Abstract
Antlions are larvae of the Myrmeleontidae family that live on other insects1 by sucking out the body fluid from their prey, after first paralysing it with a toxin produced by salivary bacteria. Here we show that the paralysing toxin produced by bacterial endosymbionts in the saliva of Myrmeleon bore larvae is a homologue of GroEL, a protective heat-shock protein known as a molecular chaperone. The amino-acid residues critical for this protein's toxicity are located away from the regions essential to its protein-folding activity, indicating that the dual function of this GroEL homologue may benefit both the antlion and the endosymbiont.
Similar content being viewed by others
Main
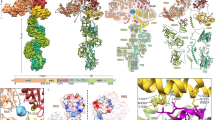
The saliva of the larvae of M. bore (Fig. 1a), a pit-building antlion, contains the endosymbiont Enterobacter aerogenes (Fig. 1b), which, when grown up in culture and injected into German cockroaches (Blattella germanica), rapidly paralyses them. We purified one of the insecticidal proteins from culture broth and found that it migrated on a denaturing SDS–polyacrylamide gel as a single band at a position corresponding to a relative molecular mass of about 63K (Fig. 1c). Partial amino-acid sequencing of this toxin indicated that it was a GroEL homologue.
a, Larva of M. bore. Scale bar, 5 mm. b, Scanning electron micrograph of the bacterial endosymbiont E. aerogenes. Scale bar, 1 μm. c, Electrophoresis on a 10 % SDS–polyacrylamide gel of the toxic protein purified from a culture of E. aerogenes. d, The three-dimensional structure of a subunit in a 14-mer GroEL molecule from E. coli (created using a PDB file,1DER). The apical, intermediate and equatorial domains are coloured yellow, blue and green, respectively. Locations of the residues in which mutation confers toxicity are shown in red. Sites at which mutation blocks the binding of polypeptide and GroES in the apical domain8 and the residues involved in ATP binding in the equatorial domain6,7 are shown in purple and orange, respectively.
GroEL, also known as chaperonin, is a product of the groE gene of Escherichia coli and was first discovered as a mutant that inhibits bacteriophage growth2. GroEL forms a homo-oligomer with a double-toroid structure and, in combination with the protein GroES and ATP, acts as a molecular chaperone to ensure correct folding and assembly of proteins2,3,4. However, the paralytic and insecticidal activities described here cannot be explained in terms of a chaperonin function.
The GroEL homologue from E. aerogenes showed no acute toxicity towards mice, but it rapidly paralysed and killed cockroaches when injected at a minimum dose of 2.7 ± 1.6 ng (mean ± s.e.m.; n = 3); the recombinant protein encoded by its complementary DNA and expressed in E. coli was equally toxic. By contrast, GroEL from E. coli did not paralyse the insects even at doses as high as 2 μg.
We cloned the groE gene from E. aerogenes and found that only 11 residues in the GroEL homologue had alignments different from the residues in GroEL from E. coli (Table 1) — except at the carboxy terminus, where the GroEL homologue has a methionine residue and is shorter than GroEL by three residues. The amino-acid residues Val 100 (valine at the 100th residue), Asn 101, Asp 338 and Ala 471 are crucial for toxicity, as shown by the marked reduction in toxicity of mutants carrying the substitutions Ile 100, Thr 101, Glu 338 and Gly 471 (Table 1). The importance of these residues was confirmed by reversing the individual mutations (substituting valine for isoleucine at residue 100, and so on) in E. coli GroEL, which conferred toxicity on the protein (Table 1).
In the crystal structure of E. coli GroEL5,6,7, Glu 338 is located in the apical domain, far from the polypeptide- and GroES-binding sites7,8, whereas Ile 100, Thr 101 and Gly 471 are in the equatorial domain, far from the ATP-binding pocket that faces the central cavity (Fig. 1d)6,7. Thus, neither GroES binding nor ATP binding to the GroEL homologue is effective in generating toxicity. Injection of the GroES homologue from E. aerogenes together with ATP and the GroEL homologue scarcely influenced the minimum paralysing dose (3.9 ± 0.3 ng; n = 3).
Chaperonins from bacterial pathogens can function as cell-signalling molecules, stimulating human monocytes, leukocytes, fibroblasts and epithelial cells to release pro-inflammatory cytokines9. The toxicity of the GroEL homologue towards insects can be seen as an effect of a bacterial extracellular chaperonin on eukaryotic cells. The homologue may act on particular receptors in insects to induce paralysis, having evolved this non-chaperone function to establish a mutually beneficial antlion–symbiont relationship. Our finding that insecticidal proteins are produced by endosymbionts to help in capturing prey is likely to extend to many other fluid-feeding carnivorous insects.
References
Matsura, T. Ecol. Res. 1, 15–24 (1986).
Zeilstra-Ryalls, J., Fayet, O. & Georgopoulos, C. Annu. Rev. Microbiol. 45, 301–325 (1991).
Sigler, P. B. et al. Annu. Rev. Biochem. 67, 581–608 (1998).
Ranson, N. A., White, H. E. & Saibil, H. R. Biochem. J. 333, 233–242 (1998).
Braig, K. et al. Nature 371, 578–586 (1994).
Boisvert, D. C., Wang, J., Otwinowski, Z., Horwich, A. L. & Sigler, P. B. Nature Struct. Biol. 3, 170–177 (1996).
Xu, Z., Horwich, A. L. & Sigler, P. B. Nature 388, 741–750 (1997).
Fenton, W. A., Kashi, Y., Furtak, K. & Horwich, A. L. Nature 371, 614–619 (1994).
Ranford, J. C., Coates, A. R. M. & Henderson, B. Exp. Rev. Mol. Med. http://www-ermm.cbcu.cam.ac.uk/00002015h.htm (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yoshida, N., Oeda, K., Watanabe, E. et al. Chaperonin turned insect toxin. Nature 411, 44 (2001). https://doi.org/10.1038/35075148
Issue Date:
DOI: https://doi.org/10.1038/35075148
This article is cited by
-
Identification of a cytotoxic factor from a non-pigmented entomopathogenic Serratia marcescens isolate toxic towards human carcinoma cell lines
Archives of Microbiology (2023)
-
Cuticular Sensilla on the Larval Mouthparts of Antlion Myrmeleon sp. and Its Role in Predatory Behavior
Proceedings of the National Academy of Sciences, India Section B: Biological Sciences (2022)
-
Blood feeding tsetse flies as hosts and vectors of mammals-pre-adapted African Trypanosoma: current and expected research directions
BMC Microbiology (2018)
-
Deep divergence and rapid evolutionary rates in gut-associated Acetobacteraceae of ants
BMC Microbiology (2016)
-
A novel insecticidal GroEL protein from Xenorhabdus nematophila confers insect resistance in tobacco
Transgenic Research (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.