblasts and myotubes from patients with glycogen storage disease type II leads to high level expression of enzyme and corrects glycogen accumulation. Hum Mol Genet 1998; 7: 1695–1702.
Van den Hout H et al. Recombinant human alpha-glucosidase from rabbit milk in Pompe patients. Lancet 2000; 356: 397–398.
Van den Hout JM et al. Enzyme therapy for Pompe disease with recombinant human alphaglucosidase from rabbit milk. J Inherit Metab Dis 2001; 24: 266–274.
Amalfitano A et al. Recombinant human acid alpha-glucosidase enzyme therapy for infantile glycogen storage disease type II: results of a phase I/II clinical trial. Genet Med 2001; 3: 132–138.
Moerschell RP, Tsunasawa S, Sherman F . Transformation of yeast with synthetic oligonucleotides. Proc Natl Acad Sci USA 1998; 85: 524–528.
Yamamoto T et al. Strand-specificity in the transformation of yeast with synthetic oligonucleotides. Genetics 1992; 131: 811–819.
Igoucheva O, Alexeev V, Yoon K . Targeted gene correction by small single-stranded oligonucleotides in mammalian cells. Gene Therapy 2001; 8: 391–399.
Liu L, Rice MC, Kmiec EB . In vivo gene repair of point and frameshift mutations directed by chimeric RNA/DNA oligonucleotides and modified single-stranded oligonucleotides. Nucleic Acids Res 2001; 29: 4238–4250.
Parekh-Olmedo H, Czymmek K, Kmiec EB . Targeted gene repair in mammalian cells using chimeric RNA/DNA oligonucleotides and modified single-stranded vectors. Sci STKE 2001; 2001: PL1.
Lin CY, Hwang B, Hsiao KJ, Jin YR . Study of α-D-glucosidase activity in patients with Pompe's disease. J Formosan Med Assoc 1986; 85: 766–770.
Lin CY, Shieh JJ . Molecular study on the infantile form of Pompe disease in Chinese in Taiwan. Acta Paed Sin 1996; 37: 115–121.
Jiaang WT, Tseng PH, Chen ST . Facile solid phase synthesis of YEE(ah-GalNAc)3, a ligand with known high affinity for the asialoglycoprotein receptor. Synletter 2000; 6: 797–800.
Hangeland JJ, Levis TT, Lee YC, Ts'o POP . Cell-type specific and ligand specific enhancement of cellular uptake of oligonucleoside methylphosphonates covalently linked with a neoglycopeptide, YEE(ah-GalNAc)3 . Bioconjugate Chem 1995; 6: 695–701.
Sambrook J, Fritsch EF, Maniatis T . Molecular Cloning, A Laboratory Use 2nd edn. 1989, pp. I.90–I.104.
David A et al. Alpha-1,4-glucosidase activity in Pompe's disease. FASEB J 2002; 16: 754–756.
Geffen I, Spiess M . Asialoglycoprotein receptor. Int Rev Cytol 1992; 137B: 181–219.
Plank C et al. Gene transfer into hepatocytes using asialoglycoprotein receptor mediated endocytosis of DNA complexed with an artificial tetra-antennary galactose ligand. Bioconjugate Chem 1992; 3: 533–539.
Seow YY, Tan MG, Woo KT . Expression of a functional asialoglycoprotein receptor in human renal proximal tubular epithelial cells. Nephron 2002; 91: 431–438.
Hu H, Serra D, Amalfitano A . Persistence of an [E1-,polymerase-] adenovirus vector despite transduction of a neoantigen into immune-competent mice. Hum Gene Ther 1999; 10: 355–364.
Amalfitano A et al. Systemic correction of the muscle disorder glycogen storage disease type II after hepatic targeting of a modified adenovirus vector encoding human acid-alpha-glucosidase. Proc Natl Acad Sci USA 1999; 96: 8861–8866.
Acknowledgements
This work was supported by NSC grants. We thank Dr Kong-Bung Choo for critically reading the manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lu, IL., Lin, CY., Lin, SB. et al. Correction/mutation of acid α-D-glucosidase gene by modified single-stranded oligonucleotides: in vitro and in vivo studies. Gene Ther 10, 1910–1916 (2003). https://doi.org/10.1038/sj.gt.3302096
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302096