Abstract
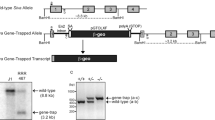
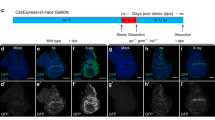
Programmed cell death is a fundamental requirement for embryogenesis, organ metamorphosis and tissue homeostasis. In mammals, release of mitochondrial cytochrome c leads to the cytosolic assembly of the apoptosome—a caspase activation complex involving Apaf1 and caspase-9 that induces hallmarks of apoptosis. There are, however, mitochondrially regulated cell death pathways that are independent of Apaf1/caspase-9. We have previously cloned a molecule associated with programmed cell death called apoptosis-inducing factor (AIF). Like cytochrome c, AIF is localized to mitochondria and released in response to death stimuli. Here we show that genetic inactivation of AIF renders embryonic stem cells resistant to cell death after serum deprivation. Moreover, AIF is essential for programmed cell death during cavitation of embryoid bodies—the very first wave of cell death indispensable for mouse morphogenesis. AIF-dependent cell death displays structural features of apoptosis, and can be genetically uncoupled from Apaf1 and caspase-9 expression. Our data provide genetic evidence for a caspase-independent pathway of programmed cell death that controls early morphogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Jacobson, M. D., Weil, M. & Raff, M. C. Programmed cell death in animal development. Cell 88, 347–354 (1997).
Vaux, D. L. & Korsmeyer, S. J. Cell death in development. Cell 96, 245–254 (1999).
Green, D. R. & Reed, J. C. Mitochondria and apoptosis. Science 281, 1309–1312 (1998).
Kerr, J. F., Wyllie, A. H. & Currie, A. R. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br. J. Cancer 26, 239–257 (1972).
Ellis, H. M. & Horvitz, H. R. Genetic control of programmed cell death in the nematode C. elegans. Cell 44, 817–829 (1986).
Hengartner, M. O. & Horvitz, H. R. C. elegans cell survival gene ced-9 encodes a functional homolog of the mammalian proto-oncogene bcl-2. Cell 76, 665–676 (1994).
Zou, H., Henzel, W. J., Liu, X., Lutschg, A. & Wang, X. Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell 90, 405–413 (1997).
Yuan, J., Shaham, S., Ledoux, S., Ellis, H. M. & Horvitz, H. R. The C. elegans cell death gene ced-3 encodes a protein similar to mammalian interleukin-1 beta-converting enzyme. Cell 75, 641–652 (1993).
Newmeyer, D. D., Farschon, D. M. & Reed, J. C. Cell-free apoptosis in Xenopus egg extracts: inhibition by Bcl-2 and requirement for an organelle fraction enriched in mitochondria. Cell 79, 353–364 (1994).
Kroemer, G. & Reed, J. C. Mitochondrial control of cell death. Nature Med. 6, 513–519 (2000).
Kluck, R. M., Bossy-Wetzel, E., Green, D. R. & Newmeyer, D. D. The release of cytochrome c from mitochondria: a primary site for Bcl-2 regulation of apoptosis. Science 275, 1132–1136 (1997).
Yang, J. et al. Prevention of apoptosis by Bcl-2: release of cytochrome c from mitochondria blocked. Science 275, 1129–1132 (1997).
Liu, X., Kim, C. N., Yang, J., Jemmerson, R. & Wang, X. Induction of apoptotic program in cell-free extracts: requirement for dATP and cytochrome c. Cell 86, 147–157 (1996).
Li, P. et al. Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91, 479–489 (1997).
Zou, H., Li, Y., Liu, X. & Wang, X. An APAF-1.cytochrome c multimeric complex is a functional apoptosome that activates procaspase-9. J. Biol. Chem. 274, 11549–11556 (1999).
Thompson, C. B. Apoptosis in the pathogenesis and treatment of disease. Science 267, 1456–1462 (1995).
Yoshida, H. et al. Apaf1 is required for mitochondrial pathways of apoptosis and brain development. Cell 94, 739–750 (1998).
Cecconi, F., Alvarez-Bolado, G., Meyer, B. I., Roth, K. A. & Gruss, P. Apaf1 (CED-4 homolog) regulates programmed cell death in mammalian development. Cell 94, 727–737 (1998).
Hakem, R. et al. Differential requirement for caspase 9 in apoptotic pathways in vivo. Cell 94, 339–352 (1998).
Kuida, K. et al. Reduced apoptosis and cytochrome c-mediated caspase activation in mice lacking caspase 9. Cell 94, 325–337 (1998).
Li, K. et al. Cytochrome c deficiency causes embryonic lethality and attenuates stress-induced apoptosis. Cell 101, 389–399 (2000).
Amarante-Mendes, G. P. et al. Anti-apoptotic oncogenes prevent caspase-dependent and independent commitment for cell death. Cell Death Differ. 5, 298–306 (1998).
Haraguchi, M. et al. Apoptotic protease activating factor 1 (Apaf-1)-independent cell death suppression by Bcl-2. J. Exp. Med. 191, 1709–1720 (2000).
Susin, S. A. et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 397, 441–446 (1999).
Daugas, E. et al. Mitochondrio-nuclear translocation of AIF in apoptosis and necrosis. FASEB J. 14, 729–739 (2000).
Ferri, K. F. et al. Apoptosis control in syncytia induced by the HIV type 1-envelope glycoprotein complex. Role of mitochondria and caspases. J. Exp. Med. 192, 1081–1092 (2000).
Chen, J., Lansford, R., Stewart, V., Young, F. & Alt, F. W. RAG-2-deficient blastocyst complementation: an assay of gene function in lymphocyte development. Proc. Natl Acad. Sci. USA 90, 4528–4532 (1993).
Martin, G. R. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc. Natl Acad. Sci. USA 78, 7634–7638 (1981).
Bautch, V. L. et al. Blood island formation in attached cultures of murine embryonic stem cells. Dev. Dyn. 205, 1–12 (1996).
Coucouvanis, E. & Martin, G. R. Signals for death and survival: a two-step mechanism for cavitation in the vertebrate embryo. Cell 83, 279–287 (1995).
Robertson, E. J. Embryo-Derived Stem Cell Lines (IRL, Oxford, 1987).
Coucouvanis, E. & Martin, G. R. BMP signaling plays a role in visceral endoderm differentiation and cavitation in the early mouse embryo. Development 126, 535–546 (1999).
Leahy, A., Xiong, J. W., Kuhnert, F. & Stuhlmann, H. Use of developmental marker genes to define temporal and spatial patterns of differentiation during embryoid body formation. J. Exp. Zool. 284, 67–81 (1999).
McCarthy, N. J., Whyte, M. K., Gilbert, C. S. & Evan, G. I. Inhibition of Ced-3/ICE-related proteases does not prevent cell death induced by oncogenes, DNA damage, or the Bcl-2 homologue Bak. J. Cell Biol. 136, 215–227 (1997).
Lorenzo, H. K., Susin, S. A., Penninger, J. & Kroemer, G. Apoptosis inducing factor (AIF): a phylogenetically old, caspase-independent effector of cell death. Cell Death Differ. 6, 516–524 (1999).
Schmitt, R. M., Bruyns, E. & Snodgrass, H. R. Hematopoietic development of embryonic stem cells in vitro: cytokine and receptor gene expression. Genes Dev. 5, 728–740 (1991).
Shalaby, F. et al. Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature 376, 62–66 (1995).
Cho, S. K. et al. Functional characterization of B lymphocytes generated in vitro from embryonic stem cells. Proc. Natl Acad. Sci. USA 96, 9797–9802 (1999).
Acknowledgements
We thank M. Saunders for scientific editing; A. Oliveira-dos-Santos, K. Bachmaier, T. Wada, V. Stambolic, L. Zhang, M. Crackower, C. Krawzcyk, I. Kozieradzki, Q. Liu, J. Irie-Sasaki, M. Nghiem, R. Sarao, E. Griffith, L. Barra and A. Manoukian for comments; D. Métivier and B. Calvieri for technical assistance; and J. Rossant and A. Bernstein for lacZ-expressing ES cells. N.J. and J.M.P. are supported by the Canadian Institute of Health Research (CIHR), Amgen, and the National Cancer Institute of Canada. W.L.S. is supported by the Karyn Glick Memorial Special Fellowship and CIHR. E.D. is supported by Assistance Publique-Hôpitaux de Paris and CANAM. G.K. is supported by grants from Ligue Nationale Contre le Cancer, European Commission and Agence Nationale pour la Recherche Sur le SIDA. J.M.P. holds a Canadian Research Chair in Cell Biology.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Rights and permissions
About this article
Cite this article
Joza, N., Susin, S., Daugas, E. et al. Essential role of the mitochondrial apoptosis-inducing factor in programmed cell death. Nature 410, 549–554 (2001). https://doi.org/10.1038/35069004
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35069004
This article is cited by
-
Quantitative Proteomics Reveals Manganese Alleviates Heat Stress of Broiler Myocardial Cells via Regulating Nucleic Acid Metabolism
Biological Trace Element Research (2024)
-
p20BAP31 induces cell apoptosis via both AIF caspase-independent and the ROS/JNK mitochondrial pathway in colorectal cancer
Cellular & Molecular Biology Letters (2023)
-
Mitochondrial Dysfunction in Cardiotoxicity Induced by BCR-ABL1 Tyrosine Kinase Inhibitors -Underlying Mechanisms, Detection, Potential Therapies
Cardiovascular Toxicology (2023)
-
An insight into the associations between microRNA expression and mitochondrial functions in cancer cell and cancer stem cell
Molecular Biology Reports (2023)
-
Apoptosis in post-streptococcal glomerulonephritis and mechanisms for failed of inflammation resolution
Pediatric Nephrology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.