Abstract
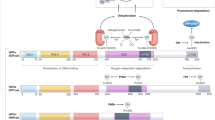
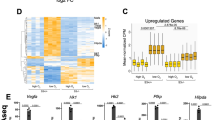
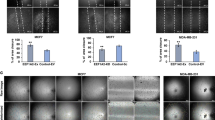
Alteration of gene expression is a crucial component of adaptive responses to hypoxia. These responses are mediated by hypoxia-inducible transcription factors (HIFs)1,2. Here we describe an inhibitory PAS (Per/Arnt/Sim) domain protein, IPAS, which is a basic helix-loop-helix (bHLH)/PAS protein structurally related to HIFs. IPAS contains no endogenous transactivation function but demonstrates dominant negative regulation of HIF-mediated control of gene expression. Ectopic expression of IPAS in hepatoma cells selectively impairs induction of genes involved in adaptation to a hypoxic environment, notably the vascular endothelial growth factor (VEGF) gene, and results in retarded tumour growth and tumour vascular density in vivo. In mice, IPAS was predominantly expressed in Purkinje cells of the cerebellum and in corneal epithelium of the eye. Expression of IPAS in the cornea correlates with low levels of expression of the VEGF gene under hypoxic conditions. Application of an IPAS antisense oligonucleotide to the mouse cornea induced angiogenesis under normal oxygen conditions, and demonstrated hypoxia-dependent induction of VEGF gene expression in hypoxic corneal cells. These results indicate a previously unknown mechanism for negative regulation of angiogenesis and maintenance of an avascular phenotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bunn, H. F. & Poyton, R. O. Oxygen sensing and molecular adaptation to hypoxia. Physiol. Rev. 76, 839–885 (1996).
Wenger, R. H. Mammalian oxygen sensing, signalling and gene regulation. J. Exp. Biol. 203, 1253–1263 (2000).
Krogh, A., Brown, M., Mian, I. S., Sjolander, K. & Haussler, D. Hidden Markov models in computational biology. Applications to protein modeling. J. Mol. Biol. 235, 1501–1531 (1994).
Wang, G. L., Jiang, B. H., Rue, E. A. & Semenza, G. L. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc. Natl Acad. Sci. USA 92, 5510–5514 (1995).
Ema, M. et al. A novel bHLH-PAS factor with close sequence similarity to hypoxia-inducible factor 1α regulates the VEGF expression and is potentially involved in lung and vascular development. Proc. Natl Acad. Sci. USA 94, 4273–4278 (1997).
Tian, H., McKnight, S. L. & Russell, D. W. Endothelial PAS domain protein 1 (EPAS1), a transcription factor selectively expressed in endothelial cells. Genes Dev. 11, 72–82 (1997).
Semenza, G. L. HIF-1 and human disease: one highly involved factor. Genes Dev. 14, 1983–1991 (2000).
Kallio, P. J., Wilson, W. J., O'Brien, S., Makino, Y. & Poellinger, L. Regulation of the hypoxia-inducible transcription factor 1α by the ubiquitin-proteasome pathway. J. Biol. Chem. 274, 6519–6525 (1999).
Maxwell, P. H. et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 399, 271–275 (1999).
Wiesener, M. S. et al. Induction of endothelial PAS domain protein-1 by hypoxia: characterization and comparison with hypoxia-inducible factor-1α. Blood 92, 2260–2268 (1998).
Li, H., Ko, H. P. & Whitlock, J. P. Induction of phosphoglycerate kinase 1 gene expression by hypoxia. Roles of Arnt and HIF1α. J. Biol. Chem. 271, 21262–21267 (1996).
Forsythe, J. A. et al. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol. Cell Biol. 16, 4604–4613 (1996).
Poellinger, L. in Inducible Gene Expression (ed. Baeuerle, P. A.) 177–205 (Birkhäuser, Boston, 1995).
Thakur, A., Willcox, M. D. & Stapleton, F. The proinflammatory cytokines and arachidonic acid metabolites in human overnight tears: homeostatic mechanisms. J. Clin. Immunol. 18, 61–70 (1998).
Sack, R. A., Beaton, A. R. & Sathe, S. Diurnal variations in angiostatin in human tear fluid: a possible role in prevention of corneal neovascularization. Curr. Eye Res. 18, 186–193 (1999).
Gradin, K. et al. Functional interference between hypoxia and dioxin signal transduction pathways: competition for recruitment of the Arnt transcription factor. Mol. Cell. Biol. 16, 5221–5231 (1996).
Ravi, R. et al. Regulation of tumor angiogenesis by p53-induced degradation of hypoxia-inducible factor 1α. Genes Dev. 14, 34–44 (2000).
Zundel, W. et al. Loss of PTEN facilitates HIF-1-mediated gene expression. Genes Dev. 14, 391–396 (2000).
Li, J. et al. PR 39, a peptide regulator of angiogenesis. Nature Med. 6, 49–55 (2000).
Norton, J. D., Deed, R. W., Craggs, G. & Sablitzky, F. Id helix-loop-helix protein in cell growth and differentiation. Trends Cell Biol. 8, 58–65 (1998).
Eddy, S. R. Multiple alignment using hidden Markov models. Ismb 3, 114–120 (1995).
Tanimoto, K., Makino, Y., Pereira, T. & Poellinger, L. Mechanism of regulation of the hypoxia-inducible factor-1α by the von Hippel-Lindau tumor suppressor protein. EMBO J. 19, 4298–4309 (2000).
Bertilsson, G. et al. Identification of a human nuclear receptor defines a new signaling pathway for CYP3A induction. Proc. Natl Acad. Sci. USA 95, 12208–12213 (1998).
Cao, R. et al. Suppression of aniogenesis by a novel inhibitor, K1-5, generated by plasmin-mediated proteolysis. Proc. Natl Acad. Sci. USA 96, 5728–5733 (1999).
Acknowledgements
We thank Y. Fujii-Kuriyama and I. Pongratz for reagents. This work was supported by grants from the Swedish Medical Research Council, Pharmacia Corporation, Swedish Cancer Society, the Human Frontiers Science Program, the Uehara Memorial Foundation, and the Japanese Ministry of Education, Culture, Sports, Science and Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Makino, Y., Cao, R., Svensson, K. et al. Inhibitory PAS domain protein is a negative regulator of hypoxia-inducible gene expression. Nature 414, 550–554 (2001). https://doi.org/10.1038/35107085
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35107085
This article is cited by
-
Targeting hypoxia-inducible factors: therapeutic opportunities and challenges
Nature Reviews Drug Discovery (2024)
-
The role of HIF in angiogenesis, lymphangiogenesis, and tumor microenvironment in urological cancers
Molecular Biology Reports (2024)
-
Targeting angiogenesis in oncology, ophthalmology and beyond
Nature Reviews Drug Discovery (2023)
-
Effects of acute hypoxia on nutrient metabolism and physiological function in turbot, Scophthalmus maximus
Fish Physiology and Biochemistry (2023)
-
COVID-19 instigates adipose browning and atrophy through VEGF in small mammals
Nature Metabolism (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.