Abstract
Immunohistochemical studies are increasingly used for the routine diagnosis of lymphomas as it is widely accepted that lymphomas of different cell lineages vary in their prognosis and response to therapy. A case of peripheral T-cell lymphoma with aberrant expression of B-cell–associated antigens L-26 (CD20) and mb-1 (CD 79a) is described. The disease pursued an aggressive clinical course, and the patient died of disease 6 weeks after presentation.
Immunohistochemical studies demonstrated expression of both T- and B-cell–associated antigens, including CD3, CD8, CD43, TIA-1, CD20, and CD79a. Other markers expressed by the tumor cells included CD56 and S-100. Of interest, βF-1 staining for the β chain of T-cell receptor (TCR) complex was positive in the small admixed T lymphocytes but was negative in the tumor cells, raising the possibility of a γ/δ T-cell lymphoma. Molecular studies by polymerase chain reaction (PCR) demonstrated clonal TCR-γ chain gene rearrangement without evidence for a clonal rearrangement of the immunoglobulin heavy chain gene. PCR for HHV-8 related sequences was negative.
Mb-1 is an IgM-associated protein that was thought to be restricted to normal and neoplastic B cells. Although its coexpression has been reported in up to 10% cases of precursor T-cell lymphoblastic lymphoma, the coexpression of both CD20 and CD79a has not been described in mature T-cell malignancies. Biphenotypic lymphomas associated with HHV-8 have been reported in immunodeficiency, but no evidence of immune deficiency was identified, and studies for EBV and HHV-8 were negative. This case illustrates that no marker has absolute lineage specificity and that immunophenotypic studies should always be performed with panels of monoclonal antibodies. Moreover, cases with ambiguous phenotypes may require genotypic studies for precise lineage assignment.
Similar content being viewed by others
INTRODUCTION
Current lymphoma classifications have incorporated immunophenotyping as an integral part of lymphoma diagnosis. Immunophenotyping is increasingly routinely used in the diagnosis and classification of non-Hodgkin's lymphomas (NHL). The recent Revised European-American Lymphoma (REAL) and World Health Organization (WHO) classifications use lineage as a starting point for classification. Lymphomas are broadly divided into T-cell and B-cell types based on immunophenotyping by immunohistochemistry or flow cytometry (1, 2). The new classifications identify distinct disease entities that have distinctive clinicopathologic features and response to therapy (3).
CD20 and CD3 are the most widely used markers for B- and T-cell lineage. However, in some cases a wide battery of immunophenotypic and genotypic tools must be employed for accurate classification. We report a novel case of T-cell lymphoma that expressed both CD20 and CD79a, in addition to the T-cell–associated antigens CD3 and CD8. CD79a has been considered highly specific for B-cell lineage because it has a major functional role in the immunoglobulin-antigen receptor complex. Although CD79a has been reported in infrequent examples of precursor T-cell and myeloid malignancies, the expression of both CD20 and CD79a has not previously been reported in mature, normal or in neoplastic T cells (4, 5, 6).
CASE REPORT
Clinical History
A case was submitted in consultation to the Hematopathology Section of the Laboratory of Pathology at the National Cancer Institute (NCI) from a local hospital. The patient was a 76-year-old white male who presented with a 3-month history of 10 to 12 pounds' weight loss and abdominal discomfort. He denied having fever, chills, or night sweats. His past medical history was remarkable for surgery for bleeding duodenal ulcer in 1986, and he was on Prevacid. He had quit smoking about 18 years ago and his alcohol intake was occasional. Physical exam results were unremarkable. A computed tomography exam of the abdomen revealed a left adrenal mass that appeared to be solid and measured 5.5 to 6 cm. The adrenal mass appeared to be rapidly enlarging, and a laparotomy was performed that resulted in left adrenalectomy and nephrectomy. The specimen was examined by the referring pathologist and was thought to be a high-grade lymphoma. The case was referred to the NCI for diagnosis and classification.
MATERIALS AND METHODS
Immunohistochemical studies were performed on formalin-fixed, paraffin-embedded tissue sections from the submitted tissue blocks of the specimen using the avidin-biotin-peroxidase complex method as described elsewhere (7). The antibodies used are summarized in Table 1. Antigen retrieval was performed using a Tender Cooker (Nordicware, Minneapolis, MN) with citrate buffer. Staining was performed on an automated immunostainer (Ventana Medical Systems, Inc., Tucson, AZ) according to the company's protocols. Known positive and negative tissues were used as controls.
The EBER RNA in situ hybridization for Epstein-Barr viral sequences was performed as described elsewhere in detail (8). Briefly, 5-μm sections of paraffin-embedded tissue were prepared on silanated slides. Tissue sections were deparaffinized, rehydrated, and placed into the Ventana Gen II automated in situ hybridization instrument (Ventana Medical Systems). The EBER-1 probe was used along with U6 control.
Polymerase chain reaction (PCR) studies were performed using paraffin sections. DNA was extracted from the paraffin sections and PCR amplified using primers to Framework Region 3 of the immunoglobulin heavy chain gene (VJ-PCR), according to the method of Segal et al. (9), The products were run on 16% polyacrylamide gels and stained with ethidium bromide. For T-cell receptor gene rearrangement, DNA was amplified using primers to the TCR-γ chain gene. Two separate reactions were performed, one with primers Vγ101, Vγ11 and Jγ12 (set 1) and a second with primers Vγ101, Vγ11 and Jp12, according to the method of McCarthy et al. (10) The DNA was also amplified to detect HHV-8 viral DNA sequences as previously described (11). The products were analyzed by agarose gel electrophoresis.
Pathologic Findings
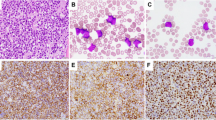
In routine histologic sections, abnormal lymphoid cells (Figure 1) diffusely infiltrated the peri-renal adipose and soft tissue. The cells were large (the nuclei were twice or more the size of the admixed small lymphocytes), with a rim of pale-staining cytoplasm. The nuclei were round to oval in shape with hyperchromatic, coarsely clumped chromatin. Occasional tumor cells showed greater nuclear pleomorphism, and some multinucleated tumor cells were present. Most cells had inconspicuous basophilic nucleoli, with rare cells containing larger nucleoli. The coarsely clumped chromatin gave the impression of a plasmacytoid appearance. Mitotic figures were very frequent (>5/high-power field), and scattered apoptotic debris was present. Large areas of necrosis were also seen. Vascular proliferation was not appreciated. Scattered mature plasma cells, small lymphocytes, and occasional eosinophils were admixed with tumor cells, particularly at the edge of the tumor infiltrate.
An extensive battery of immunohistochemical studies was performed (Figure 1). The tumor cells were uniformly positive for B-cell–associated antigens CD20 (L-26) and CD79a (mb-1), with staining restricted to the cytoplasm, suggesting the absence of a functional Ig receptor on the cell membrane. Stains for κ and λ identified infrequent plasma cells, but the tumor cells were Ig negative. The neoplastic cells were strongly positive for T-cell–associated antigens CD3, CD8, CD43 (Leu-22), and TIA-1. They were also positive for CD56, S-100, and Bcl-2. However, CD45 (LCA) was only focally positive in the tumor cells. Almost all tumor cells were immunoreactive for MIB-1, indicating a very high proliferative rate. The tumor cells were nonimmunoreactive for other T-cell–associated antigens tested, including CD5, CD4, and CD45RO (A6). CD4 and KP-1 stained infrequent histiocytes. βF-1 antibody staining for the β chain of the T-cell receptor complex was positive in the admixed small, benign-appearing T cells but was clearly negative in the tumor cells, raising the possibility of a γ/δ T-cell lymphoma. However, frozen tissue was not available for further studies to confirm the γ/δ T-cell origin of the tumor. Cyclin D1, CD10, CD30, P53, EBV- associated protein LMP-1, myeloperoxidase, lysozyme, AE1/AE3, and EMA were negative in the tumor cells. (Figs. 2 and 3.)
Immunohistochemical studies performed on paraffin sections. Neoplastic cells show cytoplasmic staining for B-cell antigens CD20 and CD79a (mb-1), despite a T-cell phenotype and genotype. A. CD20. B. CD79a. C. CD3. D. CD8. E. CD 56. F. TIA-1. Staining for CD79a is weaker and more variable in intensity than CD20 but is clearly positive in tumor cells (all images taken at ×400, hematoxylin counterstain).
PCR studies for IgH and T-cell receptor γ gene rearrangement indicate a T-cell derivation. A. T-cell receptor γ chain gene analysis. Clonal T-cell receptor γ chain gene rearrangements are present in both the Vγ/Jγ reaction (Lane 2) and the Vγ/Jp reaction (Lane 5). B. Immunoglobulin heavy chain gene analysis. A polyclonal ladder pattern is present without evidence of a clonal B-cell population (Lane 2).
Molecular studies for T-cell receptor gamma chain and immunoglobulin heavy chain gene rearrangements (VJ-PCR) were performed by PCR. VJ-PCR revealed a polyclonal ladder pattern without evidence of a clonal population. In contrast, PCR for TCR-γ chain gene rearrangement revealed a strong band by both Primer Set I and Primer Set II, indicating a clonal rearrangement of the TCR gene. Studies for HHV-8 viral sequences by PCR were negative. In situ hybridization for EBV was performed, but no positive signals were detected in either the normal or neoplastic cells, whereas the U6 control demonstrated presence of preserved mRNA in the tissue.
DISCUSSION
Recent studies in lymphoma classification have used immunophenotyping as an important tool aiding in the diagnosis of hematopoietic and lymphoid disorders. Recent technical advances such as antigen retrieval and new antibodies that can be applied on routine formalin- or B-5–fixed, paraffin-embedded tissues have also greatly improved and facilitated routine applications of immunohistochemical studies.
Mb-1 is a newly described antibody that can be applied to paraffin material. It detects CD79a, a 47-kDa subunit found in the disulfide-linked heterodimer associated with IgM on normal and neoplastic B-lymphocytes (12, 13). CD79a is expressed in precursor B-cells in the bone marrow and is retained throughout B-cell differentiation, with positive staining in most plasma cells. In contrast to CD20, staining intensity tends to vary among B-cell populations, with weaker staining in normal germinal centers, follicular lymphomas, and large B-cell lymphomas. Its high degree of specificity for B cells is thought to be in keeping with its important functional role in the antigen receptor complex, which initiates a cellular response after antigen binding. Mb-1 subserves a function in B cells very similar to that of CD3 in T cells (13).
The current case represents a very rare phenomenon in which a highly specific B-cell marker, mb-1, was expressed in a lymphoma that demonstrated evidence of T-cell derivation at the phenotypic and genotypic level. Interestingly, this tumor also expressed CD20. Expression of L26 has been reported in rare cases of T-cell lymphoma and on a small subpopulation of normal T-cells (14). Blakolmer et al. (6) recently reported the expression of CD79a in three cases of cytotoxic T-cell lymphoma, but their cases were CD20 negative. Thus, this case represents a novel phenomenon in which both CD20 and CD79a were expressed in a T-cell lymphoma. Arguing against a B-cell derivation in this case was clonal rearrangement of the TCR-γ chain gene, and a polyclonal pattern by VJ-PCR for the immunoglobulin heavy chain gene. The neoplastic cells failed to express immunoglobulin in the cytoplasm, and the T-cell–associated antigens CD3 and CD8 were present. Therefore, the preponderance of the evidence favored a T-cell derivation with aberrant expression of both CD20 and CD79a.
Mason et al. (13) reported the largest series to date analyzing the expression of Mb-1 in lymphomas and nonlymphoid malignancies. They studied 454 cases including a broad spectrum of B-cell (356 cases) and T-cell (70 cases) lymphomas and leukemias. Mb-1 also was analyzed in 28 cases of nonlymphoid malignancy. Mb-1 was immunoreactive in nearly 100% of all B-cell malignancies tested, including 41 cases of precursor B lymphoblastic lymphoma/leukemia. Mb-1–negative B-cell neoplasms were rare and were nearly restricted to the plasma cell stage of differentiation when CD79a is down-regulated. Mb-1 was positive in 10/20 cases of myeloma/plasmacytoma and in 13/15 cases of so-called B-cell anaplastic lymphoma. All other B-cell lineage neoplasms were Mb-1 positive. In contrast, none of the 70 cases of T cell lymphomas studied showed mb-1 positivity, including 9 cases of T lymphoblastic lymphoma/leukemia, 10 cases of mycosis fungoides, 32 cases of peripheral T cell lymphoma, 8 cases of angioimmunoblastic T cell lymphoma, and 11 cases of anaplastic large-cell lymphoma. All 28 cases of nonlymphoid malignancies were negative for mb-1.
In Hodgkin's lymphoma, mb-1 was positive in 100% of the cases of the lymphocyte predominance subtype, whereas only 20% of classical Hodgkin's lymphoma cases were positive (15). These results are in keeping with recent molecular findings indicating a B-cell lineage derivation for all types of Hodgkin's lymphoma. The absence of CD79a in classical Hodgkin's lymphoma is consistent with the view of the Reed-Sternberg cell as a crippled B cell, incapable of immunoglobulin secretion (16).
Pilozzi et al. (5) studied a larger number of immature lymphoid neoplasms and found coexpression of CD79a and CD3 in 10% of 149 cases of T-lymphoblastic lymphoma tested using the JCB117 antibody. Lineage infidelity and bi-lineage phenotypes are relative common in precursor lymphoid and myeloid malignancies while in contrast; lineage fidelity has been found to be the general rule in mature T and B cell tumors (4, 17).
Our laboratory and others have described cases of immunodeficiency-associated lymphoma with lineage infidelity at both the genotypic and phenotypic levels (18, 19, 20). These cases usually have been positive for EBV or HHV-8. In the current case, the patient was not positive for the human immunodeficiency virus and did not exhibit features of immunosuppression. The patient was 76 years old, and some authors have considered old age a time of immune senescence (21). Notably, both HHV-8 and EBV sequences were absent according to PCR and in situ hybridization.
Some of the findings in the current case suggested that it was of γδ T-cell origin. The neoplastic cells were negative for βF1, despite positive staining of admixed small lymphocytes. Studies for TCR δ expression could not be performed as frozen tissue was not available. The neoplastic cells had a cytotoxic phenotype and expressed CD56, features of most γδ T-cell lymphomas (22, 23). The clinical course of this case was highly aggressive, in keeping with the aggressive clinical behavior of T-cell lymphomas in general and of γδ T-cell lymphomas in particular (23). Interestingly, the tumor cells also were positive for S-100 protein. A subpopulation of normal cytotoxic CD8+ T-cells are S-100 positive (24, 25), and S-100 protein expression has been reported in cases of aggressive T-cell lymphoma/leukemia (26, 27). It is interesting that all of the cases of CD79a-positive T-cell lymphoma recently reported also had a cytotoxic phenotype (6).
In conclusion, this case calls attention to the complexity of lineage assignment in high-grade lymphomas. It suggests that no marker has absolute lineage specificity. It had been thought that CD79a, linked to the expression of immunoglobulin, was highly specific of B-cell lineage. Previously, it had been reported in precursor T-cell lymphoblastic malignancies, but lineage infidelity is common at the early stages of hematopoietic differentiation. Our case illustrates that no single marker can be considered specific and that immunophenotypic analysis should be performed with a broad panel of reagents, especially in high-grade tumors. Cases with aberrant phenotypes may require genotypic studies for lineage assignment. The aberrant expression of B-cell–associated antigens in this T cell lymphoma is most likely incidental but could be indicative of a normal antigenic phenotype not previously recognized.
References
Harris NL, Jaffe ES, Stein H, Banks PM, Chan JK, Cleary ML, et al. A revised European-American classification of lymphoid neoplasms: a proposal from the International Lymphoma Study Group. Blood 1994; 84: 1361–92.
Jaffe ES, Harris NL, Diebold J, Muller-Hermelink HK . World Health Organization classification of lymphomas: a work in progress. Ann Oncol 1998; 9 (5 Suppl):S25–30.
The Non-Hodgkin's Lymphoma Classification Project. A clinical evaluation of the International Lymphoma Study Group classification of non-Hodgkin's lymphoma. Blood 1997; 89: 3909–18.
Buccheri V, Mihaljevic B, Matutes E, Dyer MJ, Mason DY, Catovsky D . mb-1: a new marker for B-lineage lymphoblastic leukemia. Blood 1993; 82: 853–7.
Pilozzi E, Pulford K, Jones M, Muller-Hermelink HK, Falini B, Ralfkiaer E, et al. Co-expression of CD79a (JCB117) and CD3 by lymphoblastic lymphoma. J Pathol 1998; 186: 140–3.
Blakolmer K, Vesely M, Kummer JA, Jurecka W, Mannhalter C, Chott A . Immunoreactivity of B-cell markers (CD79a, L26) in rare cases of extranodal cytotoxic peripheral T- (NK/T-) cell lymphomas. Mod Pathol 2000; 13: 766–72.
Elenitoba-Johnson KSJ, Zarate-Osorno A, Meneses A, Krenacs L, Kingma DW, Raffeld M, et al. Cytotoxic granular protein expression, Epstein-Barr virus strain type, and latent membrane protein-1 oncogene deletions in nasal T-lymphocyte/natural killer cell lymphomas from Mexico. Mod Pathol 1998; 11: 754–61.
Kingma DW, Medeiros LJ, Barletta J, Raffeld M, Mann RB, Ambinder RF, et al. Epstein-Barr virus is infrequently identified in non-Hodgkin's lymphomas associated with Hodgkin's disease. Am J Surg Pathol 1994; 18: 48–61.
Segal GH, Jorgensen T, Scott M, Braylan RC . Optimal primer selection for clonality assessment by polymerase chain reaction analysis: II. follicular lymphomas. Hum Pathol 1994; 25: 1276–82.
McCarthy KP, Sloane JP, Kabarowski JH, Matutes E, Wiedemann LM . A simplified method of detection of clonal rearrangements of the T-cell receptor-gamma chain gene. Diagn Mol Pathol 1992; 1: 173–9.
Otsuki T, Kumar S, Ensoli B, Kingma DW, Yano T, Stetler-Stevenson M, et al. Detection of HHV-8/KSHV DNA sequences in AIDS-associated extranodal lymphoid malignancies. Leukemia 1996; 10: 1358–62.
Mason D, Cordell J, Tse A, van Dongen J, van Noesel C, Micklem K, et al. The IgM-associated protein mb-1 as a marker of normal and neoplastic B-cells. J Immunol 1991; 147: 2474–82.
Mason DY, Cordell JL, Brown MH, Borst J, Jones M, Pulford K, et al. CD79a: a novel marker for B-cell neoplasms in routinely processed tissue samples. Blood 1995; 86: 1453–9.
Quintanilla-Martinez L, Preffer F, Rubin D, Ferry JA, Harris NL . CD20+ T-cell lymphoma. Neoplastic transformation of a normal T-cell subset. Am J Clin Pathol 1994; 102: 483–9.
Korkolopoulou P, Cordell J, Jones M, Kaklamanis L, Tsenga A, Gatter KC, et al. The expression of the B-cell marker mb-1 (CD79a) in Hodgkin's disease. Histopathology 1994; 24: 511–5.
Kanzler H, Kuppers R, Hansmann ML, Rajewsky K . Hodgkin and Reed-Sternberg cells in Hodgkin's disease represent the outgrowth of a dominant tumor clone derived from (crippled) germinal center B cells. J Exp Med 1996; 184: 1495–505.
Cossman J, Uppenkamp M, Sundeen J, et al. Molecular genetics and the diagnosis of lymphoma. Arch J Pathol 1988; 134: 117–27.
Hollingsworth HC, Stetler SM, Gagneten D, Kingma DW, Raffeld M, Jaffe ES . Immunodeficiency-associated malignant lymphoma. Three cases showing genotypic evidence of both T- and B-cell lineages. Am J Surg Pathol 1994; 18: 1092–101.
Waller EK, Ziemianska M, Bangs CD, Cleary M, Weissman I, Kamel OW . Characterization of posttransplant lymphomas that express T-cell-associated markers: immunophenotypes, molecular genetics, cytogenetics, and heterotransplantation in severe combined immunodeficient mice. Blood 1993; 82: 247–61.
Knowles DM . Molecular pathology of acquired immunodeficiency syndrome-related non- Hodgkin's lymphoma. Semin Diagn Pathol 1997; 14: 67–82.
Lukes RJ, Tindle BH . Immunoblastic lymphadenopathy: a hyperimmune entity resembling Hodgkin's disease. N Engl J Med 1975; 292: 1–8.
Cooke CB, Krenacs M, Stetler-Stevenson M, Greiner TC, Raffeld M, Kingma DW, et al. Hepatosplenic gamma/delta T-cell lymphoma: a distinct clinicopathologic entity of cytotoxic gamma/delta T-cell origin. Blood 1996; 88: 4265–74.
Arnulf B, Copie-Bergman C, Delfau-Larue MH, Lavergne-Slove A, Bosq J, Wechsler J, et al. Nonhepatosplenic gammadelta T-cell lymphoma: a subset of cytotoxic lymphomas with mucosal or skin localization. Blood 1998; 91: 1723–31.
Kanamori M, Endo T, Shirakawa S, Sakurai M, Hidaka H . S-100 antigen in human T lymphocytes. Biochem Biophys Res Commun 1982; 108: 1447–53.
Sansoni P, Rowden G, Manara GC, Ferrari C, De Panfilis G . S-100-positive T cells are largely restricted to a CD8-positive, 9.3-negative subset. Virchows Arch B Cell Pathol Incl Mol Pathol 1987; 53: 301–7.
Zarate OA, Raffeld M, Berman EL, Ferguson MM, Andrade R, Jaffe ES . S-100-positive T-cell lymphoproliferative disorder. A case report and review of the literature. Am J Clin Pathol 1994; 102: 478–82.
Hanson CA, Bockenstedt PL, Schnitzer B, Fox DA, Kueck B, Braun DK . S100-positive, T-cell chronic lymphoproliferative disease: an aggressive disorder of an uncommon T-cell subset. Blood 1991; 78: 1803–13.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yao, X., Teruya-Feldstein, J., Raffeld, M. et al. Peripheral T-Cell Lymphoma with Aberrant Expression of CD79a and CD20: A Diagnostic Pitfall. Mod Pathol 14, 105–110 (2001). https://doi.org/10.1038/modpathol.3880265
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3880265
Keywords
This article is cited by
-
Cytotoxic peripheral T-cell lymphomas and EBV-positive T/NK-cell lymphoproliferative diseases: emerging concepts, recent advances, and the putative role of clonal hematopoiesis. A report of the 2022 EA4HP/SH lymphoma workshop
Virchows Archiv (2023)
-
Prognostic significance of aberrant expression of CD markers in acute lymphoblastic leukemia
memo - Magazine of European Medical Oncology (2017)
-
CD20-positive primary gastric T-cell lymphoma poorly responding to initial treatment with rituximab plus CHOP, and a literature review
International Journal of Hematology (2015)
-
CD20-positive T-cell lymphoma involving bone marrow: report of four cases
Journal of Hematopathology (2013)
-
CD20-positive NK/T-cell lymphoma with indolent clinical course: report of case and review of literature
Diagnostic Pathology (2012)