Abstract
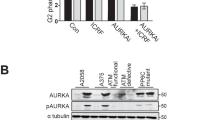
The rare diseases ataxia-telangiectasia (AT), caused by mutations in the ATM gene, and Nijmegen breakage syndrome (NBS), with mutations in the p95/nbs1 gene, share a variety of phenotypic abnormalities such as chromosomal instability, radiation sensitivity and defects in cell-cycle checkpoints in response to ionizing radiation1,2,3,4. The ATM gene encodes a protein kinase that is activated by ionizing radiation or radiomimetic drugs5,6, whereas p95/nbs1 is part of a protein complex that is involved in responses to DNA double-strand breaks3,7. Here, because of the similarities between AT and NBS, we evaluated the functional interactions between ATM and p95/nbs1. Activation of the ATM kinase by ionizing radiation and induction of ATM-dependent responses in NBS cells indicated that p95/nbs1 may not be required for signalling to ATM after ionizing radiation. However, p95/nbs1 was phosphorylated on serine 343 in an ATM-dependent manner in vitro and in vivo after ionizing radiation. A p95/nbs1 construct mutated at the ATM phosphorylation site abrogated an S-phase checkpoint induced by ionizing radiation in normal cells and failed to compensate for this functional deficiency in NBS cells. These observations link ATM and p95/nbs1 in a common signalling pathway and provide an explanation for phenotypic similarities in these two diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Carney,J. P. et al. The hMre11/hRad50 protein complex and Nijmegen breakage syndrome: linkage of double-strand break repair to the cellular DNA damage response. Cell 93, 477–486 (1998).
Varon,R. et al. Nibrin, a novel DNA double-strand break repair protein, is mutated in Nijmegen breakage syndrome. Cell 93, 467–476 (1998).
Featherstone,C. & Jackson,S. P. DNA repair: the Nijmegen breakage syndrome protein. Curr. Biol. 8, R622–R625 (1998).
Shiloh,Y. Ataxia-telangiectasia and the Nijmegen breakage syndrome: related disorders but genes apart. Annu. Rev. Genet. 31, 635 –662 (1997).
Canman,C. E. et al. Activation of the ATM kinase by ionizing radiation and phosphorylation of p53. Science 281, 1677– 1679 (1998).
Banin,S. et al. Enhanced phosphorylation of p53 by ATM in response to DNA damage. Science 281, 1674–1677 (1998).
Petrini,J. H. The mammalian Mre11-Rad50-nbs1 protein complex: integration of functions in the cellular DNA-damage response. Am. J. Hum. Genet. 64, 1264–1269 (1999).
Kastan,M. B. et al. A mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is defective in ataxia-telangiectasia. Cell 71, 587–597 (1992).
Canman,C. E., Wolff,A. C., Chen,C. Y., Fornace,A. J. Jr & Kastan,M. B. The p53-dependent G1 cell cycle checkpoint pathway and ataxia-telangiectasia. Cancer Res. 54, 5054–5058 (1994).
Khanna,K. K. et al. Nature of G1/S cell cycle checkpoint defect in ataxia-telangiectasia. Oncogene 11, 609–618 (1995).
Siliciano,J. D. et al. DNA damage induces phosphorylation of the amino terminus of p53. Genes Dev. 11, 3471– 3481 (1997).
Jongmans,W. et al. Nijmegen breakage syndrome cells fail to induce the p53-mediated DNA damage response following exposure to ionizing radiation. Mol. Cell. Biol. 17, 5016–5022 (1997).
Yamazaki,V., Wegner,R. D. & Kirchgessner, C. U. Characterization of cell cycle checkpoint responses after ionizing radiation in Nijmegen breakage syndrome cells. Cancer Res. 58, 2316–2322 (1998).
Hartley,K. O. et al. DNA-dependent protein kinase catalytic subunit: a relative of phosphatidylinositol 3-kinase and the ataxia telangiectasia gene product. Cell 82, 849–856 (1995).
Cimprich,K. A., Shin,T. B., Keith,C. T. & Schreiber,S. L. cDNA cloning and gene mapping of a candidate human cell cycle checkpoint protein. Proc. Natl Acad. Sci. USA 93, 2850– 2855 (1996).
Kim,S. T., Lim,D. S., Canman,C. E. & Kastan,M. B. Substrate specificities and identification of putative substrates of ATM kinase family members. J. Biol. Chem. 274, 37538–37543 (1999).
Nelms,B. E., Maser,R. S., MacKay,J. F., Lagally,M. G. & Petrini,J. H. In situ visualization of DNA double-strand break repair in human fibroblasts. Science 280, 590–592 (1998).
Trujillo,K. M., Yuan,S. S., Lee,E. Y. & Sung,P. Nuclease activities in a complex of human recombination and DNA repair factors Rad50, Mre11, and p95. J. Biol. Chem. 273, 21447– 21450 (1998).
Paull,T. T. & Gellert,M. Nbs1 potentiates ATP-driven DNA unwinding and endonuclease cleavage by the Mre11/Rad50 complex. Genes Dev. 13, 1276–1288 ( 1999).
Maser,R. S., Monsen,K. J., Nelms,B. E. & Petrini,J. H. hMre11 and hRad50 nuclear foci are induced during the normal cellular response to DNA double-strand breaks. Mol. Cell. Biol. 17, 6087–6096 (1997).
Maser,R. S., Mressan,D. A. & Petrini, J. H. The Mre11-Rad50 Complex: Diverse Functions in the Cellular DNA Damage Response (Humana, Totowa, 1999).
Khosravi,R. et al. Rapid ATM-dependent phosphorylation of MDM2 precedes p53 accumulation in response to DNA damage. Proc. Natl Acad. Sci. USA 96, 14973–14977 (1999).
Matsuoka,S., Huang,M. & Elledge,S. J. Linkage of ATM to cell cycle regulation by the Chk2 protein kinase. Science 282, 1893– 1897 (1998).
Brown,A. L. et al. A human Cds1-related kinase that functions downstream of ATM protein in the cellular response to DNA damage. Proc. Natl Acad. Sci. USA 96, 3745–3750 (1999).
Cortez,D., Wang,Y., Qin,J. & Elledge,S. J. Requirement of ATM-dependent phosphorylation of brca1 in the DNA damage response to double-strand breaks. Science 286, 1162– 1166 (1999).
Zhong,Q. et al. Association of BRCA1 with the hRad50-hMre11-p95/nbs1 complex and the DNA damage response. Science 285, 747–750 (1999).
Ziv,Y. et al. Recombinant ATM protein complements the cellular A-T phenotype. Oncogene 15, 159–167 (1997).
Morgan,S. E., Lovly,C., Pandita,T. K., Shiloh,Y. & Kastan,M. B. Fragments of ATM which have dominant-negative or complementing activity. Mol. Cell. Biol. 17, 2020– 2029 (1997).
Acknowledgements
We thank T. M. Gilmer and K. Kilpatrick (Glaxo-Wellcome) for the mouse ATM monoclonal antibodies; Y. Shiloh for AT complemented cell lines; C. Bakkenist, K. McLure and C. Canman for helpful comments on the manuscript; and D. Woods and A. Justman for technical assistance. This work was supported by grants from the NIH (M.B.K, the St. Jude Cancer Centre Core Grant, J.H.J.P. and J.L.). M.B.K. was also supported by Glaxo-Wellcome and the American Lebanese Syrian Associated Charities (ALSAC) of the St. Jude Children's Research Hospital, and J.H.J.P. by the Milwaukee Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lim, DS., Kim, ST., Xu, B. et al. ATM phosphorylates p95/nbs1 in an S-phase checkpoint pathway. Nature 404, 613–617 (2000). https://doi.org/10.1038/35007091
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35007091
This article is cited by
-
Protein post-translational modifications in the regulation of cancer hallmarks
Cancer Gene Therapy (2023)
-
miR-4796 enhances the sensitivity of breast cancer cells to ionising radiation by impairing the DNA repair pathway
Breast Cancer (2023)
-
Caspase-2 regulates S-phase cell cycle events to protect from DNA damage accumulation independent of apoptosis
Oncogene (2022)
-
Stress-sensing in the human greying hair follicle: Ataxia Telangiectasia Mutated (ATM) depletion in hair bulb melanocytes in canities-prone scalp
Scientific Reports (2020)
-
NBS1 interacts with HP1 to ensure genome integrity
Cell Death & Disease (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.