Key Points
-
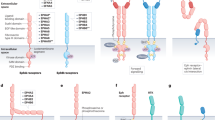
The four ErbB receptors and their many neuregulins and EGF-like ligands form a layered signalling network. The network structure allows the diversification, tuning and robustness of cell-to-cell signalling.
-
The network evolved from a simple signalling module that controls inductive morphogenesis in invertebrates. In mammals, specific ligands and their respective homo- or heterodimeric ErbB complexes specify different cell lineages.
-
Oncogenic animal viruses harness the network through diverse molecular processes that promote ErbB signalling or prevent it from being switched off.
-
Autocrine loops, mutant ErbB1 molecules and enhanced expression of ErbB receptors are frequently observed in human cancers of epithelial and neuronal origins. Most frequent is overexpression of ErbB2, a ligandless co-receptor that amplifies ErbB signalling.
-
Current attempts to block the network in human disease include small-molecule inhibitors of tyrosine kinases and chaperones, and various gene-therapy strategies. But immunotherapy directed at ErbB2 is already widely used, in combination with chemotherapy, to inhibit metastasizing breast cancers.
-
Future pharmacological advances and deeper understanding of the network will allow selective inhibition or activation of its many routes, with the aim of curing neuronal and skin disorders, as well as cancer.
Abstract
When epidermal growth factor and its relatives bind the ErbB family of receptors, they trigger a rich network of signalling pathways, culminating in responses ranging from cell division to death, motility to adhesion. The network is often dysregulated in cancer and lends credence to the mantra that molecular understanding yields clinical benefit: over 25,000 women with breast cancer have now been treated with trastuzumab (Herceptin®), a recombinant antibody designed to block the receptor ErbB2. Likewise, small-molecule enzyme inhibitors and monoclonal antibodies to ErbB1 are in advanced phases of clinical testing. What can this pathway teach us about translating basic science into clinical use?
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Burden, S. & Yarden, Y. Neuregulins and their receptors: a versatile signalling module in organogenesis and oncogenesis. Neuron 18, 847–855 ( 1997).
Borg, J. -P. et al. ERBIN: a basolateral PDZ protein that interacts with the mammalian ERBB2/HER2 receptor. Nature Cell Biol. 2, 407–414 (2000).
Monilola, A. O., Neve, R. M., Lane, H. A. & Hynes, N. E. The ErbB signaling network: receptor heterodimerization in development and cancer. EMBO J. 19, 3159–3167 ( 2000).
Baselga, J. et al. Phase II study of weekly intravenous recombinant humanized anti-p185HER2 monoclonal antibody in patients with HER2/neu-overexpressing metastatic breast cancer. J. Clin. Oncol. 14, 737–744 (1996).First full report of clinical tests of an anti-ErbB2 antibody as a single agent. Patients with metastatic breast cancer were intravenously treated with the recombinant drug. Toxicity was minimal and objective response was observed in several organs of a small group of patients.
Cobleigh, M. A. et al. Multinational study of the efficacy and safety of humanized anti-HER2 monoclonal antibody in women who have HER2-overexpressing metastatic breast cancer that has progressed after chemotherapy for metastatic disease . J. Clin. Oncol. 17, 2639– 2648 (1999).
Dickson, R. B. & Lippman, M. E. Estrogenic regulation of growth and polypeptide growth factor secretion in human breast carcinoma. Endocrin. Rev. 8, 29–43 (1987).
Prenzel, N. et al. EGF receptor transactivation by G-protein-coupled receptors requires metalloproteinase cleavage of proHB-EGF. Nature 402, 884–888 (1999).
Jones, J. T., Akita, R. W. & Sliwkowski, M. X. Binding specificities and affinities of EGF domains for ErbB receptors. FEBS Lett. 447, 227– 231 (1999).Relative binding affinities of the EGF domains of 11 ErbB ligands were measured on 6 ErbB receptor combinations using soluble receptors. This format allowed precise determination of the effect of heterodimerization on ligand affinity and specificity.
Tzahar, E. et al. Bivalence of EGF-like ligands drives the ErbB signalling network . EMBO J. 16, 4938–4950 (1997).
Landgraf, R. & Eisenberg, D. Heregulin reverses the oligomerization of HER3. Biochemistry 39, 8503– 8511 (2000).
Ferguson, K. M., Darling, P. J., Mohan, M. J., Macatee, T. L. & Lemmon, M. A. Extracellular domains drive homo- but not heterodimerization of erbB receptors. EMBO J. 19, 4632–4643 (2000).
Guy, P. M., Platko, J. V., Cantley, L. C., Cerione, R. A. & Carraway, K. L. Insect cell-expressed p180ErbB3 possesses an impaired tyrosine kinase activity. Proc. Natl Acad. Sci. USA 91, 8132–8136 ( 1994).
Klapper, L. N. et al. The ErbB2/HER2 oncoprotein of human carcinomas may function solely as a shared coreceptor for multiple stroma-derived growth factors. Proc. Natl Acad. Sci. USA 96, 4995– 5000 (1999).
Tzahar, E. et al. A hierarchical network of interreceptor interactions determines signal transduction by Neu differentiation factor/neuregulin and epidermal growth factor. Mol. Cell. Biol. 16, 5276 –5287 (1996).
Graus Porta, D., Beerli, R. R., Daly, J. M. & Hynes, N. E. ErbB2, the preferred heterodimerization partner of all ErbB receptors, is a mediator of lateral signalling. EMBO J. 16, 1647–1655 (1997).
Elenius, K. et al. Characterization of a naturally occurring ErbB4 isoform that does not bind or activate phosphatidyl inositol 3-kinase. Oncogene 18, 2607–2615 ( 1999).
Olayioye, M. A. et al. ErbB1 and ErbB2 acquire distinct signalling properties dependent upon their dimerization partner. Mol. Cell. Biol. 18 , 5042–5051 (1998).
Soltoff, S. P. & Cantley, L. C. p120cbl is a cytosolic adapter protein that associates with phosphoinositide 3-kinase in response to epidermal growth factor in PC12 and other cells. J. Biol. Chem. 271, 563–567 (1996).
Schaeffer, L., Duclert, N., Huchet Dymanus, M. & Changeux, J. P. Implication of a multisubunit Ets-related transcription factor in synaptic expression of the nicotinic acetylcholine receptor. EMBO J. 17, 3078–3090 (1998).
Fedi, P., Pierce, J., Di Fiore, P. P. & Kraus, M. H. Efficient coupling with phosphatidylinositol 3-kinase, but not phospholipase Cγ or GTPase-activating protein, distinguishes ErbB3 signalling from that of other ErbB/EGFR family members. Mol. Cell. Biol. 14, 492–500 (1994).
Pinkas-Kramarski, R. et al. Diversification of Neu differentiation factor and epidermal growth factor signalling by combinatorial receptor interactions. EMBO J. 15, 2452–2467 ( 1996).References 21 and 22 describe analyses of signal transduction by individual ErbB proteins and their combinations expressed in isolation in myeloid cells. This allows the pan-ErbB stimulatory effect of ErbB2 to be demonstrated.
Riese, D. J. II, van Raaij, T. M., Plowman, G. D., Andrews, G. C. & Stern, D. F. The cellular response to neuregulins is governed by complex interactions of the ErbB receptor family. Mol. Cell. Biol. 15, 5770– 5776 (1995).
Kokai, Y. et al. Synergistic interaction of p185c-neu and the EGF receptor leads to transformation of rodent fibroblasts. Cell 58, 287–292 (1989).
Alimandi, M. et al. Cooperative signalling of ErbB3 and ErbB2 in neoplastic transformation of human mammary carcinoma cells. Oncogene 15, 1813–1821 (1995).
Wallasch, C. et al. Heregulin-dependent regulation of HER2/neu oncogenic signalling by heterodimerization with HER3. EMBO J. 14, 4267–4275 (1995).
Chausovsky, A. et al. Molecular requirements for the effect of neuregulin on cell spreading, motility and colony organization. Oncogene 19, 878–888 (2000).
Kainulainen, V. et al. A natural ErbB4 isoform that does not activate phosphoinositide 3-kinase mediates proliferation but not survival or chemotaxis. J. Biol. Chem. 275, 8641–8649 (2000).
Vaskovsky, A., Lupowitz, Z., Erlich, S. & Pinkas-Kramarski, R. ErbB4 activation promotes neurite outgrowth in PC12 cells. J. Neurochem. 74, 979–987 ( 2000).
Carpenter, G. Employment of the epidermal growth factor receptor in growth factor-independent signalling pathways. J. Cell Biol. 146, 697–702 (1999).
Daub, H., Wallasch, C., Lankenau, A., Herrlich, A. & Ullrich, A. Signal characteristics of G protein-transactivated EGF receptor. EMBO J. 16, 7032– 7044 (1997).Elucidation in cultured cells of crosstalk between G-protein-coupled receptors and ErbB signalling. Ectopic expression of G q - or G i -coupled receptors revealed the essential function of ErbB1 in downstream signalling of these G proteins to mitogen-activated protein kinases.
Luttrell, L. M., Della Rocca, G. J., van Biesen, T., Luttrell, D. K. & Lefkowitz, R. J. Gβγ subunits mediate Src-dependent phosphorylation of the epidermal growth factor receptor. A scaffold for G protein-coupled receptor-mediated Ras activation. J. Biol. Chem. 272, 4637–4644 ( 1997).
Dikic, I., Tokiwa, G., Lev, S., Courtneidge, S. A. & Schlessinger, J. A role for Pyk2 and Src in linking G-protein-coupled receptors with MAP kinase activation. Nature 383, 547–550 (1996).
Luttrell, L. M. et al. β-Arrestin-dependent formation of β2 adrenergic receptor–Src protein kinase complexes. Science 283, 655–661 (1999).
Yamauchi, T. et al. Tyrosine phosphorylation of the EGF receptor by the kinase Jak2 is induced by growth hormone. Nature 390, 91–96 (1997).
Wong, A. J. et al. Structural alterations of the epidermal growth factor receptor gene in human gliomas. Proc. Natl Acad. Sci. USA 89 , 2965–2969 (1992).
Qiu, Y., Ravi, L. & Kung, H. J. Requirement of ErbB2 for signalling by interleukin-6 in prostate carcinoma cells. Nature 393, 83 –85 (1998).
Miettinen, P. et al. Epithelial immaturity and multiorgan faliure in mice lacking epidermal growth factor receptor. Nature 376, 337–341 (1995).
Sibilia, M., Steinbach, J. P., Stingl, L., Aguzzi, A. & Wagner, E. F. A strain-independent postnatal neurodegeneration in mice lacking the EGF receptor. EMBO J. 17, 719–731 (1998).
Threadgill, D. W. et al. Targeted disruption of mouse EGF receptor: effect of genetic background on mutant phenotype. Science 269, 230–234 (1995).
Mann, G. et al. Mice with null mutations of the TGFα gene have abnormal skin architecture, wavy hair, and curly whiskers and often develop corneal inflammation. Cell 73, 249– 261 (1993).
Luetteke, N. C. et al. TGFα deficiency results in hair follicles and eye abnormalities in targeted and Waved-1 mice. Cell 73, 263 –278 (1993).
Liu, X. et al. Domain-specific gene disruption reveals critical regulation of neuregulin signalling by its cytoplasmic tail. Proc. Natl Acad. Sci. USA 95, 13024–13029 (1998).
Kramer, R. et al. Neuregulins with an Ig-like domain are essential for mouse myocardial and neuronal development. Proc. Natl Acad. Sci. USA 93, 4833–4838 ( 1996).
Britsch, S. et al. The ErbB2 and ErbB3 receptors and their ligand, neuregulin-1, are essential for development of the sympathetic nervous system. Genes Dev. 12, 1825–1836 (1998).By targeting ErbB2, ErbB3 and NRG1, the authors revealed that the ternary complex has an essential role in the developing sympathetic nervous system. Apparently, signalling by this complex drives migration of sympathetic cells from the neural crest.
Erickson, S. L. et al. ErbB3 is required for normal cerebellar and cardiac development: a comparison with ErbB2-and heregulin-deficient mice. Development 124, 4999–5011 ( 1997).
Riethmacher, D. et al. Severe neuropathies in mice with targeted mutations in the ErbB3 receptor. Nature 389, 725– 730 (1997).
Fischbach, G. D. & Rosen, K. M. ARIA: a neuromuscular junction neuregulin. Annu. Rev. Neurosci. 20, 429–458 (1997).
Ozaki, M., Sasner, M., Yano, R., Lu, H. S. & Buonanno, A. Neuregulin-β induces expression of an NMDA-receptor subunit. Nature 390, 691– 694 (1997).Synaptogenesis in the central nervous system involves marked changes in the composition of N -methyl- d -aspartate receptors. This in vitro study implies that neuregulins regulate the composition of the neurotransmitter receptor in maturing synapses in the brain, in a manner analogous to the neuromuscular junction.
Dong, J. et al. Metalloprotease-mediated ligand release regulates autocrine signalling through the epidermal growth factor receptor. Proc. Natl Acad. Sci. USA 96, 6235–6240 ( 1999).
Modjtahedi, H. & Dean, C. The receptor for EGF and its ligands: Expression, prognostic value and target for therapy in cancer. Int. J. Oncol. 4, 277– 296 (1994).
Salomon, D. S., Brandt, R., Ciardiello, F. & Normanno, N. Epidermal growth factor-related peptides and their receptors in human malignancies . Crit. Rev. Oncol. Hematol. 19, 183– 232 (1995).
Scher, H. I. et al. Changing pattern of expression of the epidermal growth factor receptor and transforming growth factor-α in the progression of prostatic neoplasms. Clin. Cancer Res. 1, 545– 550 (1995).
Wikstrand, C. J., Reist, C. J., Archer, G. E., Zalutsky, M. R. & Bigner, D. D. The class III variant of the epidermal growth factor receptor (EGFRvIII): characterization and utilization as an immunotherapeutic target. J. Neurovirol. 4, 148–158 (1998).
Moscatello, D. K. et al. Frequent expression of a mutant epidermal growth factor receptor in multiple human tumours. Cancer Res. 55, 5536–5539 (1995).
Slamon, D. J. et al. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235 , 177–182 (1987). First demonstration of the prognostic value of ErbB2 amplification in breast cancer. Gene amplification was correlated with several disease parameters, and was a significant predictor of patient survival and time to relapse.
Ross, J. S. & Fletcher, J. A. The HER-2/neu oncogene in breast cancer: prognostic factor, predictive factor, and target for therapy. Stem Cells 16, 413–428 ( 1998).
Paik, S. & Liu, E. T. HER2 as a predictor of therapeutic response in breast cancer. Breast Dis. 11, 91–102 (2000).
Press, M. F. et al. HER-2/neu gene amplification characterized by fluorescence in situ hybridization: poor prognosis in node-negative breast carcinomas . J. Clin. Oncol. 15, 2894– 2904 (1997).
Muss, H. B. c-erbB-2 expression and response to adjuvant therapy in women with node-positive early breast cancer. N. Engl. J. Med. 330, 1260–1266 (1994). First evidence that ErbB2 can predict therapeutic response in breast cancer. Using tissue blocks obtained from patients treated with adjuvant chemotherapy, the authors found that patients randomly assigned to a high-dose regimen of adjuvant chemotherapy had significantly longer disease-free and overall survival if their tumours overexpressed c-ErbB2.
Borg, A. et al. ERBB2 amplification is associated with tamoxifen resistance in steroid-receptor positive breast cancer. Cancer Lett. 81, 137–144 (1994).
Carlomagno, C. et al. c-erb B2 overexpression decreases the benefit of adjuvant tamoxifen in early-stage breast cancer without axillary lymph node metastases . J. Clin. Oncol. 14, 2702– 2708 (1996).
Pietras, R. J. HER-2 tyrosine kinase pathway targets estrogen receptor and promotes hormone-independent growth in human breast cancer cells. Oncogene 10, 2435–2446 (1995).
Giani, C. et al. Increased expression of c-ErbB2 in hormone-dependent breast cancer cells inhibits cell growth and induces differentiation. Oncogene 17, 425–432 ( 1998).
Xia, W. et al. Combination of EGFR, HER-2/neu, and HER-3 is a stronger predictor for the outcome of oral squamous cell carcinoma than any individual family members. Clin. Cancer Res. 5, 4164– 4174 (1999).
Lyne, J. C. et al. Tissue expression of neu differentiation factor/heregulin and its receptor complex in prostate cancer and its biologic effects on prostate cancer cells in vitro. Cancer J. Sci. Am. 3, 21–30 (1997).
Kew, T. Y. et al. c-ErbB4 protein expression in human breast cancer. Br. J. Cancer 82, 1163–1170 (2000).
Gilbertson, R. J., Perry, R. H., Kelly, P. J., Pearson, A. D. & Lunec, J. Prognostic significance of HER2 and HER4 co-expression in childhood medulloblastoma. Cancer Res. 57, 3272–3280 (1997).
Sliwkowski, M. X. et al. Nonclinical studies addressing the mechanism of action of trastuzumab (Herceptin). Semin. Oncol. 26, 60–70 (1999).
Clynes, R. A., Towers, T. L., Presta, L. G. & Ravetch, J. V. Inhibitory Fc receptors modulate in vivo cytotoxicity against tumor targets. Nature Med. 6, 443– 446 (2000).
Disis, M. L. & Cheever, M. A. HER-2/neu protein: a target for antigen-specific immunotherapy of human cancer. Adv. Cancer Res. 71, 343–371 ( 1997).
Sampson, J. H. et al. Unarmed, tumour-specific monoclonal antibody effectively treats brain tumours. Proc. Natl Acad. Sci. USA 97, 7503–7508 (2000).
Wu, X. et al. Involvement of p27KIP1 in G1 arrest mediated by an anti-epidermal growth factor receptor monoclonal antibody. Oncogene 12, 1397–1403 (1996).
Levitzki, A. & Gazit, A. Tyrosine kinase inhibition: an approach to drug development. Science 267, 1782– 1788 (1995).
Fry, D. W. et al. Specific, irreversible inactivation of the epidermal growth factor receptor and erbB2, by a new class of tyrosine kinase inhibitor. Proc. Natl Acad. Sci. USA 95, 12022– 12027 (1998).
Brugge, J. S. New intracellular targets for drug design, Science 260, 918–919 (1993).
Chang, J. Y. et al. The tumour suppression activity of E1A in HER-2/neu-overexpressing breast cancer. Oncogene 14, 561– 568 (1997).
Beerli, R. R., Wels, W. & Hynes, N. E. Intracellular expression of single chain antibodies reverts ErbB2 transformation. J. Biol. Chem. 269, 23931–23936 (1994).
Alvarez, R. D. & Curiel, D. T. A phase I study of recombinant adenovirus vector-mediated delivery of an anti-ErbB2 single chain (sFv) antibody gene for previously treated ovarian and extraovarian cancer patients. Hum. Gene Ther. 8, 229– 242 (1997).
Ebbinghaus, S. W. et al. Triplex formation inhibits HER-2/neu transcription in vitro . J. Clin. Invest. 92, 2433– 2439 (1993).
Vaughn, J. P. et al. Antisense DNA downregulation of the ERBB2 oncogene measured by a flow cytometric assay. Proc. Natl Acad. Sci. USA 92, 8338–8342 (1995).
Hsieh, S. S. et al. ERBB-2 expression is rate-limiting for epidermal growth factor-mediated stimulation of ovarian cancer cell proliferation. Int. J. Cancer 86, 644–651 ( 2000).
Qian, X. et al. Kinase-deficient neu proteins suppress epidermal growth factor receptor function and abolish cell transformation. Oncogene 9, 1507–1514 (2000).
Kalmes, A., Vesti, B., Daum, G., Abraham, J. A. & Clowes, A. W. Heparin blockade of thrombin-induced smooth muscle cell migration involves inhibition of epidermal grwoth factor (EGF) receptor transactivation by heparin-binding EGF-like growth factor. Circulation Res. 87, 92–98 ( 2000).
Ben-Bassat, H. & Klein, B. Y. Inhibitors of tyrosine kinases in the treatment of psoriasis. Curr. Pharm. Des. 6, 933–942 ( 2000).
Nakata, A. et al. Localization of heparin-binding epidermal growth factor-like growth factor in human coronary arteries. Possible roles of HB-EGF in the formation of coronary atherosclerosis. Circulation 94, 2778–2786 (1996).
Wells, A. EGF receptor. Int. J. Biochem. Cell Biol. 31, 637–643 (1999).
Patel, N. V. et al. Neuregulin-1 and human epidermal growth factor receptors 2 and 3 play a role in human lung development in vitro. Am. J. Respir. Cell Mol. Biol. 22, 432– 440 (2000).
Levi, A. D. et al. The influence of heregulins on human Schwann cell proliferation . J. Neurosci. 15, 1329– 1340 (1995).
Levi, A. D. et al. The role of cultured Schwann cell grafts in the repair of gaps within the peripheral nervous system of primates. Exp. Neurol. 143, 25–36 ( 1997).
Cohen, S. & Carpenter, G. Human epidermal growth factor: isolation and chemical and biological properties. Proc. Natl Acad. Sci. USA 72, 1317–1321 ( 1975).
Bray, D. & Lay, S. Computer simulated evolution of a network of cell-signalling molecules. Biophys. J. 66, 972–977 (1994).
Aroian, R. V. & Sternberg, P. W. Multiple functions of let-23, a Caenorhabditis elegans receptor tyrosine kinase gene required for vulval induction. Genetics 128, 251– 267 (1991).
Volk, T. Signalling out Drosophila tendon cells. Trends Genet. 15, 448–453 (1999).
Schweitzer, R., Shaharabany, M., Seger, R. & Shilo, B.-Z. Secreted Spitz triggers the DER signalling pathway and is a limiting component in embryonic ventral ectoderm determination. Genes Dev. 10, 1518–1529 (1995).
Schweitzer, R. et al. Inhibition of Drosophila EGF receptor activation by the secreted protein Argos. Nature 376, 699–702 (1995).
Baulida, J., Kraus, M. H., Alimandi, M., Di Fiore, P. P. & Carpenter, G. All ErbB receptors other than the epidermal growth factor receptor are endocytosis impaired. J. Biol. Chem. 271, 5251–5257 (1996).Comparative analysis of ErbB proteins that shows that there are substantial differences in mechanisms of endocytosis-based attenuation.
Waterman, H., Sabanai, I., Geiger, B. & Yarden, Y. Alternative intracellular routing of ErbB receptors may determine signalling potency. J. Biol. Chem. 273, 13819–13827 (1998).
Worthylake, R. & Wiley, H. S. Structural aspects of the epidermal growth factor receptor required for transmodulation of ErbB2/neu . J. Biol. Chem. 272, 8594– 8601 (1997).
Lenferink, A. E. et al. Differential endocytic routing of homo- and hetero-dimeric ErbB tyrosine kinases confers signalling superiority to receptor heterodimers . EMBO J. 17, 3385–3397 (1998).
Sorkin, A., Di Fiore, P. P. & Carpenter, G. The carboxyl terminus of epidermal growth factor receptor/ErbB2 chimera is internalization impaired. Oncogene 8, 3021–3028 (1993).
Muthuswamy, S. K., Gilman, M. & Brugge, J. Controlled dimerization of ErbB receptors provide evidence for differential signalling by homo- and heterodimers. Mol. Cell. Biol. 19, 6845–6857 ( 1999).
Levkowitz, G. et al. c-Cbl/Sli-1 regulates endocytic sorting and ubiquitination of the epidermal growth factor receptor. Genes Dev. 12, 3663–3674 (1998). First report of the endocytic-sorting function of c-Cbl, a ubiquitin ligase that is a major substrate of ErbB1. Unlike sorting by c-Cbl to the late endosome, the oncogenic viral form, v-Cbl, shunts internalized receptors to the recycling pathway.
Menzo, S. et al. Transactivation of epidermal growth factor receptor gene by the hepatitis B virus X-gene product. Virology 196, 878–882 (1993).
Miller, W. E., Earp, H. S. & Raab-Traub, N. The Epstein-Barr virus latent membrane protein 1 induces expression of the epidermal growth factor receptor. J. Virol. 69, 4390–4398 (1995).
Opgenorth, A., Nation, N., Graham, K. & McFadden, G. Transforming growth factor-α, Shope fibroma virus factor, and vaccinia growth factor can replace myxoma growth factor in the induction of myxomatosis in rabbits . Virology 192, 701–709 (1993).
Adelsman, M. A., Huntley, B. K. & Maihle, N. J. Ligand-independent dimerization of oncogenic v-erbB products involves covalent interactions. J. Virol. 70, 2533–2544 (1996).
Straight, S. W., Herman, B. & McCance, D. J. The E5 oncoprotein of human papillomavirus type 16 inhibits the acidification of endosomes in human keratinocytes. J. Virol. 69, 3185–3192 (1995).
Yamanaka, Y. et al. Co-expression of epidermal growth factor receptor and ligands in human pancreatic cancer is associated with enhanced tumour aggressiveness . Anticancer Res. 13, 565– 569 (1993).
Krane, I. M. & Leder, P. NDF/heregulin induces persistence of terminal end buds and adenocarcinomas in the mammary glands of transgenic mice. Oncogene 12, 1781– 1788 (1996).
Gorgoulis, V. et al. Expression of EGF, TGF-α and EGFR in squamous cell lung carcinomas. Anticancer Res. 12, 1183– 1187 (1992).
Irish, J. C. & Bernstein, A. Oncogenes in head and neck cancer . Laryngoscope 103, 42– 52 (1993).
Shintani, S., Funayama, T., Yoshihama, Y., Alcalde, R. E. & Matsumura, T. Prognostic significance of ERBB3 overexpression in oral squamous cell carcinoma. Cancer Lett. 95, 79–83 (1995).
Acknowledgements
Y.Y. acknowledges support by the Israel Science Fund, the US Army Medical Research and Material Command and the M.D. Institute for Cancer Research.
Author information
Authors and Affiliations
Related links
Related links
DATABASE LINKS
FURTHER INFORMATION
ENCYCLOPEDIA OF LIFE SCIENCES
Glossary
- MESENCHYME
-
Immature connective tissue that consists of cells embedded in extracellular matrix.
- NEUREGULINS
-
EGF-like ligands whose primary receptor is ErbB3 and/or ErbB4. Four types of neuregulin are known.
- STROMA
-
Supporting connective tissue in which a glandular or other epithelium is embedded.
- HUMANIZED MONOCLONAL ANTIBODY
-
An antibody, usually from a rodent, engineered to contain mainly human sequences. This process reduces the immune response to the antibody in humans.
- ADAPTOR PROTEINS
-
Proteins that augment cellular responses by recruiting other proteins to a complex. They usually contain several protein–protein interaction domains.
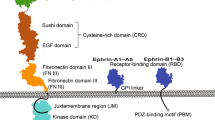
- EGF-LIKE DOMAIN
-
A motif with ∼50 amino acids, including six cysteine residues and a mainly β-sheet structure, found in all ErbB-binding growth factors and in extracellular matrix proteins.
- IMMUNOGLOBULIN-LIKE DOMAIN
-
A protein domain composed of two β-pleated sheets held together by a disulphide bond.
- METALLOPROTEINASES
-
Proteinases that have a metal ion at their active sites.
- MESODERM
-
The middle germ layer of the developing embryo. It gives rise to the musculoskeletal, vascular and urinogenital systems, and to connective tissue (including that of the dermis).
- ECTODERM
-
The outermost germ layer of the developing embryo. It gives rise to the epidermis and the nerves.
- AKT PATHWAY
-
Akt (or protein kinase B) is a serine/threonine protein kinase activated by the phosphatidylinositol-3-OH kinase pathway that activates survival responses.
- STRESS-ACTIVATED PROTEIN KINASES
-
Members of the mitogen-activated protein kinase (MAPK) family that respond to stress. They include the Jun amino-terminal kinases (JNKs) and the p38 MAPKs.
- UBIQUITIN LIGASES
-
Enzymes that catalyse the last stage of ubiquitylation, in which the small protein ubiquitin is transferred from a ubiquitin-conjugating enzyme (UBC or E2) to its target protein. They are also known as E3 enzymes.
- GAPS
-
Proteins that inactivate small GTP-binding proteins, such as Ras family members, by increasing their rate of GTP hydrolysis.
- GLIA
-
Supporting cells of the nervous system, including oligodendrocytes and astrocytes in the central nervous system, and Schwann cells in the peripheral nervous system. Glia surround neurons, providing mechanical and physical support, and electrical insulation between neurons.
- SCHWANN CELLS
-
Cells that produce myelin and ensheath axons in the peripheral nervous system.
- TRABECULAE
-
Finger-like projections of cardiac muscle cells that form ridges in the ventricular wall.
- SYMPATHETIC GANGLIA
-
Clusters of sympathetic neurons in which a glandular or other epithelium is embedded.
- NEURAL CREST
-
A group of embryonic cells that separate from the embryonic neural plate and migrate, giving rise to the spinal and autonomic ganglia, peripheral glia, chromaffin cells, melanocytes and some haematopoietic cells.
- CLONAL EXPANSION
-
Growth of a population of cells from a single precursor cell.
- CARCINOMA
-
A malignant tumour of epithelial origin.
- PROGNOSIS
-
The likely outcome or course of a disease.
- ANDROGEN-DEPENDENT PROSTATE CANCER
-
An early form of prostate cancer that is responsive to androgens and anti-androgen therapy.
- AUTOCRINE
-
Activation of cellular receptors by ligands produced by the same cell.
- GENE AMPLIFICATION
-
A differential increase in a specific portion of the genome. Amplification is associated with neoplastic transformation and acquisition of drug resistance.
- DUCTAL BREAST CANCER
-
Cancer arising from the lining of the milk ducts, as opposed to the lobules of the breast (lobular breast cancer).
- ANEUPLOIDY
-
An abnormal number of chromosomes caused by their inaccurate segregation during cell division.
- FLUORESCENCE IN SITU HYBRIDIZATION
-
Visualizing a genetic marker on a chromosome by using a fluorescently labelled polynucleotide probe that hybridizes to a gene on a chromosome during metaphase.
- FARNESYLTRANSFERASE INHIBITORS
-
Inhibitors that block the activity of Ras by preventing the addition of a farnesyl group that targets it to the plasma membrane.
- TYRPHOSTINS
-
A type of tyrosine kinase inhibitor.
Rights and permissions
About this article
Cite this article
Yarden, Y., Sliwkowski, M. Untangling the ErbB signalling network. Nat Rev Mol Cell Biol 2, 127–137 (2001). https://doi.org/10.1038/35052073
Issue Date:
DOI: https://doi.org/10.1038/35052073
This article is cited by
-
Gentiopicroside inhibits the progression of gastric cancer through modulating EGFR/PI3K/AKT signaling pathway
European Journal of Medical Research (2024)
-
Non-coding RNAs as potential therapeutic targets for receptor tyrosine kinase signaling in solid tumors: current status and future directions
Cancer Cell International (2024)
-
Based on whole-exome sequencing to explore the rule of Herceptin and TKI resistance in breast cancer patients
BMC Medical Genomics (2024)
-
Fluorescent in situ hybridization has limitations in screening NRG1 gene rearrangements
Diagnostic Pathology (2024)
-
Development of an invasion score based on metastasis-related pathway activity profiles for identifying invasive molecular subtypes of lung adenocarcinoma
Scientific Reports (2024)