Abstract
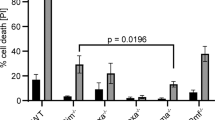
WILD-TYPE p53 protein has many properties consistent with its being the product of a tumour suppressor gene1–3. Although the normal roles of tumour suppressor genes are still largely unknown, it seems that they could be involved in promoting cell differentiation4–6 as well as in mediating growth arrest by growth-inhibitory cytokines7–9. Hence, the abrogation of wild-type p53 expression, which is a common feature of many tumours, could eliminate these activities. We have now tested this notion by restoring the expression of p53 in a murine myeloid leukaemic cell line that normally lacks p53. The use of a temperature-sensitive p53 mutant10 allowed us to analyse cells in which the introduced p53 had either wild-type or mutant properties. Although there seemed to be no effect on differentiation, the introduction of wild-type p53 resulted in rapid loss of cell viability in a way characteristic of apoptosis (programmed cell death). The effect of wild-type p53 was counteracted by interleukin-6. Thus products of tumour suppressor genes could be involved in restricting precursor cell populations by mediating apoptosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Levine, A. J. & Momand, J. Biochim. biophys. Acta 1032, 119–136 (1990).
Lane, D. P. & Benchimol, S. Genes Dev. 4, 1–8 (1990).
Michalovitz, D., Halevy, O. & Oren, M. J. cell. Biochem. 45, 22–29 (1991).
Stanbridge, E. J. A. Rev. Genet. 24, 615–657 (1990).
Sachs, L. in Genetic Analysis of Tumour Suppression (eds Bock, G. & Marsh, J.) 217–233 (Ciba Foundation Symposium Vol. 142, Wiley, Chichester, 1989).
Harris, H. J. in Genetic Analysis of Tumour Suppression (eds Bock, G. & Marsh, J.) 199–213 (Ciba Foundation Symposium, Vol. 142, Wiley, Chichester, 1989).
Laiho, M., DeCaprio, J. A., Ludlow, J. W., Livingston, D. M. & Massague, J. Cell 62, 175–185 (1990).
Pietenpol, J. A. et al. Cell 61, 777–785 (1990).
Deiss, L. & Kimchi, A. Science 252, 117–120 (1991).
Michalovitz, D., Halevy, O. & Oren, M. Cell 62, 671–680 (1990).
Resnitzky, D. & Kimchi, A. Cell Growth Diff. 2, 33–41 (1991).
Ichikawa, Y. J. Cell Physiol. 74, 223–234 (1969).
Fibach, E., Hayashi, M. & Sachs, L. Proc. natn. Acad Sci. U.S.A. 70, 343–346 (1973).
Resnitzky, D., Yarden, A., Zipori, D. & Kimchi, A. Cell 46, 31–40 (1986).
Southern, P. J. & Berg, P. J. molec. appl. Genet. 1, 327–341 (1982).
Milner, J. & Medcalf, E. A. J. molec. Biol. 216, 481–484 (1990).
Ginsberg, D., Michalovitz, D., Ginsberg, D. & Oren, M. Molec. cell. Biol. 11, 582–585 (1991).
Duvall, E. & Wyllie, A. H. Immun. Today 7, 115–119 (1986).
Williams, G. T., Smith, C. A., Spooncer, E., Dexter, T. M. & Taylor, D. R. Nature 343, 76–79 (1990).
Rodriguez-Tarducy, G., Collins, M. & Lopez-Rivas, A. EMB0 J. 9, 2997–3002 (1990).
Duke, R. C. & Cohen, J. J. Lymphokine Res. 5, 289–299 (1986).
Lotem, J., Cragoe, E. J. & Sachs, L. Blood (in the press).
Sachs, L. Cancer 65, 2196–2206 (1990).
Shabo, Y. et al. Blood 72, 2070–2073 (1988).
Wolf, D. & Rotter, V. Proc. natn. Acad. Sci. U.S.A. 82, 790–794 (1985).
Prokocimer, M. et al. Blood 68, 113–119 (1986).
Lubbert, M., Miller, C. W., Crawford, L. V. & Koeffler, H. P. J. exp. Med. 167, 873–886 (1988).
Fearon, E. R. & Vogelstein, B. Cell 61, 759–767 (1990).
Stanbridge, E. J. & Nowell, P. C. Cell 63, 867–874 (1990).
Halevy, O., Michalovitz, D. & Oren, M. Science 250, 113–116 (1990).
Eliyahu, D., Michalovitz, D., Eliyahu, S., Pinhasi-Kimhi, O. & Oren, M. Proc. natn. Acad. Sci. U.S.A. 86, 8763–8767 (1989).
Finlay, C. A., Hinds, P. W. & Levine, A. J. Cell 57, 1083–1093 (1989).
Baker, S. J., Markowltz, S., Fearon, E. R., Willson, J. K. V. & Vogelstein, B. Science 249, 912–915 (1990).
Mercer, W. E. et al. Proc. natn. Acad. Sci. U.S.A. 87, 6166–6170 (1990).
Chen, P.-L., Chen, Y., Bookstein, R. & Lee, W.-H. Science 250, 1576–1580 (1990).
Diller, L. et al. Molec. cell. Biol. 10, 5772–5781 (1990).
Nunez, G. et al. J. Immun. 144, 3602–3610 (1990).
Vaux, D. L., Cory, S. & Adams, J. M. Nature 335, 440–442 (1988).
Hockenbery, D., Nunez, G., Milliman, C., Schreiber, R. & Korsmeyer, S. J. Nature 348, 334–336 (1990).
Gregory, C. D. et al. Nature 349, 612–614 (1991).
Pinhasi, O. & Oren, M. Molec. cell. Biol. 4, 2180–2186 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yonish-Rouach, E., Resnftzky, D., Lotem, J. et al. Wild-type p53 induces apoptosis of myeloid leukaemic cells that is inhibited by interleukin-6. Nature 352, 345–347 (1991). https://doi.org/10.1038/352345a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/352345a0
This article is cited by
-
Changes in calpain-2 expression during glioblastoma progression predisposes tumor cells to temozolomide resistance by minimizing DNA damage and p53-dependent apoptosis
Cancer Cell International (2023)
-
Paracrine Factors Released from Tonsil-Derived Mesenchymal Stem Cells Inhibit Proliferation of Hematological Cancer Cells Under Hyperthermia in Co-culture Model
Applied Biochemistry and Biotechnology (2023)
-
Molekulare Klassifikation beim Endometriumkarzinom
Die Gynäkologie (2023)
-
Severe cellular stress drives apoptosis through a dual control mechanism independently of p53
Cell Death Discovery (2022)
-
Green tea EGCG effectively alleviates experimental colitis in middle-aged male mice by attenuating multiple aspects of oxi-inflammatory stress and cell cycle deregulation
Biogerontology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.