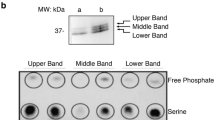
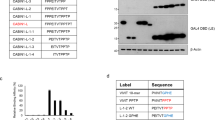
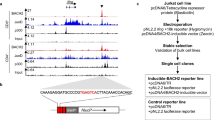
Abstract
IN the presence of costimulation1, Ca2 + influx in T cells leads to activation (transcription of interleukin-2; ref. 2) via calcineurin3,4. In the absence of costimulation, Ca2+ influx results in anergy (interleukin-2 transcriptional block5) through an unknown mechanism. Specific attenuation of interleukin-2 transcriptional induction occurs in Jurkat T cells following pretreatment with a Ca2+ ionophore. A >90% block of inducible interleukin-2 reporter gene activity was initiated by transfection of a constitutively active mutant of multifunctional Ca2 + /calmodulin-dependent protein kinase (CaM kinase or CaM kinase II)6, but not by constitutive mutants of CaM kinase IV, calcineurin or protein kinase C. The block was complete six hours after kinase transfection and showed specificity for interleukin-2; there was no change in β-actin transcription or in c-fos transcription induced by phorbol myristyl acetate, and a Rous sarcoma virus promoter was stimulated threefold. Multifunctional CaM kinase also attenuated interleukin-2 activation by calcineurin plus phorbol ester. T-cell receptor signalling activates multifunctional CaM kinase. These findings suggest that two Ca2 + /calmodulin-responsive enzymes, multifunctional CaM kinase and calcineurin, could mediate the divergent effects of Ca2 + signals in T-lymphocyte regulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Schwartz, R. H. Cell 71, 1065–1068 (1992).
Crabtree, G. R. Science 243, 355–360 (1989).
O'Keefe, S. J., Tamura, J., Kincaid, R. L., Tocci, M. J. & O'Neill, E. A. Nature 357, 692–694 (1992).
Clipstone, N. A. & Crabtree, G. R. Nature 357, 695–697 (1992).
Schwartz, R. H. Science 248, 1349–1356 (1990).
Schulman, H. Curr. Opin. Cell Biol. 5, 247–253 (1993).
MacNicol, M., Jefferson, A. B. & Schulman, H. J. biol. Chem. 265, 18055–18058 (1990).
Gardner, P. Cell 59, 15–20 (1989).
Meyer, T., Hanson, P. I., Stryer, L. & Schulman, H. Science 256, 1199–1202 (1992).
Schulman, H., Hanson, P. I., Meyer, T. Cell Calcium 13, 401–411 (1992).
Kang, S.-M. et al. Science 257, 1134–1138 (1992).
Durand, D. B. et al. Molec. cell. Biol. 8, 1715–1724 (1988).
Nghiem, P., Saati, S. M., Martens, C. L., Gardner, P. & Schulman, H. J. biol. Chem. 268, 5471–5479 (1993).
Li, X., Nghiem, P., Schulman, H. & Francke, U. Cytogenet. Cell Genet. 66, 113–116 (1994).
Traystman, M. D. & Beisel, K. W. Clin. exp. Immun. 86, 291–298 (1991).
Muramatsu, M.-A., Kaibuchi, K. & Arai, K.-I. Molec. cell. Biol. 9, 831–836 (1989).
Cruzalegui, F. H. & Means, A. R. J. biol. Chem. 268, 26171–26178 (1993).
Hanson, P. I. & Schulman, H. A. Rev. Biochem. 61, 559–601 (1992).
Hanissian, S. H., Frangakis, M., Bland, M. M., Jawahar, S. & Chatila, T. A. J. biol. Chem. 268, 20055–20063 (1993).
Kapiloff, M. S., Mathis, J. M., Nelson, C. A., Lin, C. R. & Rosenfeld, M. G. Proc. natn. Acad. Sci. U.S.A. 88, 3710–3714 (1991).
MaCaffrey, P. G., Perrino, B. A., Soderling, T. R. & Rao, A. J. biol. Chem. 268, 3747–3752 (1993).
McCaffrey, P. G. et al. Science 262, 750–754 (1993).
Lisman, J. Proc. natn. Acad. Sci. U.S.A. 86, 9574–9578 (1989).
Malinow, R., Schulman, H. & Tsien, R. W. Science 245, 862–866 (1989).
Silva, A. J., Stevens, C. F., Tonegawa, S. & Wang, Y. Science 257, 201–206 (1992).
Mulkey, R. M., Endo, S., Shenolikar, S. & Malenka, R. C. Nature 369, 486–488 (1994).
Grynkiewicz, G., Poenie, M. & Tsien, R. Y. J. biol. Chem. 260, 3440–3450 (1985).
Mattila, P. S. et al. EMBO J. 9, 4425–4433 (1990).
Jamal, S. & Ziff, E. Nature 344, 463–466 (1990).
Takebe, Y. et al. Molec. cell. Biol. 8, 466–472 (1988).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nghiem, P., Ollick, T., Gardner, P. et al. lnterleukin-2 transcriptional block by multifunctional Ca2+/ calmodulin kinase. Nature 371, 347–350 (1994). https://doi.org/10.1038/371347a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/371347a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.