Abstract
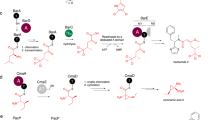
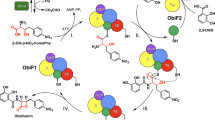
In the biosynthesis of many macrocyclic natural products by multidomain megasynthases, a carboxy-terminal thioesterase (TE) domain is involved in cyclization and product release1,2; however, it has not been determined whether TE domains can catalyse macrocyclization (and elongation in the case of symmetric cyclic peptides) independently of upstream domains. The inability to decouple the TE cyclization step from earlier chain assembly steps has precluded determination of TE substrate specificity, which is important for the engineered biosynthesis of new compounds1. Here we report that the excised TE domain from tyrocidine synthetase efficiently catalyses cyclization of a decapeptide-thioester to form the antibiotic tyrocidine A, and can catalyse pentapeptide-thioester dimerization followed by cyclization to form the antibiotic gramicidin S. By systematically varying the decapeptide-thioester substrate and comparing cyclization rates, we also show that only two residues (one near each end of the decapeptide) are critical for cyclization. This specificity profile indicates that the tyrocidine synthetase TE, and by analogy many other TE domains, will be able to cyclize and release a broad range of new substrates and products produced by engineered enzymatic assembly lines.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cane, D. E., Walsh, C. T. & Khosla, C. Harnessing the biosynthetic code: combinations, permutations and mutations. Science 282, 63– 68 (1998).
Marahiel, M. A., Stachelhaus, T. & Mootz, H. D. Modular peptide synthetases involved in nonribosomal peptide synthesis. Chem. Rev. 97, 2651– 2674 (1997).
Lambalot, R. H. et al. A new enzyme superfamily–the phosphopantetheinyl-transferases. Chem. Biol. 3, 923–936 (1996).
Lawson, D. M. et al. Structure of a myristoyl-ACP-specific thioesterase from Vibrio harveyi. Biochemistry 33, 9382 –9388 (1994).
Li, J., Szittner, R., Derewenda, Z. S. & Meighen, E. A. Conversion of serine-114 to cysteine-114 and the role of the active site nucleophile in acyl transfer by myristoyl-ACP thioesterase from Vibrio harveyi. Biochemistry 35, 9967– 9973 (1996).
Shaw-Reid, C. A. et al. Assembly line enzymology by multimodular nonribosomal peptide synthetases: the thioesterase domain of E. coli EntF catalyses both elongation and cyclolactonization. Chem. Biol. 6, 385–400 (1999).
Aggarwal, R., Caffrey, P., Leadlay, P. F., Smith, C. J. & Staunton, J. The thioesterase domain of the erythromycin-producing polyketide synthase: mechanistic studies in vitro to investigate its mode of action and substrate specificity. J. Chem. Soc. Chem. Comm. 15, 1519– 1520 (1995).
Gokhale, R. S., Hunziker, D., Cane, D. E. & Khosla, C. Mechanism and specificity of the terminal thioesterase domain from the erythromycin polyketide synthase. Chem. Biol. 6, 117– 125 (1998).
Cortes, J. et al. Repositioning of a domain in a modular polyketide synthase to promote specific chain cleavage. Science 268, 1487–1489 (1995).
Kao, C. M. et al. Gain of function mutagenesis of the erythromycin polyketide synthase. 2. Engineered biosynthesis of an eight-membered ring tetraketide lactone. J. Am. Chem. Soc. 119, 11339– 11340 (1997).
Jacobsen, J. R., Hutchinson, C. R., Cane, D. E. & Khosla, C. Precursor-directed biosynthesis of erythromycin analogs by an engineered polyketide synthase. Science 277, 367– 369 (1997).
McDaniel, R. et al. Multiple genetic modifications of the erythromycin polyketide synthase to produce a library of novel “unnatural” natural products. Proc. Natl Acad. Sci. USA 96, 1846– 1851 (1999).
Tang, L., Fu, H. & McDaniel, R. Formation of functional heterologous complexes using subunits from the picromycin, erythromycin and oleandomycin polyketide synthases. Chem. Biol. 7, 77–84 ( 2000).
Xue, Y. & Sherman, D. H. Alternative modular polyketide synthase expression controls macrolactone structure. Nature 403, 571–575 (2000).
Stachelhaus, T., Schneider, A. & Marahiel, M. A. Rational design of peptide antibiotics by targeted replacement of bacterial and fungal domains. Science 269, 69–72 (1995).
de Ferra, F., Rodriguez, F., Tortora, O., Tosi, C. & Grandi, G. Engineering of peptide synthetases. J. Biol. Chem 272, 25304– 25309 (1997).
Mootz, H. D., Schwarzer, D. & Marahiel, M. A. Construction of hybrid peptide synthetases by module and domain fusions. Proc. Natl Acad. Sci. USA 97, 5848–5853 (2000).
Belshaw, P. J., Walsh, C. T. & Stachelhaus, T. Aminoacyl-CoAs as probes of condensation domain selectivity in nonribosomal peptide synthesis. Science 284, 486–489 (1999).
Mootz, H. D. & Marahiel, M. A. The tyrocidine biosynthesis operon of Bacillus brevis: complete nucleotide sequence and biochemical characterization of functional internal adenylation domains. J. Bacteriol. 179, 6843–6850 ( 1997).
Roskoski, R., Kleinkauf, H., Gevers, W. & Lipmann, F. Isolation of enzyme-bound peptide intermediates in tyrocidine biosynthesis. Biochemistry 9, 4846–4851 (1970).
Hori, K. et al. Molecular cloning and nucleotide sequence of the gramicidin S synthetase 1 gene. J. Biochem. (Tokyo) 106, 639–645 (1989).
Turgay, K., Krause, M. & Marahiel, M. A. Four homologous domains in the primary structure of GrsB are related to domains in a superfamily of adenylate-forming enzymes. Mol. Microbiol. 6, 529– 546 (1992).
Boger, D. L. & Ichikawa, S. Total syntheses of thiocoraline and BE-22179: establishment of relative and absolute stereochemistry. J. Am. Chem. Soc. 122, 2956–2957 (2000).
Jackson, D. Y. et al. A designed peptide ligase for total synthesis of ribonuclease A with unnatural catalytic residues. Science 266, 243–247 (1994).
Acknowledgements
This work was supported by grants from the NIH (to C.T.W.), the Deutsche Forschungsgemeinschaft and the Fonds der Chemischen Industrie (to M.A.M.), a NIH postdoctoral fellowship (to J.W.T.) and a PhD fellowship of the Stiftung Stipendien-Fonds des Verbandes der Chemischen Industrie (to H.D.M.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Trauger, J., Kohli, R., Mootz, H. et al. Peptide cyclization catalysed by the thioesterase domain of tyrocidine synthetase. Nature 407, 215–218 (2000). https://doi.org/10.1038/35025116
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35025116
This article is cited by
-
Chemoenzymatic tandem cyclization for the facile synthesis of bicyclic peptides
Communications Chemistry (2024)
-
Discovery and biosynthesis of tricyclic copper-binding ribosomal peptides containing histidine-to-butyrine crosslinks
Nature Communications (2023)
-
Heterochiral coupling in non-ribosomal peptide macrolactamization
Nature Catalysis (2020)
-
Trapping biosynthetic acyl-enzyme intermediates with encoded 2,3-diaminopropionic acid
Nature (2019)
-
Refining and expanding nonribosomal peptide synthetase function and mechanism
Journal of Industrial Microbiology and Biotechnology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.