Abstract
Biological activities of oxysterols seem tightly regulated. Therefore, the ability to induce cell death of structurally related oxysterols, such as those oxidized at C7(7α-, 7β-hydroxycholesterol, and 7-ketocholesterol), was investigated on U937 cells at different times of treatment in a concentration range of 5–80 μg/ml. Whereas all oxysterols accumulate inside the cells, strong inhibition of cell growth and increased permeability to propidium iodide were observed only with 7β-hydroxycholesterol and 7-ketocholesterol, which trigger an apoptotic process characterized by the occurrence of cells with fragmented and/or condensed nuclei, and by various cellular dysfunctions: loss of mitochondrial transmembrane potential, cytosolic release of cytochrome c, activation of caspase-9 and -3 with subsequent enhanced activity of caspase-3, degradation of poly(ADP-ribose) polymerase, and increased accumulation of cellular C16 : 0 and C24 : 1 ceramide species. This ceramide generation is not attributed to caspase activation since inhibition of 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis by Z-VAD-fmk (100 μM), a broad spectrum caspase inhibitor, did not reduce C16 : 0 and C24 : 1 ceramide species accumulation. Conversely, when U937 cells were treated with 7β-hydroxycholesterol and 7-ketocholesterol in the presence of fumonisin B1 (100 μM), a specific inhibitor of ceramide synthase, C16 : 0 and C24 : 1 ceramide species production was completely abrogated whereas apoptosis was not prevented. Noteworthy, 7α-hydroxycholesterol induced only a slight inhibition of cell growth. Collectively, these results are consistent with the notion that the α or β hydroxyl radical position of oxysterols oxidized at C7 plays a key role in the induction of the apoptotic process. In addition, our findings demonstrate that 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis involve the mitochondrial signal transduction pathway and they suggest that C16 : 0 and C24 : 1 ceramide species generated through ceramide synthase play a minor role in the commitment of 7β-hydroxycholesterol- and 7-ketocholesterol-induced cell death.
Similar content being viewed by others
Introduction
Oxysterols are 27-carbon derivatives of cholesterol that contain additional oxygen atoms either on the steroid nucleus or on the side chain.1 These compounds constitute a wide family of molecules resulting either from the auto-oxidation of cholesterol in air, or from the enzyme-catalyzed transformation of cholesterol in various cell species.2,3 To date, oxysterols, which are present in various amounts in different kinds of foods,3,4,5 have been shown to possess diverse biological activities,6,7 and some of them are strongly cytotoxic to both normal and tumoral cells of human origin and from various animal species.8,9 However, from one oxysterol to another, important variations in the ability to induce cell death were observed. Thus, in cultured aortic smooth muscle cells the toxic potency differed with the oxysterol considered.10 In bovine aortic endothelial cells, we demonstrated that 7β-hydroxycholesterol and 7-ketocholesterol were more cytotoxic than 19-hydroxycholesterol, cholesterol 5α, 6α-epoxide, and 25-hydroxycholesterol, and that their cytotoxicity was accompanied by an enhanced proportion of cells with fragmented and/or condensed nuclei characteristic of apoptotic cells.11 In human umbilical venous endothelial cells (HUVECs),12 in human, rabbit, and rat smooth muscle cells,12,13 as well as in leukemic cells such as U937 promonocytic human leukemia cells,9,14,15 typical features of apoptosis were also observed under treatment with 7β-hydroxycholesterol and 7-ketocholesterol but not with 7α-hydroxycholesterol.16 Taken together, these different observations underline the complexity of the biological activities of oxysterols, mainly of those oxidized at C7 (7α-, 7β-hydroxycholesterol and 7-ketocholesterol). Indeed, these oxysterols are detected in appreciable quantities in human tissues and fluids, including human plasma and atheromatous lesions,17 in oxidized low density lipoproteins (oxLDL) which play a key role at different stages of the atherosclerotic process,18,19 as well as in various kinds of food (powdered milk, cheese and egg products) especially when they are heated in air during processing and stored for a lengthy period.20 Therefore, it is of importance to investigate the cytotoxicity of these oxysterols oxidized at C7, to precisely determine the mode of cell death that they induce,21,22 and to elucidate the signaling pathway(s) triggered by these compounds.
Since the biological activities of 7α-,7β-hydroxycholesterol and 7-ketocholesterol seem tightly regulated,16,19 the cytotoxicities of these molecules were investigated in U937 cells in a range of concentrations varying from 5 to 80 μg/ml (12.5–200 μM) after 30 min to 30 h of treatment by using different criteria: cell counting; flow cytometric measurement of cell permeability with propidium iodide;11 observation by transmission electron microscopy; determination by fluorescence microscopy of the proportion of apoptotic cells displaying condensed and/or fragmented nuclei after DNA staining with Hoechst 33342;23 quantification of cellular oxysterol concentration by gaseous phase chromatography coupled with mass spectrometry;24 flow cytometric quantification of mitochondrial transmembrane potential (Δψm) with the cationic lipophilic dye DiOC6(3);25 Western blot analysis of the release of cytochrome c into the cytosol; activation of caspase-9 (MACH6/ICE-LAP6) and -3 (CPP32/apopain/Yama), and degradation of poly(ADP-ribose) polymerase (PARP).26 In addition, caspase-3 activity was simultaneously measured by a fluorogenic assay. According to these different criteria, 7α-hydroxycholesterol was not cytotoxic and induced only a slight inhibition of cell growth, whereas it accumulates in the cells in higher amounts than 7β-hydroxycholesterol and 7-ketocholesterol. As for 7β-hydroxycholesterol and 7-ketocholesterol, they were potent inducers of cell death by apoptosis characterized by the following dysfunctions: inhibition of cell growth, increased permeability to propidium iodide, fragmentation and/or condensation of the nuclei, mitochondrial depolarization, cytochrome c release into the cytosol, activation of caspase-9 and -3, and PARP degradation.
Since ceramides seem of fundamental importance in some signaling pathways leading to apoptosis,27,28,29 we further asked whether the different potencies of oxysterols to induce apoptosis could depend on their ability to stimulate ceramide generation. Thus, by electrospray ionization mass spectrometry, an important increase of cellular C16 : 0 and C24 : 1 ceramide accumulation was detected after treatment with 7β-hydroxycholesterol and 7-ketocholesterol, but not with 7α-hydroxycholesterol. Interestingly, simultaneous incubation of 7β-hydroxycholesterol and 7-ketocholesterol with Z-VAD-fmk (100 μM), a wide spectrum caspase inhibitor,30 impaired the apoptotic process but not ceramide generation, and otherwise simultaneous incubation of 7β-hydroxycholesterol and 7-ketocholesterol with fumonisin B1, a specific inhibitor of ceramide synthase,31 completely inhibited C16 : 0 and C24 : 1 ceramide species generation without inhibiting apoptosis.
The reported data confirm that 7α-hydroxycholesterol has no cytoxic effects, underlining that the pro-apoptotic effects of oxysterols oxidized at C7 depend not only on the radical (hydroxyl or keto) present at C7, but also on the α or β position of the hydroxyl radical, and they demonstrate that 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis characterized by a loss of mitochondrial transmembrane potential (Δψm), a release of cytochrome c into the cytosol, an activation of caspase-9 and -3, and a PARP degradation, is independent of the endogeneously generated C16 : 0 and C24 : 1 ceramide species.
Results
Effects of 7α-,7β-hydroxycholesterol and 7-ketocholesterol on cell growth and on cell permeability to propidium iodide
Characterization of the effects of 7α-,7β-hydroxycholesterol and 7-ketocholesterol (5, 10, 20, 40 and 80 μg/ml) on cell growth and on cell permeability to propidium iodide was performed on U937 cells at 18, 24 and 30 h of treatment. As shown in Figure 1, treatment of U937 cells with 7β-hydroxycholesterol and 7-ketocholesterol was associated with an inhibition of cell growth occuring in a dose-and time-dependent manner. Thus, as early as 18 h after treatment has begun, 7β-hydroxycholesterol and 7-ketocholesterol significantly (P<0.05) inhibited cell growth when they were used at minimum concentrations of 10 and 20 μg/ml, respectively, and the most significant inhibition of cell proliferation for each compound was observed at 30 h of treatment (Figure 1). Interestingly, 7β-hydroxycholesterol was the strongest inhibitor of cell proliferation regardless of the time of treatment. When cells were treated with 7α-hydroxycholesterol, a slight but significant inhibition of cell growth (P<0.05) was observed only at 30 h of culture, when this oxysterol was used at the highest concentration investigated (80 μg/ml) (Figure 1). Because of the complex relationships between inhibition of cell growth and modification of cellular membrane integrity, flow cytometric analyses of cell permeability were performed with propidium iodide, which distinguishes between dead and living cells.32 In those conditions, strong increases in the proportions of propidium iodide positive cells were detected after 18 h of treatment with 7β-hydroxycholesterol and 7-ketocholesterol when these compounds were used at minimum concentrations of 20 and 40 μg/ml, respectively, and the highest proportions of propidium iodide permeable cells were identified at 30 h of culture (Figure 1). Noteworthy, among the oxysterols oxidized at C7 (7α-,7β-hydroxycholesterol and 7-ketocholesterol), 7β-hydroxycholesterol induced the highest proportion of cells permeable to propidium iodide regardless of the time of treatment, whereas no effect was observed with 7α-hydroxycholesterol (Figure 1).
Concentration and time-dependent effects of 7α, 7β-hydroxycholesterol and 7-ketocholesterol on cell growth and on cell permeability to propidium iodide. U937 cells were treated for 18, 24, and 30 h with either 7α-hydroxycholesterol, 7β-hydroxycholesterol, or 7-ketocholesterol in a range of concentrations from 5 to 80 μg/ml. Cell growth was determined by cell counting, and the permeability to propidium iodide was quantified by flow cytometry. Data are mean±S.D. of four independent experiments performed in triplicate
Characterization of oxysterol-induced cell death
When U937 cells were cultured in the presence of 7β-hydroxycholesterol or 7-ketocholesterol (5, 10, 20, 40 and 80 μg/ml) for 18, 24, and 30 h, some cells with fragmented and/or condensed nuclei characteristic of apoptotic cells were identified by transmission electron microscopy, as well as by fluorescence microscopy after nuclei staining with Hoechst 33342 (Figures 2 and 3). The latter method, which was used to quantify apoptosis, showed a significant (P<0.05) increase in apoptotic cells as early as 18 h of treatment when 7β-hydroxycholesterol and 7-ketocholesterol were used at minimum concentrations of 20 and 40 μg/ml, respectively, and whatever the time of treatment the highest proportion of apoptotic cells was obtained with 7β-hydroxycholesterol (Figure 4). When U937 cells were treated with 7α-hydroxycholesterol, the morphological aspect of the cells and of their nuclei were the same as those of untreated cells when the observations were performed either by transmission electron microscopy or by fluorescence microscopy after nuclei staining with Hoechst 33342 (Figures 2 and 3). Thus, regardless of the time of treatment considered (18, 24, and 30 h), 7α-hydroxycholesterol used at 5, 10, 20, 40, and 80 μg/ml never induced apoptosis when this mode of cell death was characterized by the occurrence of cells with fragmented and/or condensed nuclei (Figure 4).
Morphological characterization of 7α-, 7β-hydroxycholesterol and 7-ketocholesterol-treated cells by transmission electron microscopy. U937 cells were cultured for 24 h in the absence (control) or in the presence of 7α-hydroxycholesterol (80 μg/ml), 7β-hydroxycholesterol (20 μg/ml), or 7-ketocholesterol (40 μg/ml), and the morphological aspects of the cells were analyzed by transmission electron microscopy (magnification×3000): (A), control corresponding to untreated cells; (B), 7α-hydroxycholesterol-treated cells. 7α-hydroxycholesterol-treated cells are morphologically similar to untreated cells; (C), 7β-hydroxycholesterol-treated cells; (D), 7-ketocholesterol-treated cells. Apoptotic cells with fragmented nuclei (large arrows) obtained after treatment with 7β-hydroxycholesterol and 7-ketocholesterol are shown in C and D
Morphological characterization of 7α-,7β-hydroxycholesterol and 7-ketocholesterol-treated cells by fluorescence microscopy. U937 cells were cultured for 24 h in the absence (control) or in the presence of 7α-hydroxycholesterol, 7β-hydroxycholesterol, or 7-ketocholesterol in a range of concentrations from 5 to 80 μg/ml, and the morphological aspects of the cells were analyzed by fluorescence microscopy after nuclei staining with Hoechst 33342 (magnification×450): (A), control corresponding to untreated cells; (B), 7α-hydroxycholesterol-treated cells (80 μg/ml); (C), 7β-hydroxycholesterol-treated cells (20 μg/ml); (D), 7-ketocholesterol-treated cells (40 μg/ml). Apoptotic cells with fragmented (large arrows) and/or condensed nuclei (small arrows) were observed after treatment with 7β-hydroxycholesterol and 7-ketocholesterol but not among 7α-hydroxycholesterol-treated cells, which were morphologically similar to untreated cells
Concentration and time-dependent effect of 7α-,7β-hydroxycholesterol and 7-ketocholesterol on the induction of apoptotic cells. To quantify the percentage of apoptotic cells, U937 cells were treated for 18, 24, and 30 h with either 7α-hydroxycholesterol (80 μg/ml), 7β-hydroxycholesterol (20 μg/ml), or 7-ketocholesterol (40 μg/ml). Apoptotic cells characterized by condensed and/or fragmented nuclei were identified by fluorescence microscopy after DNA staining with Hoechst 33342. Data are mean±S.D. of four independent experiments performed in triplicate
For further characterization of cell death, 7β-hydroxycholesterol and 7-ketocholesterol were used at concentrations of 20 and 40 μg/ml, respectively, since these were the lowest concentrations able to induce maximal proportions of apoptotic cells at 24 and 30 h of treatment (Figure 4). As 7α-hydroxycholesterol was not an inducer of apoptosis, it was used at 80 μg/ml corresponding to the highest concentration investigated.
Quantification of the intracellular uptake of 7α-,7β-hydroxycholesterol and 7-ketocholesterol
In order to define whether the differences of toxicity between oxysterols oxidized at C7 were due to different uptakes from one oxysterol to another, the quantity of oxysterol (per cell) was quantified at 24 h of treatment by capillary gas chomatography coupled to mass spectrometry. In these conditions, when the cells were treated for 24 h in the presence of oxysterols used at final concentrations of 5, 10, 20, 40 and 80 μg/ml, the uptake of 7α-hydroxycholesterol was approximately twofold higher than those obtained with 7β-hydroxycholesterol and 7-ketocholesterol. Thus, when cells were treated with 7α-hydroxycholesterol at concentrations of 5 to 80 μg/ml, the uptake of this oxysterol varied from 8.22±1.8 to 104.95±14.21 μg/106 cells (Table 1). When cells were treated with 7β-hydroxycholesterol and 7-ketocholesterol at final concentrations of 5 to 80 μg/ml, the uptakes of these oxysterols were similar and varied from 3.40±1.30 to 65.00±5.50 μg/106 cells, and from 4.30±1.38 to 53.08±7.94 μg/106 cells, respectively (Table 1).
Effect of 7α-,7β-hydroxycholesterol and 7-ketocholesterol on mitochondrial transmembrane potential (Δψm) and on cytochrome c release into the cytosol
Disruption of Δψm and cytochrome c release from the intermembrane space of mitochondria into the cytosol seems a general feature of apoptosis.33 Therefore, in the presence of 7β-hydroxycholesterol (20 μg/ml) and 7-ketocholesterol (40 μg/ml), the evolution of Δψm and the processing of cytochrome c release were simultaneously quantified after various treatment times (18, 24, and 30 h) by flow cytometry with the cationic lipophilic dye DiOC6(3), and by Western blot with a mouse monoclonal antibody directed against cytochrome c (molecular weight 15 kD), respectively. The levels of cytochrome c release into the cytosol were quantified comparatively to the constant level of a cross reacting protein of around 65 kD present in the cytosolic extract. When compared to untreated cells, treatment with 7β-hydroxycholesterol and 7-ketocholesterol resulted in a time-dependent increase of the percentage of depolarized cells (characterized by low values of Δψm) which were observed as early as 18 h after treatment, and the effects were more marked with 7β-hydroxycholesterol than with 7-ketocholesterol (Table 2). This increase in the percentage of cells with low Δψm was associated with a cytosolic release of cytochrome c which was already significant at 18 h of treatment; with 7-ketocholesterol, the cytosolic release of cytochrome c was slightly detected at 18 h, but was more and more pronounced at 24 and 30 h of treatment (Table 2). 7α-hydroxycholesterol induced only a slight but not significant increase in the proportion of depolarized cells, and when treated with this oxysterol no cytosolic release of cytochrome c was observed (Table 2).
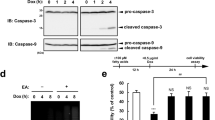
Analysis of caspase-9 and -3 activation, and of poly(ADP-ribose) polymerase degradation in 7α-, 7β-hydroxycholesterol- and 7-ketocholesterol-treated U937 cells
As the loss of mitochondrial transmembrane potential (Δψm) and the release of cytochrome c generally leads to subsequent activation of caspase-9, and -3, as well as to PARP degradation,34,35,36 the occurrence of this cascade of events was investigated during 7β-hydroxycholesterol and 7-ketocholesterol-induced apoptosis. To this end, U937 cells were treated for 18, 24, and 30 h with 7β-hydroxycholesterol (20 μg/ml), or 7-ketocholesterol (40 μg/ml), and Western blot analysis was used to demonstrate the involvement of caspase-9 and -3, as well as PARP degradation. In addition, caspase-3 activity was simultaneously measured by a fluorogenic assay based on the cleavage of Ac-DEVD-7-amino-4-methylcoumarin. When compared to untreated cells, incubation with 7β-hydroxycholesterol resulted in the degradation of procaspase-9 as well as in the cleavage of procaspase-3 (32 kD) in its active subunit (17 kD) as early as 18 h after treatment (Table 3). In the presence of 7-ketocholesterol, degradation of procaspase-9 and cleavage of procaspase-3 were not observed at 18 h but they began to be detected at 24 h, and were more pronounced at 30 h (Table 3). This cleavage of procaspase-3 occurring when treated with 7β-hydroxycholesterol and with 7-ketocholesterol was associated with an increase of caspase-3 activity (Table 3). In untreated cells and in the presence of 7α-hydroxycholesterol (80 μg/ml), no degradation of procaspase-9, no cleavage of procaspase-3, and no caspase-3 activity were ever observed (Table 3).
The findings that oxysterols induce the activation of caspase-3 prompted an analysis of poly(ADP-ribose) polymerase (PARP) cleavage since one of the potential substrates of caspase-3 during apoptosis is PARP (115 kD), an enzyme that appears to be involved in DNA repair, genome surveillance, and integrity.37 So, PARP degradation was also investigated by Western blot analysis of 7β-hydroxycholesterol (20 μg/ml), and 7-ketocholesterol (40 μg/ml)-treated U937 cells taken at 18, 24 and 30 h of incubation. Immunoblot analysis with an antibody recognizing both the full length and the cleaved form of PARP (115 and 85 kD) revealed an early cleavage of PARP occurring after 18 h of treatment with 7β-hydroxycholesterol, whereas in the presence of 7-ketocholesterol, PARP degradation was observed only at 24 and 30 h of treatment (Table 3). In untreated cells, as well as in 7α-hydroxycholesterol (80 μg/ml)-treated cells, no PARP degradation was found regardless of the time of treatment (Table 3).
Generation of C16 : 0 and C24 : 1 ceramide species when treated with 7β-hydroxycholesterol and 7-ketocholesterol
Ceramide is a group of neutral sphingolipids that mediate several cellular processes38 and constitute important secondary messenger molecules mainly in the regulation of cell growth, and in apoptosis signaling.27 As cellular ceramide generation is frequently observed after treatment with various apoptosis inducers,39 we asked whether ceramide synthesis was upregulated during 7β-hydroxycholesterol and 7-ketocholesterol-induced apoptosis. To this end, U937 cells were treated for 30 min, 1, 6, 12, 18, 24, and 30 h with 7β-hydroxycholesterol (20 μg/ml), or 7-ketocholesterol (40 μg/ml), and ceramide species were characterized and quantified by electrospray ionization mass spectrometry. As previously mentioned, the major ceramide species present in U937 cells were C16 : 0 and C24 : 1.40 The cellular content of these predominant ceramides increased with the time of treatment (Figure 5), whereas there was no generation of any previously undetected ceramide species following cell stimulation at any time (data not shown). Thus, significant increases (P<0.05) of C16 : 0 were observed after 6 and 12 h of treatment with 7β-hydroxycholesterol and 7-ketocholesterol, respectively, and with each oxysterol, a plateau was reached at 18 h of treatment (Figure 5A). Similar profiles were found with C24 : 1 (Figure 5B). At the plateau, C16 : 0 and C24 : 1 content (per cell) were three to four times higher in 7β-hydroxycholesterol- than in 7-ketocholesterol-treated cells, and C24 : 1 content (per cell) was always four to five times lower than C16 : 0 content (Figure 5). In untreated cells, as well as in the presence of 7α-hydroxycholesterol (80 μg/ml), no ceramide generation was ever observed (Figure 5). Therefore, even at a high concentration (80 μg/ml), 7α-hydroxycholesterol is unable to induce apoptosis or to stimulate ceramide generation. As the aim of further experiments was to establish the relationship between apoptosis and ceramide synthesis, the following investigations were only performed on 7β-hydroxycholesterol- and 7-ketocholesterol-treated cells.
Effects of 7α-,7β-hydroxycholesterol and 7-ketocholesterol on ceramide generation. C16 : 0 (A) and C24 : 1 (B) ceramide changes quantified by electrospray ionization mass spectrometry were studied in U937 cells cultured for 30 min, 1, 6, 12, 18, 24, and 30 h in the absence (control) or in the presence of 7α-hydroxycholesterol (80 μg/ml), 7β-hydroxycholesterol (20 μg/ml), or 7-ketocholesterol (40 μg/ml). Data are mean±S.D. of three independent experiments performed in triplicate
Z-VAD-fmk counteracts 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis but does not reduce ceramide generation
The mechanisms by which ceramide generation is related to apoptosis have not yet been fully addressed. Because ceramide accumulation occurred concomitantly with enhanced caspase-3 activation, which is an important effector caspase with numerous substrates,41,42 we asked whether C16 : 0 and C24 : 1 ceramide generation depended on caspase-3. To this end, U937 cells were treated for 24 h with 7β-hydroxycholesterol (20 μg/ml) or 7-ketocholesterol (40 μg/ml) in the absence or in the presence of Z-VAD-fmk (100 μM), which is a broad spectrum cell-permeable caspase inhibitor.30 Taken alone, Z-VAD-fmk did not increase the percentage of apoptotic cells, of propidium iodide permeable cells, or of cells with depolarized mitochondria (cells with low Δψm) (Table 4). Z-VAD-fmk had also no effect on either ceramide generation, caspase-3 activity, caspase-3 cleavage or PARP degradation (Table 4). When Z-VAD-fmk was added to the culture medium 30 min before 7β-hydroxycholesterol (20 μg/ml) or 7-ketocholesterol (40 μg/ml), the percentage of apoptotic cells characterized by condensed and/or fragmented nuclei after staining with Hoechst 33342 was significantly reduced, cleavage in the small subunit of caspase-3 was completely inhibited, caspase-3 activity was strongly impaired and PARP-degradation was counteracted (Table 4). However, as shown in Table 4, pretreatment of cells with Z-VAD-fmk had no effect on cell permeability to propidium iodide and on mitochondrial depolarization measured with the cationic lipophilic dye DiOC6(3). Likewise, C16 : 0 and C24 : 1 ceramide generation was similar when the cells were treated with 7β-hydroxycholesterol- or 7-ketocholesterol in the absence or in the presence of Z-VAD-fmk (Table 4). Thus, Z-VAD-fmk is able to inhibit certain related events that occur during 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis (morphological changes of the nuclei, induction of caspase-3 activity, cleavage of caspase-3 and PARP degradation), but not cell permeability to propidium iodide, mitochondrial depolarization, and C16 : 0 and C24 : 1 ceramide generation. Consequently, in our model of apoptosis, ceramide generation does not depend on caspase-3.
Fumonisin B1 blocks C16 : 0 and C24 : 1 ceramide species generation but not 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis
Various metabolic pathways can contribute to ceramide synthesis,38,39 but the late occurrence of ceramide generation when treated with 7β-hydroxycholesterol (20 μg/ml) and 7-ketocholesterol (40 μg/ml) rather suggests a stimulation of ceramide synthesis by the enzyme ceramide synthase.43 Therefore, U937 cells were treated for 24 h with 7β-hydroxycholesterol (20 μg/ml) or 7-ketocholesterol (40 μg/ml) in the absence or in the presence of fumonisin B1 (100 μM), a specific inhibitor of ceramide synthase.31 Under those conditions, similar proportions of apoptotic cells, of propidium iodide permeable cells, and of cells with depolarized mitochondria (characterized by low values of Δψm) were detected in untreated- and in fumonisin B1-treated cells (Table 5). Noteworthy, when fumonisin B1 was added to the culture medium 30 min before 7β-hydroxycholesterol (20 μg/ml) or 7-ketocholesterol (40 μg/ml), ceramide generation was strongly reduced but no significant decreases in the percentages of apoptotic cells, of propidium iodide permeable cells, and of cells with depolarized mitochondria were observed (Table 5). Therefore, 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis does not seem mediated by a de novo pathway of C16 : 0 and C24 : 1 ceramide species generation involving ceramide synthase. In addition, our data also suggest that C16 : 0 and C24 : 1 ceramide species generation through ceramide synthase activity is not involved in the occurrence of cell permeability to propidium iodide and in the loss of transmembrane mitochondrial potential (Δψm).
Discussion
Due to the fundamental role of apoptosis in physiological and in pathological processes, it is of importance to identify the molecules capable of inducing this form of cell death and to characterize their mechanisms.33,44 Among these pro-apoptotic molecules, oxysterols (which are oxygenated derivatives of cholesterol, and which result from the oxidation of cholesterol essentially on its B ring or on its side chain) constitute a wide class of compounds1,3 that could be involved in Alzheimer's disease45 and that probably play a key role in the initiation and in the development of atherosclerosis.46 Indeed, some of them, such as 7β-hydroxycholesterol and 7-ketocholesterol are potent inducers of apoptosis in tumoral and normal cells,14,47,48 especially those of the nervous system49,50,51 and of the vascular wall.12 As 7α-hydroxycholesterol is not cytotoxic, the biological effects of oxysterols oxidized at C7 (7α-, 7β-hydroxycholesterol, and 7-ketocholesterol) seem tightly regulated, and it was therefore of interest to compare, and to characterize their cytotoxic effects. To this end, human U937 promyelocytic leukemia cells were treated with oxysterol oxidized at C7 at concentrations (5–80 μg/ml) that were in the range of levels measured in the plasma of hypercholesterolemic patients52 and of cholesterol-rich fed rabbits.53 U937 cells were chosen because they are sensitive to oxysterols in the same range of concentrations as those observed in nerve cells49,50 as well as in endothelial and smooth muscle cells.12 In addition, U937 cells are frequently used as macrophage-like reference models to investigate the cytotoxicity of oxysterols in humans,9,14,15 as it is well admitted that macrophages play an important role in the atherosclerotic process.47
Under those conditions, with the use of different criteria (cell counting, permeability to propidium iodide, morphological aspect of cellular nuclei, mitochondrial depolarization, cytochrome c release into the cytosol, caspase-9 and -3 activation, PARP degradation, quantification and characterization of ceramide species) we report that 7α-hydroxycholesterol induces only a slight inhibition of cell growth, and that the apoptotic potency is higher with 7β-hydroxycholesterol than with 7-ketocholesterol, although these oxysterols trigger similar signaling pathways.
So, as reported before on a wide variety of normal or tumoral cell types deriving from the digestive, immune, nervous and cardiovascular systems,11,49,54,55 the present in vitro investigation underlines again the important differences of toxicity from one oxysterol to another even when these compounds are structurally identical as 7α- and 7β-hydroxycholesterol. Noteworthy, as no cytoxicity of 7α-hydroxycholesterol was previously observed on lymphoma and hepatoma cells,56,57 the present investigation suggests that the activity of this compound would not depend on the cell type considered as is frequently the case with some oxysterols. Thus, 25-hydroxycholesterol displays slight cytotoxic activities towards lymphoma and hepatoma cells as well as towards endothelial cells, but it is strongly cytotoxic against microglial cells while 7β-hydroxycholesterol acts in the opposite manner.11,49,56,57 Moreover, our data also confirm the important cytotoxic effects of 7β-hydroxycholesterol and of 7-ketocholesterol that were described to induce either apoptosis or necrosis according to the cells considered,12,55 and they raise further the interest in determining the cell death characteristics linked to these compounds.
In the present work, in agreement with our previous studies,9,15 we also report that 7β-hydroxycholesterol and 7-ketocholesterol- induced cell death is a form of apoptosis characterized by a reduction of cell growth, an enhanced permeability to propidium iodide (which is in agreement with the alterations of the physical properties of lipid membranes observed in the presence of various oxysterols by angle-resolved fluorescence depolarization microscopy and electron spin resonance),58 and an increased proportion of cells with condensed and/or fragmented nuclei. The significant reduction of cell growth associated with a marked increase permeability to propidium iodide occuring during treatment with 7β-hydroxycholesterol and 7-ketocholesterol is probably a complex phenomenon that could be the consequence of the simultaneous downregulation of proliferation signals and of the complete disruption of cellular structures in late steps of the apoptotic process, as it is frequently the case with various pro-apoptotic compounds.59 Similarly, the occurrence of cells with fragmented and/or condensed nuclei, which is a characteristic feature of apoptosis,23 also involves complex signaling pathways that have not yet been completely delineated, and a variety of models associated with apoptotic morphology have been described.21 Nowadays, three compartments are implicated in the execution of apoptotic processes;60 these compartments are the plasma membrane where both death and survival receptors reside, the mitochondrion, which is home for several proteins that regulate apoptosis, and the endoplasmic reticulum involving caspase-12.33,60,61 When treated with 7β-hydroxycholesterol and 7-ketocholesterol, the loss of the mitochondrial trasmembrane potential (Δψm) and the simultaneous release of cytochrome c into the cytosol show that the mitochondrial compartment is involved in the apoptotic process, and the requirement of these mitochondrial events in the induction of the downstream biochemical changes leading to caspase-9 and -3 activation, and to PARP degradation, is suggested by the ability of various antioxidants (glutathion, N-acetylcysteine, and vitamin E) to counteract 7-ketocholesterol-induced cell death.15,62 Indeed, we previously demonstrated that impairment of 7-ketocholesterol-induced apoptosis by glutathion, N-acetylcysteine and vitamin E correlates with the prevention of mitochondrial dysfunctions, i.e, loss of the trasmembrane mitochondrial potential (Δψm) and simultaneous release of cytochrome c into the cytosol.62 Therefore, there is considerable evidence that the disruption of mitochondrial functions are major events in 7β-hydroxycholesterol and 7-ketocholesterol-induced apoptosis. In addition, similarly to numerous inducers of apoptosis,34,63 7β-hydroxycholesterol and 7-ketocholesterol also induce an apoptotic mode of cell death where casapases play important roles. Indeed, we report in the present study that Z-VAD-fmk (a broad-spectrum caspase inhibitor)30 counteracts the cleavage and activity of caspase-3, and consequently PARP degradation and morphological changes associated with apoptosis (occurence of cells with fragmented and / or condensed nuclei). However, Z-VAD-fmk does not inhibit the loss of transmembrane mitochondrial potential and the permeability to propidium iodide known to enter only in dead cells.32 Therefore, in oxysterol-induced apoptosis as in numerous other types of apoptotic processes,64 the inhibition of casapases-dependent events by Z-VAD-fmk is not sufficient to inhibit the commitment to cell death. Taken together these different data led us to speculate that 7β-hydroxycholesterol and 7-ketocholesterol would trigger the following post-mitochondrial cascade of events: cytochrome c release into the cytosol would contribute to the activation of a complex of apoptosis activating factor 1 (Apaf-1) and of pro-caspase-9, leading to the cytochrome c dependent processing of pro-caspase-3 which in turn would cleave several death substrates including poly (ADP-ribose) polymerase (PARP).33,65,66
Because cell signals upstream of mitochondrial dysfunctions resulting from 7β-hydroxycholesterol and 7-ketocholesterol-induced apoptosis are yet unknown, and since some ceramide species that play an important role in the transduction of the apoptotic signal27,38 were also found to induce mitochondrial dysfunctions such as loss of mitochondrial transmembrane potential and cytochrome c release,67,68,69 we further investigated changes of ceramide concentration (per cell) after various treatment times in the presence of 7β-hydroxycholesterol and 7-ketocholesterol. The present study is consistent with the findings that the predominant ceramide species present in U937 cells are C16 : 0 and C24 : 1,40 and it demonstrates with the use of Z-VAD-fmk that ceramide generation occurring in U937 cells following treatment with 7β-hydroxycholesterol and 7-ketocholesterol does not depend on caspase activation, especially activation of caspase-3, which is an important effector caspase with numerous substrates.42 Indeed, in U937 cells incubated with 7β-hydroxycholesterol (or 7-ketocholesterol), activation of caspase-3 was inhibited and the percentage of cells with fragmented and/or condensed nuclei characteristic of apoptotic cells was reduced in the presence of Z-VAD-fmk whereas the accumulation of C16 : 0 and C24 : 1 ceramide species was not modified. In addition, the present work underlines that ceramide generation occuring during treatment with 7β-hydroxycholesterol and 7-ketocholesterol (but not with 7α-hydroxycholesterol) is mediated through ceramide synthase, as it is in daunorubicin-induced apoptosis in hen granulosa cells,70 in CPT-11-induced apoptosis in L929 cells,71 and in 12-O-tetradecanoylphorbol-13-acetate-induced apoptosis in LNCaP cells.72 So, in U937 cells, hen granulosa cells, L929 cells, and LNCaP cells, incubated in the presence of oxysterol, daunorubicin, CPT-11, and 12-O-tetradecanoylphorbol-13-acetate, respectively, it was possible to abrogate ceramide accumulation with fumonisin B1, a fungal toxin that specifically inhibits ceramide synthase.31 However, contrary to the data obtained for daunorubicin-, CPT-11-, and 12-O-tetradecanoylphorbol-13-acetate-treated cells, abrogation of ceramide generation in oxysterol-treated cells did not abolish apoptosis. Therefore, as in the case of apoptosis triggered by oxidized low density lipoproteins73 which are known to contain high levels of 7β-hydroxycholesterol and of 7-ketocholesterol,74 our results rather suggest that ceramide generation (especially C16 : 0 and C24 : 1 ceramide species) is not a main event in the death response to 7β-hydroxycholesterol and 7-ketocholesterol probably because ceramide generation (in our model of cell death) has minor effects on central executioners of the apoptotic process such as mitochondria. Indeed, fumonisin B1, which strongly reduces the accumulation of C16 : 0 and C24 : 1 ceramide species induced by 7β-hydroxycholesterol and 7-ketocholesterol, does not prevent the loss of transmembrane mitochondrial potential (Δψm). In addition, our results also suggest that C16 : 0 and C24 : 1 ceramide species are probably not involved in the regulation of cell permeability to propidium iodide since fumonisin B1, which strongly inhibits ceramide generation triggered by 7β-hydroxycholesterol or by 7-ketocholesterol, does not simultaneously reduce the proportion of propidium iodide permeable cells. In support of the idea that ceramide generation is however implied in the control of several cellular activities particularly in apoptotically dying cells,38 further studies are therefore needed to clarify the role of ceramide generation during oxysterol-induced cell death. However, as some oxysterols such as 25-hydroxycholesterol75 and 7β-hydroxycholesterol76 have been shown to favor the accumulation of the cells in the G0/G1 phase of the cell cycle, and as increase ceramide generation has been described in yeast77 as well as in Molt-4 cells78 blocked in G1, we can suppose a link between ceramide generation and regulation of the cell cycle during oxysterol-induced apoptosis.
Noteworthy, contrary to 7β-hydroxycholesterol and 7-ketocholesterol, no cytoxicity was observed in the presence of 7α-hydroxycholesterol, and the only cellular dysfunction identified was a slight inhibition of cell growth at the highest concentration considered (80 μg/ml). As all oxysterols oxidized at C7 (7α-hydroxycholesterol, 7β-hydroxycholesterol and 7-ketocholesterol) accumulate inside the cells, the differences of activity between 7α-hydroxycholesterol and 7β-hydroxycholesterol, which are structurally analogous but stereologically different, suggest that the biological activities of oxysterols oxidized at C7 are probably mediated by a receptor. Nowadays, various kinds of oxysterol binding molecules have been identified but nothing is known about their ability to trigger an apoptotic signal. Among these molecules, some studies revealed that several oxysterols can bind to the antiestrogen-binding site (AEBS) located predominantly in the microsomal fraction, and ubiquitously distributed in animal and human tissues.79 However, as the affinities of oxysterols oxidized at C7 for the AEBS are in the following order (7α-hydroxycholesterol>7-ketocholesterol>7β-hydroxycholesterol), we can suppose that this receptor does not deliver a death signal.80 Another putative receptor of oxysterols is the oxysterol-binding protein (OSBP) present in the cytosol of many cell types,81 whose gene has been mapped to the long arm of human chromosome 11 and the proximal end of mouse chromosome 19.82 However, 7α-hydroxycholesterol, 7β-hydroxycholesterol, and 7-ketocholesterol are weakly recognized by this receptor, contrary to the cholesterol derivatives hydroxylated on the side chain.83 Therefore, OSBP is probably not involved in the apoptotic process induced by 7β-hydroxycholesterol and 7-ketocholesterol. As it was shown that sterols derived from cholesterol by the introduction of a second functional group (hydroxyl or ketone) in the 6, 7, 15, 20, 22, 24, or 25 positions are potent inhibitors of 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMG-CoA reductase),84 and that some statins (synthetic inhibitors of HMG-CoA reductase)85 can induce an apoptotic mode of cell death,86,87 the different potency of oxysterols oxidized at C7 to induce apoptosis might also depend on their ability to inhibit HMG-CoA reductase. Indeed, HMG-CoA reductase activity has major consequences on the transmission of growth factor-dependent survival signals by contributing to the synthesis of farnesylpyrophosphate, which is not only a cholesterol precursor but also an intermediate in different isoprenoid-dependent metabolic pathways such as dolichol and ubiquinone synthesis, but also protein farnesylation (Ras) and protein geranyl-geranylation (Rho, Rab), which seem essential for the delivery of anti-apoptotic signals through pathways involving the activation of phosphatidylinositol-3-kinase (PI(3)K) and of its downstream effector, the serine / threonine kinase Akt.87,88,89,90,91 So, if we suppose that 7α-hydroxycholesterol, contrary to 7β-hydroxycholesterol and 7-ketocholesterol, is not a potent inhibitor of HMG-CoA reductase, it could subsequently not induce a suppression of Ras farnesylation and of Rho geranyl-geranylation. In turn, according to this hypothesis, 7α-hydroxycholesterol could not lead to the suppression of phosphorylation through the pathway involving the activation of PI(3)K/Akt. As a consequence, numerous molecules such as Bad or Bax would not be dephosphorylated and could not interact with Bcl-2 at the mitochondrial level92,93,94 to induce a loss of transmembrane mitochondrial potential (Δψm), cytosolic release of cytochrome c, activation of caspases-9 and -3, and PARP degradation. More recently, new receptors (LXRs) have been identified, and two of them, the closely related transcription factors LXRα and LXRβ, are activated by hydroxylated cholesterol as ligands,95,96 but their roles in oxysterol-induced cell death remain to be defined. However, it seems that these receptors are involved in the activation of SREB-1, which results from the cleavage of sterol regulatory element binding proteins (SREBP) under the action of CPP32/SCA-1,97,98 reinforcing the hypothesis that molecules implied in cholesterol biosynthesis and lipoprotein uptake are probably implied in the program of apoptosis.
In conclusion, our data underline the complexity of the mechanisms of oxysterols oxidized at C7 (7α-hydroxycholesterol, 7β-hydroxycholesterol and 7-ketocholesterol), and they demonstrate the following points: (1) induction of apoptosis depends on the radical (hydroxyl or keto) present at C7 as well as on the α or β hydroxyl radical position; thus, 7β-hydroxycholesterol and 7-ketocholestrol are potent inducers of apoptosis but 7α-hydroxycholesterol is not; (2) the mitochondrial signaling pathway involving loss of the mitochondrial transmembrane potential (Δψm), release of cytochrome c, activation of caspase-9 and -3, and PARP degradation is a common feature of 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis; (3) C16 : 0 and C24 : 1 ceramide species are generated under treatment with 7β-hydroxycholesterol and 7-ketocholesterol (but not with 7α-hydroxycholesterol); (4) Z-VAD-fmk counteracts 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis but does not reduce C16 : 0 and C24 : 1 ceramide species generation; (5) the inhibition of significant amounts of C16 : 0 and C24 : 1 ceramide species by fumonisin B1 is not associated with an impairment of 7β-hydroxycholesterol and of 7-ketocholesterol-induced apoptosis. Thus, the present comparative study performed on oxysterols oxidized at C7 brings new insights on the metabolic pathways involved in oxysterol-induced apoptosis, and further understanding of the cytotoxicity of oxysterols, which probably play important roles in atherosclerosis,46 might allow for the development of new systemic therapies for this disease.
Materials and Methods
Cells
U937 cells were grown in suspension in culture medium consisting of RPMI 1640 medium (Gibco, Eragny, France), 2 mM L-glutamine (Gibco), antibiotics (100 U/ml penicillin, 100 mg/ml streptomycin) (Gibco) and supplemented with 10% (v/v) heat-inactived fetal calf serum (Gibco). The cells were seeded at 5×105 per ml of culture medium, passaged twice a week, and incubated at 37°C under a 5% CO2/ 95% air atmosphere.
Cell treatments
The purity of 7α-, 7β-hydroxycholesterol (purchased from Steraloids, Wilton, USA), and 7-ketocholesterol (Sigma-Aldrich, L'Isles d'Abeau-Chesnes, France) was determined to be 100% by gaseous phase chromatography-mass spectrometry. For all experiments, initial solutions of 7α-, 7β-hydroxycholesterol and 7-ketocholesterol were prepared extemporaneously at a concentration of 800 μg/ml as previously described.15 The initial oxysterol solutions were prepared dissolving 800 μg of oxysterol in 50 μl of absolute ethanol, 950 μl of culture medium were further added, and the solution was sonicated. 7α-, 7β-hydroxycholesterol and 7-ketocholesterol were studied at final concentrations of 5, 10, 20, 40, and 80 μg/ml (12.5–200 μM) by adding 6.25, 12.5, 25, 50, or 100 μl of the initial oxysterol solution (800 μg/ml) per ml of culture medium. The oxysterol concentrations used are in the range of levels measured in the plasma of hypercholesterolemic patients52 and of cholesterol-rich fed rabbits.53 Under these experimental conditions, the ethanol concentration in the culture medium did not exceed 0.5%. In those conditions, ethanol had no effect on cell growth nor on cell viability measured with propidium iodide, and did not increase the proportion of apoptotic cells when compared to untreated cells.12 All oxysterols were introduced in the culture medium at the beginning of the culture. N-benzyloxycarbonyl-valinyl-alaninyl-aspartyl fluoromethylketone (Z-VAD-fmk) (Bachem Biochimie, Voisins-le-Bretonneux, France), which is a broad spectrum cell-permeable caspase inhibitor,30 was dissolved in dimethylsulfoxide (Sigma) to a concentration of 2 mM, and added to the culture medium at a final concentration of 100 μM. As for fumonisin B1 (Sigma), a specific inhibitor of ceramide synthase,31 it was dissolved in sterile saline (0.9%) to a concentration of 500 μM and used at a final concentration of 100 μM. When the cells were simultaneously treated with an oxysterol and with Z-VAD-fmk (or with fumonisin B1), the latter compound was introduced in the culture medium 30 min before the oxysterol.
Cell counting
Cell counting was performed with a hemacytometer under a Laborlux IX 70 inverted phase contrast microscope (Olympus, Tokyo, Japan) on cells seeded in 6-well plates (Falcon/Becton Dickinson, Plymouth, UK) at a concentration of 1.5×106 cells in 3 ml of culture medium per well.
Identification and quantification of apoptotic cells after nuclei staining with Hoechst 33342
Nuclear morphology of control and treated cells was studied by fluorescence microscopy after staining with Hoechst 33342 (λEx max: 346 nm, λEm max: 420 nm) (Sigma). Apoptotic cells were characterized by nuclear condensation of chromatin and/or nuclear fragmentation.23 Hoechst 33342 was freshly prepared in distilled water at 1 mg/ml and used at the final concentration of 10 μg/ml. After 30 min of incubation at 37°C, cells were washed twice in cold PBS and resuspended to a concentration of 106 cells/ml in PBS containing 1% (w/v) paraformaldehyde. Cell deposits of about 40 000 cells were applied to glass slides by cytocentrifugation for 5 min at 1000 r.p.m. with a cytospin 2 (Shandon, Cheshire, UK), mounted in Fluoprep (Biomérieux, Marcy l'Etoile, France), and stored in the dark at 4°C. The morphological aspect of cell nuclei was observed with an Axioskop light microscope (Zeiss, Jena, Germany) by using UV light excitation. Images were acquired with an image analysis system (Biocom, Les Ulis, France), and 300 cells were examined for each sample.
Transmission electron microscopy
For transmission electron microscopy, 20×106 cells were fixed for 1 h with 2% glutaraldehyde prepared in a 0.1 M cacodylate buffer (pH 7.4), postfixed in osmium tetroxide, deshydrated using a graded ethanol series, and embedded in Epon. Sections were stained with uranyl acetate and lead citrate, and were examined with a H 600 electron microscope (Hitachi, Tokyo, Japan).
Determination of cell permeability with propidium iodide
Cell permeability was determined after staining with the phenanthrene dye propidium iodide (λEx max: 540 nm, λEm max: 625 nm) (Sigma), which enters only dead cells.32 A stock solution of propidium iodide was prepared in phosphate-buffered saline (PBS) at the concentration of 10 μg/ml, and kept in the dark at room temperature. Propidium iodide was used at a final concentration of 4 μg/ml in a cell suspension adjusted to 106 cells/ml. Fluorescence was immediately quantified by flow cytometry in 10 000 cells on a logarithmic scale of fluorescence of four decades of log on a FACScan flow cytometer (Becton Dickinson, Moutain View, CA, USA) at excitation and emission wavelengths of 488 and 585/42 nm, respectively.
Characterization and quantification of oxysterols by capillary gas chomatography and mass spectrometry
U937 cells incubated for 24 h in their culture medium in the absence or presence of 7α-, 7β-hydroxycholesterol or 7-ketocholesterol used at concentrations of 5, 10, 20, 40 and 80 μg/ml for 24 h were collected by centrifugation (1000×g, 4°C, 10 min), washed twice in PBS, and enumerated with a hemacytometer. Total lipids from cells were further extracted by the methods of Folch et al.99 The extract was saponified at 60°C for 60 min with potassium hydroxide (13.2 g/l), followed by esterification at 60°C for 60 min with boron trifluoride (BF3)-methanol to yield fatty acid methyl esters, and oxysterols were analyzed by capillary gas chromatography24 on a 30×0.25 mm Hewlett-Packard HP5MS column with the use of a Hewlett-Packard 6890 gas chromatograph attached to a 5973 A mass detector (Hewlett-Packard, Palo Alto, CA, USA). Heptadecanoic acid (17 : 0) was added as an internal standard to each sample before extraction and the oxysterol content was determined from the ratio of the peak area of the sample to the peak area of the internal standard. Data are represented as the quantity of 7α-, 7β-hydroxycholesterol and 7-ketocholesterol in μg per 106 cells.
Characterization and quantification of ceramide by electrospray ionization mass spectrometry
Ceramide changes were studied in U937 cells cultured for 30 min, 1, 6, 12, 18, 24, and 30 h in the absence or in the presence of 7α-hydroxycholesterol (80 μg/ml), 7β-hydroxycholesterol (20 μg/ml), or 7-ketocholesterol (40 μg/ml). In addition, the effects of Z-VAD-fmk (100 μM) and of fumonisin B1 (100 μM) were investigated at 24 h of treatment in the presence of 7β-hydroxycholesterol (20 μg/ml), or 7-ketocholesterol (40 μg/ml). At the end of the incubation time, cells were collected by centrifugation (1000×g, 4°C, 10 min), washed twice in culture medium without fetal calf serum, and enumerated with a hemacytometer. Cells were further lysed in CH3OH/H2O/12 M HCl 95 : 5 : 0.5 (v/v), and sonicated. Lipids were extracted with CHCl3 according to the method described by Gu et al.,100 and N-acetylsphingosine (ICN, Costa Mesa, CA, USA) was used as internal standard for ceramides. Lipids were eluted by liquid chromatography on a 2.0×250 mm Hypersil ODS column (Hewlett-Packard) using 5 mM amonium acetate in CH3OH as eluant at a flow rate of 0.5 ml/min, and ceramides were characterized by positive ion electrospray ionization mass spectrometry performed on a MSD 1100 mass spectrometer (Hewlett-Packard). The orifice voltage was operated at 80 V, the capillary voltage at 3.5 kV, and the drying gas flow at 8 l/min. Ions at m/z 324, 520, and 630 were used to measure N-acetylsphingosine, palmitoylsphingosine, and nervonoylsphingosine. Palmitoylsphingosine, and nervonoylsphingosine levels were determined by comparison with a standard curve produced using known amounts of palmitoylsphingosine (Sigma), and nervonoylsphingosine (Sigma). Data are represented as the quantity of ceramide (C16 : 0 and C24 : 1) in ng per 106 cells.
Flow cytometric measurement of the mitochondrial transmembrane potential (Δψm) with the cationic lipophilic dye DiOC6(3)
The mitochondrial transmembrane potential (Δψm) was measured with 3, 3′-dihexyloxacarbocyanine iodide (DiOC6(3): λEx max 484 nm, λEx max 501 nm) (Molecular Probes, Inc., Eugene, OR, USA) used at 40 nM final concentration in U937 cells cultured for 18, 24, and 30 h in the absence or in the presence of 7α-hydroxycholesterol (80 μg/ml), 7β-hydroxycholesterol (20 μg/ml), or 7-ketocholesterol (40 μg/ml). This cyanine dye, which accumulates in the mitochondrial matrix under the influence of the Δψm,101 was initially prepared at 1 mM in DMSO (Sigma), and further diluted in distilled water in order to obtain a 20 μM intermediate solution. To obtain a 40 nM final concentration, 2 μl of this intermediate solution were added to cell suspensions adjusted to 2×106 cells/ml, and after 15 min of incubation at 37°C, DiOC6(3) mitochondrial transmembrane potential related fluorescence was immediately recorded by flow cytometry with a FACscan flow cytometer (Becton Dickinson). The green fluorescence was collected through a 524/44 nm band pass filter, and the fluorescent signals were measured on a logarithmic scale of four decades of log. For each sample, 10 000 cells were acquired and the data were analyzed with the LYSYS I software (Becton Dickinson).
Protein extraction and Western blot analysis
Degradation of caspase-9 and-3, cytochrome c release into the cytosol, and cleavage of the poly(ADP-ribose) polymerase (PARP) were investigated by Western blot analysis of U937 cells incubated for 18, 24 and 30 h in their culture medium in the absence or presence of 7α-hydroxycholesterol (80 μg/ml), 7β-hydroxycholesterol (20 μg/ml) or 7-ketocholesterol (40 μg/ml). Analyses of the degradation of caspase -9 and -3 were performed in Ripa buffer (150 mM NaCl, 50 mM TrisHCl pH8,0, 0.1% SDS, 0.5% Na desoxycholate) extracts. To this end, cells were harvested at the end of the treatment and washed one time with cold PBS. Cells were further resuspended in the lysis buffer consisting of Ripa buffer containing a mixture of protease inhibitors (0.1 mM phenylmethylsulfonylfluoride, 2.5 μg/l aprotinin, 10 μg/ml pepstatine A, 2.5 μg/ml trypsin inhibitors, and 5 μg/ml leupeptin). After 30 min of incubation at 4°C in the lysis buffer, cell debris was eliminated by centrifugation for 15 min at 10 000×g and the supernatant was collected.
For analysis of cytochrome c release into the cytosol, cells were resuspended in buffer A (20 mM HEPES-KOH pH 7.5, 10 mM KCl, 1.5 mM MgCl2, 1 mM EDTA-Na, 1 mM EGTA-Na, 1 mM dithiothreitol) containing 250 mM sucrose and a mixture of protease inhibitors (1 mM phenylmethylsulfonylfluoride, 1% aprotinin, 1 mM leupeptin, 1 μg/ml pepstatin A, and 1 μg/ml chymostatin). The cells were homogenized by successive passages through a 25 G fine needle. Unbroken cells, large plasma membrane pieces, and nuclei were removed by centrifuging the homogenates for 10 min at 1000×g and at 4°C. The resulting supernatant was subjected to 10 000×g at 4°C for 20 min, and the supernatant obtained was centrifuged again for 1 h at 100 000×g and at 4°C to generate cytosol.
For analysis of PARP, total cellular extracts were needed. The cell pellets were resuspended and incubated for 30 min at 4°C in Laemnli's lysis buffer (1% SDS, 1 mM sodium-vanadate, 10 mM Tris-HCl) in the presence of protease inhibitors (1 mM phenylmethylsulfonylfluoride, 1% aprotinin, 1 mM leupeptin, 1 μg/ml pepstatin A, and 1 μg/ml chymostatin), and the cells were homogenized by successive passages through a 25 G fine needle.
The protein concentrations were measured by using bicinchoninic acid reagent (Pierce, Rockford, IL, USA) according to the method of Smith et al.102 Thirty to 50 μg of protein were incubated in loading buffer (125 mM Tris-HCl, pH 6.8, 10% β-mercaptoethanol, 4.6% SDS, 20% glycerol, 5 M urea and 0.003% bromophenol blue), boiled for 3 min, separated by SDS-polyacrylamide gel electrophoresis and electroblotted onto polyvinylidine difluoride membrane (Biorad, Ivry sur Seine, France). After blocking nonspecific binding sites overnight by 5% nonfat milk in TPBS (PBS, Tween 20 0.1%), the membranes were incubated for 2 h at room temperature with various primary antibodies: a mouse monoclonal antibody directed against procaspase-9 used at 2 μg/ml (Pharmingen), a rabbit polyclonal antibody directed against caspase-3 used at 2 μg/ml (Pharmingen, San Diego, CA, USA), a mouse monoclonal antibody directed against cytochrome c also used at 2 μg/ml (Pharmingen), and a mouse monoclonal antibody directed against the PARP diluted 1/2000 (Boerhinger); all antibodies were diluted in TPBS. After two washes in TPBS, the membranes were incubated with horseradish peroxidase-conjugated goat anti-mouse (Dako, Coppenhagen, Denmark), or anti-rabbit antibody (Biorad) for 1 h at room temperature and washed twice in TPBS. Autoradiography of the immunoblots was performed using an enhanced chemoluminescence detection kit (Amersham, Les Ulis, France). Autoradiograms were digitized using an image analysis system (Biocom). Quantitative analysis of cytochrome c release was performed using autoradiography with a Biocom image analysis system and normalized against a cross reacting protein of around 65 kD present in the cytosolic extract;62 in these conditions, cytochrome c release was estimated with the Visiolab software (Biocom) by the ratio [cytochrome c]/[65 kD protein]. Each experiment was repeated three times with identical results.
Fluorogenic assay for caspase-3 protease activity
To assess caspase-3 activity, the cleavage of Ac-DEVD-7-amino-4-methylcoumarin (Ac-DEVD-AMC) was measured according to the manufacturer procedure with the Quanti PakTM kit (BIOMOL Research Laboratories, Plymouth Meeting, PA, USA) in U937 cells incubated for 18, 24 and 30 h in their culture medium in the absence or presence of 7α-hydroxycholesterol (80 μg/ml), 7β-hydroxycholesterol (20 μg/ml) or 7-ketocholesterol (40 μg/ml), with or without ZVAD-fmk (100 μM). Briefly, at the end of the incubation time, cells were counted, harvested by centrifugation (1000×g, 4°C, 10 min), and resuspended to 5×107 cells per ml of ice-cold lysis buffer (50 mM(N-[(2-hydroxyethyl] piperazine-N′-[2-ethanesulfonic acid]) (HEPES), pH 7.4, 0.1% (3[(3-chloramidopropyl) dimethylammonio]-1-propane-sulfonate) (CHAPS), 1 mM dithiothreitol (DTT), 0.1 mM ethylenediaminetetraacetic acid (EDTA), 1% triton X-100). After 10 min of incubation in an ice bath, cells were centrifuged (10 000×g, 4°C, 10 min), and the supernatant (cytosolic extract) was collected. For each assay, the cytosolic extract (10 μl) was mixed with 80 μl of assay buffer (50 mM HEPES, pH 7.4, 100 mM NaCl, 0.1% CHAPS, 10 mM DTT, 1 mM EDTA, 10% glycerol), and with 10 μl of substrate solution (2 mM Ac-DEVD-AMC). The fluorescence of the liberated AMC was measured at room temperature at 2 min intervals for 40 min using a Victor2 microspectrofluorometer (Wallac, Turku, Finland) (λEx 484 nm, λEm 501 nm) in order to determine the specific activity of the assay. In these conditions, three independent experiments were performed in triplicate.
Statistical analysis
Statistical analyses were performed with SYSTAT software (Evanston, III.) using a one way and a two way analysis of variance.
Abbreviations
- DiOC6(3):
-
3,3′-dihexyloxacarbocyanine iodide
- PARP:
-
poly(ADP-ribose) polymerase
- PI:
-
propidium iodide
- Z-VAD-fmk:
-
N-benzyloxycarbonyl-valinyl-alaninyl-aspartyl fluoromethylketone
- HMG-CoA reductase:
-
3-hydroxy-3-methylglutaryl coenzyme A reductase
References
Smith L . 1996 Review of progress in sterol oxidations: 1987-1995 Lipids 31: 453–457
Parish EJ, Nanduri VBB, Kohl HH and Taylor FR . 1986 Oxysterols: chemical synthesis, biosynthesis and biological activities Lipids 21: 27–30
Smith LL . 1987 Cholesterol autoxidation Chem. Phys. Lipids 44: 87–125
Maraschiello C, Esteve E and Garcia Regueiro JA . 1998 Cholesterol oxidation in meat from chicken fed alpha-tocopherol- and beta-carotene-supplemented diets with different insaturation grades Lipids 33: 705–713
Przygonski K, Jelen H and Wasowicz E . 2000 Determination of cholesterol oxidation products in milk powder and infant formulas by gas chromatography and mass spectrometry Nahrung 44: 122–125
Guardiola F, Codony R, Addis PB, Rafecas M and Boatella J . 1996 Biological effects of oxysterols: current status Food Chem. Toxicol. 34: 193–211
Morel DW and Lin CY . 1996 Cellular biochemistry of oxysterols derived from the diet or oxidation in vivo Nutr. Biochem. 7: 495–506
Bakos JT, Johnson BH and Thompson EB . 1993 Oxysterol-induced cell death in human leukemic T-cells correlates with oxysterol binding protein occupancy and is independent of glucocorticoid-induced apoptosis J. Steroid. Biochem. Mol. Biol. 46: 415–426
Lizard G, Lemaire S, Monier S, Gueldry S, Néel D and Gambert P . 1997 Induction of apoptosis and of interleukin-1β secretion by 7β-hydroxycholesterol and 7-ketocholesterol: partial inhibition by Bcl-2 overexpression FEBS Lett. 419: 276–280
Peng SK, Tham P, Taylor CB and Mikkelson B . 1979 Cytotoxicity of oxidation derivatives of cholesterol on cultured aortic smooth muscle cells and their effect on cholesterol biosynthesis Am. J. Clin. Nutr. 32: 1033–1042
Lizard G, Deckert V, Dubrez L, Moisant M, Gambert P and Lagrost L . 1996 Induction of apoptosis in endothelial cells treated with cholesterol oxides Am. J. Pathol. 148: 1625–1638
Lizard G, Monier S, Cordelet C, Gesquière L, Deckert V, Gueldry S, Lagrost L and Gambert P . 1999 Characterization and comparison of the mode of cell death, apoptosis versus necrosis, induced by 7β-hydroxycholesterol and 7-ketocholesterol in the cells of the vascular wall Arterioscl. Thromb. Vasc. Biol. 19: 1190–1200
Nishio E and Watanabe Y . 1996 Oxysterols induced apoptosis in cultured smooth muscle cells through CPP32 protease activation and bcl-2 downregulation Biochem. Biophys. Res. Comm. 226: 928–934
Aupeix K, Weltin D, Meija JE, Christ M, Marchal J, Freyssinet JM and Bischoff P . 1995 Oxysterol-induced apoptosis in human monocytic cell lines Immunobiology 194: 415–428
Lizard G, Gueldry S, Sordet O, Monier S, Athias A, Miguet C, Besséde G, Lemaire S, Solary E and Gambert P . 1998 Glutathione is implied in the control of 7-ketocholesterol-induced apoptosis, which is associated with radical oxygen species production FASEB J. 12: 1651–1663
Lemaire S, Lizard G, Monier S, Miguet C, Gueldry S, Volot F, Gambert P and Néel D . 1998 Different patterns of IL-1β secretion, adhesion molecule expression and apoptosis induction in human endothelial cells treated with 7α-, 7β-hydroxycholesterol, or 7-ketocholesterol FEBS Lett. 440: 434–439
Brown AJ and Jessup W . 1999 Oxysterols and atherosclerosis Atherosclerosis 142: 1–28
Berliner JA and Heinecke JW . 1996 The role of oxidized lipoproteins in atherogenesis Free Radical Biol. Med. 20: 707–727
Harada-Shiba M, Kinoshita M, Kamido H and Shimokado K . 1998 Oxidized low density lipoprotein induces apoptosis in cultured human umbilical vein endothelial cells by common unique mechanisms J. Biol. Chem. 273: 9681–9687
Addis PB . 1986 Occurrence of lipid oxidation products in foods Food Chem. Toxicol. 24: 1021–1030
Kitanaka C and Kuchino Y . 1999 Caspase-independent programmed cell death with necrotic morphology Cell Death Differ. 6: 508–515
Vaux DL . 1999 Caspases and apoptosis-biology and terminology Cell Death Differ. 6: 493–494
Lizard G, Fournel S, Genestier L, Dhedin N, Chaput C, Flacher M, Mutin M, Panaye M and Revillard JP . 1995 Kinetics of plasma membrane and mitochondrial alterations in cells undergoing apoptosis Cytometry 21: 275–283
Park SW and Addis PB . 1985 Capillary column gas-liquid chromatographic resolution of oxidized cholesterol derivatives Anal. Biochem. 149: 275–283
Petit PX, Lecoeur H, Zorn E, Dauget C, Mignotte B and Gougeon ML . 1995 Alterations in mitochondrial structure and function are early events of dexamethasone-induced thymocyte apoptosis J. Cell. Biol. 130: 157–167
Dubrez L, Savoy I, Hamman A and Solary E . 1996 Pivotal role of DEVD-sensitive step in eteoposide-induced and Fas-mediated apoptotic pathways EMBO J. 15: 5504–5512
Hannun YA and Obeid LM . 1995 Ceramide: an intracellular signal for apoptosis Trends Biol. Sci. 20: 73–77
Jaffrezou JP, Levade T, Bettaieb A, Andrieu N, Bezombes C, Maestre N, Vermeersch S, Rousse A and Laurent G . 1996 Daunorubicin-induced apoptosis: triggering of ceramide generation through sphingomyelin hydrolysis EMBO J. 15: 2417–2424
Bettaieb A, Record M, Come MG, Bras AC, Chap H, Laurent G and Jaffrezou JP . 1996 Opposite effects of tumor necrosis factor alpha on the sphingomyelin-ceramide pathway in two myeloid leukemia cell lines: role of transverse sphingomyelin distribution in the plasma membrane Blood 15: 1465–1472
Rodriguez I, Matsuura K, Ody C, Nagata S and Vassalli P . 1996 Systemic injection of a tripeptide inhibits the intracellular activation of CPP32-like proteases in vivo and fully protects mice against Fas-mediated fulminant liver destruction and death J. Exp. Med. 184: 2067–2072
Wang E, Norred WP, Bacon CW, Riley RT and Merril AH . 1991 Inhibition of sphingolipid biosynthesis by fumonisins. Implications for diseases associated with Fusarium noniliforme J. Biol. Chem. 5: 14486–14490
Yeh CG, Hsi B and Faulk WP . 1981 Propidium iodide as a nuclear marker in immunofluorescence. II. Use with cellular identification and viability studies J. Immunol. Methods 43: 269–275
Green D and Kroemer G . 1998 The central executioners of apoptosis: caspases or mitochondria? Trends Cell Biol. 8: 267–271
Sun XM, MacFarlane M, Zhuang J, Wolf BB, Green DR and Cohen GM . 1999 Distinct caspase cascade are initiated in receptor-mediated and chemical-induced apoptosis J. Biol. Chem. 274: 5053–5060
Susin AS, Zamzami N and Kroemer G . 1998 Mitochondria as regulators of apoptosis: doubt no more Biochim. Biophys. Acta 1366: 151–165
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES and Wang X . 1997 Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade Cell 91: 479–489
De Murcia G and Menissier De Murcia J . 1994 Poly(ADP-ribose)polymerase: a molecular nick-sensor Trends Biochem. Sci. 19: 172–176
Perry DK and Hannun YA . 1998 The role of ceramide in cell signaling Biochim. Biophys. Acta 1436: 233–243
Hannun YA . 1996 Functions of ceramide in coordinating cellular responses to stress Science 274: 1855–1859
Watts JD, Gu M, Patterson SD, Aebersold R and Polverino AJ . 1999 On the complexities of ceramide changes in cells undergoing apoptosis: lack of evidence for a second messenger funtion in apoptotic induction Cell Death Differ. 6: 105–114
Jänicke RU, Ng P, Sprengart ML and Porter AG . 1998 Caspase-3 is required for cleavage of α-fodrin but dispensable for cleavage of other death substrates in apoptosis J. Biol. Chem 273: 15540–15545
Porter AG and Jänicke RU . 1999 Emerging roles of caspases-3 in apoptosis Cell Death Differ. 6: 99–104
Bose R, Verheil M, Haimovitz-Friedman A, Scotto K, Fuks Z and Kolesnick R . 1995 Ceramide synthase mediates daunorubicin-induced apoptosis: an alternative mechanism for generating death signals Cell 82: 405–414
Samali A, Zhivotovsky, Jones D, Nagata S and Orrenius S . 1999 Apoptosis: cell death defined by caspase activation Cell Death Differ. 6: 495–496
Lutjohann D, Papassotiropoulos A, Bjorkhem I, Locatelli S, Bagli M, Oehring RD, Schlegel U, Jessen F, Rao ML, Von Bergmann K and Heun R . 2000 Plasma 24S-hydroxycholesterol (cerebrosterol) is increased in Alzheimer and vascular demented patients J. Lipid Res. 41: 195–198
Best PJM, Hasdai D, Sangiorgi G, Schwartz RS, Holmes DR, Simari RD and Leman A . 1998 Apoptosis. Basic concepts and implications in coronary artery disease Arterioscler. Thromb. Vasc. Biol. 19: 14–22
Schroepfer GJ . 2000 Oxysterols: modulators of cholesterol metabolism and other processes Physiol. Rev. 80: 362–554
Bischoff PL, Holl V, Coelho D, Dufour P, Weltin D and Luu B . 2000 Apoptosis at the interface of immunosuppressive and anticancer activities: the examples of two classes of chemical inducers, oxysterols and alkylating agents Curr. Med. Chem. 7: 693–713
Chang JY, Chavis JA, Liu LZ and Drew PD . 1998 Cholesterol oxides induce programmed cell death in microglial cells Biochem. Biophys. Res. Commun. 249: 817–821
Chang JY and Liu LZ . 1998 Neurotoxicity of cholesterol oxides on cultured cerebellar granule cells Neurochem. Int. 32: 317–323
Kolsch H, Lutjohann D, Tulke A, Bjorkhem I and Rao ML . 1999 The neurotoxic effects of 24-hydroxycholesterol on SH-5Y human neuroblastoma cells Brain Res. 818: 171–175
Gray MFT, Lawrie TDV and Brooks CJW . 1971 Isolation and identification of cholesterol α-oxide and other main sterols in human serum Lipids 6: 836–843
Vine DF, Mamo JCL, Beilin LJ, Mori TA and Croft KD . 1998 Dietary oxysterols are incorporated in plasma triglyceride-rich lipoproteins, increase their susceptibility to oxidation and increase aortic cholesterol concentration of rabbits J. Lipid Res. 39: 1995–2004
Christ M, Luu B, Meija JE, Moosbrugger I and Bischoff P . 1993 Apoptosis induced by oxysterols in murine lymphoma cells and in normal thymocytes Immunology 78: 455–460
O'Callaghan YC, Woods JA and O'Brien NM . 1999 Oxysterol-induced cell death in U937 and HepG2 cells at reduced and normal serum concentrations Eur. J. Nutr. 38: 255–262
Luu B . 1995 New insights on cholesterol and its oxidation compounds C. R. Soc. Biol. 189: 827–837
Hietter H, Bischoff P, Beck JP, Ourisson G and Luu B . 1986 Comprative effects of 7β-hydroxycholesterol towards murine lymphoma, lymphoblasts and lymphocytes: selective cytotoxicity and blastogenesis inhibition. Cancer Biochem Biophys. 9: 75–83
Verhagen JC, ter Braake P, Teunissen J, van Ginkel G and Sevanian A . 1996 Physical effects of biologically formed cholesterol oxidation products on lipid membranes investigated with fluorescence depolarization spectroscopy and electron spin resonance J. Lipid Res. 37: 1488–1502
Bonnefoy-Bérard N, Genestier L, Flacher M, Rouault JP, Lizard G, Mutin M and Revillard JP . 1994 Apoptosis induced by polyclonal antilymphocyte globulins in human B-cell lines Blood 83: 1051–1059
Mehmet H . 2000 Caspases find new place to hide Nature 403: 29–30
Nakagawa T, Zhu H, Morishima N, Li E, Xu J, Yanker BA and Yuan J . 2000 Caspase-12 mediates endoplasmic-reticulum-specific apoptosis and cytotoxicity by amyloid-β Nature 403: 98–103
Lizard G, Miguet C, Besséde G, Monier S, Gueldry S, Néel D and Gambert P . 2000 Impairment with various antioxidants of the loss of mitochondrial transmembrane potential and of the cytosolic release of cytochrome c occuring during 7-ketocholesterol-induced apoptosis Free Radical. Biol. Med. 28: 743–753
Fiers W, Rudi B, Declercq W and Vandenabeele P . 1999 More than one way to die: apoptosis, necrosis and reactive oxygen damage Oncogene 18: 7719–7730
Slee EA, Adrain C and Martin SJ . 1999 Serial killers: ordering caspase activation events in apoptosis Cell Death Differ. 6: 1067–1074
Cai J, Yang J and Jones DP . 1998 Mitochondrial control of apoptosis: the role of cytochrome c Biochim. Biophys. Acta 1366: 139–149
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES and Wang X . 1997 Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade Cell 91: 479–489
Arora AS, Jones BJ, Patel TC, Bronk SF and Gores GJ . 1997 Ceramides induces hepatocyte cell death through disruption of mitochondrial function in the rat Hepatology 25: 958–963
Ghafourifar P, Klein SD, Schucht O, Schenk U, Pruschy M, Rocha S and Richter C . 1999 Ceramide induces cytochrome c release from isolated mitochondria J. Biol. Chem. 274: 6080–6084
Cuvillier O, Edsall L and Spiegel S . 2000 Involvement of spingosine in mitochondria-dependent Fas-induced apoptosis of type II Jurkat T cells J. Biol. Chem. 275: 15691–15700
Witty JP, Brigham JT and Johnson AL . 1996 Induction of apoptotic cell death in hen granulosa cells by ceramides Endocrinology 137: 5269–5277
Suzuki A, Iwasaki M, Kato M and Wagai N . 1997 Sequential operation of ceramide synthesis and ICE cascade in CPT-11-initiated apoptotic death signaling Exp. Cell Res. 233: 41–47
Garzotto M, White-Jones M, Jiang Y, Ehleiter D, Liao WC, Haimovitz-Friedman A, Fuks Z and Kolesnick . 1998 12-O -tetradecanoylphorbol-13-acetate induced apoptosis in LNCaP cells is mediated through ceramide synthase Cancer Res. 58: 2260–2264
Escargueil-Blanc I, Andrieu-Abadie N, Caspar-Bauguil S, Brossmer R, Levade T, Nègre-Salvayre A and Salvayre R . 1998 Apoptosis and activation of the sphingomyelin-ceramide pathway induced by oxidized low density lipoproteins are not causally related in ECV-304 endothelial cells J. Biol. Chem. 273: 27389–27395
Sevanian A, Hodis HN, Hwang J, McLeod LL and Peterson H . 1995 Characterization of endothelial cell injury by cholesterol oxidation products found in oxidized LDL J. Lipid Res. 36: 1971–1986
Cornell R, Grove GL, Rothblat GH and Horwitz AF . 1977 Lipid requirement for cell cycling. The effect of selective inhibition of lipid synthesis Exp. Cell Res. 109: 299–307
Hyun JW, Weltin D, Holl V, Marchal J, Dufour P, Luu B and Bischoff P . 1997 Cytotoxic properties of phosphoglycoconjugated derivative of 7β-hydroxycholesterol upon normal and tumor cells in culture Anticancer Res. 17: 2621–2626
Nickels JT and Broach JR . 1996 Ceramide-activated protein phosphatase mediates ceramide-induced G1 arrest of Sacharomyces cerevisae Genes Dev. 10: 382–394
Jayadev S, Liu B, Bielawska AE, Lee JY, Nazaire F, Pushkareva MY, Obeid LM and Hannun YA . 1995 Role in ceramide in cell cycle arrest J. Biol. Chem. 270: 2047–2052
Lazier CB and Bapat BV . 1988 Antiestrogen binding sites : general and comparative properties J. Steroid Biochem. 31: 665–669
Lin L and Hwang PL . 1991 Antiproliferative effects of oxygenated sterols: positive correlation with binding affinities for the antiestrogen-binding sites Biochim. Biophys. Acta 1082: 177–184
Kandutsch AA, Taylor FR and Shown EP . 1984 Different forms of the oxysterol-binding protein. Binding kinetics and stability J. Biol. Chem. 259: 12388–12397
Levanon D, Hsieh CL, Francke U, Dawson PA, Ridgway ND, Brown MS and Goldstein JL . 1990 cDNA cloning of human oxysterol-binding protein and localization of the gene to human chromosome 11 and mouse chromosome 19 Genomics 7: 65–74
Beseme F, Astruc ME, Defay R and Crastes de Paulet A . 1987 Rat liver cytosol-binding protein. Characterization and comparison with the HTC cell protein FEBS Lett. 210: 97–103
Kandutsch AA and Chen HW . 1974 Inhibition of sterol synthesis in cultured mouse cells by cholesterol derivatives oxygenated in the side chain J. Biol. Chem. 249: 6057–6061
Farnier M and Davignon J . 1998 Current and future treatment of hyperlipidemia: the role of statins Am. J. Cardiol. 82: 3J–10J
Bansal N, Houle AG and Melnykovych G . 1989 Comparison of dexamethasone and lovastatin (mevinolin) as growth inhibitors in cultures of T-cell derived from human acute leukemia lines (CEM) Leuk. Res. 13: 875–882
Guijarro C, Blanco-Colio LM, Ortego M, Alonso C, Ortiz A, Plaza JJ, Diaz C, Hernandez G and Egido J . 1998 3-hydroxy-3-methylglutaryl coenzyme A reductase and isoprenylation inhibitors induce apoptosis of vascular smooth muscle cells in culture Circ. Res. 83: 490–500
Edwards PA and Ericsson J . 1999 Sterols and isoprenoids: signaling molecules derived from the cholesterol biosynthetic pathway Annu. Rev. Biochem. 68: 157–185
Gadbut AP, Wu L, Tang D, Papageorge A, Watson JA and Galper JB . 1997 Induction of the cholesterol metabolic pathway regulates the farnesylation of RAS in embryonic chick heart cells: a new role for RAS in regulating the expression of muscarinic receptors and G proteins EMBO J 16: 7250–7260
Kennedy SG, Wagner AJ, Conzen SD, Jordan J, Bellacosa A, Tsichilis PN and Hay N . 1997 The PI 3-kinase / Akt signaling pathway delivers an anti-apoptotic signal Gene Dev. 11: 701–713
Kauffmann-Zeh A, Rodriguez-Viciana P, Ulrich E, Gilbert C, Coffer P, Downward J and Evan G . 1997 Suppression of c-Myc-induced apoptosis by Ras signalling through PI(3)K and PKB Nature 386: 544–548
Zha J, Harada H, Yang E, Jockel J and Korsmeyer SJ . 1996 Serine phosphorylation of death agonist Bad in response to survival factor results in binding to 14-3-3 not Bcl-xL Cell 87: 619–628
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y and Greenberg ME . 1997 Akt phosphorylation of BAD couples survival signals to the cell-intinsic death machinery Cell 91: 231–241
Kennedy SG, Kandel ES, Cross TK and Hay N . 1999 Akt/protein kinase B inhibits cell death by preventing the release of cytochrome c from mitochondria Mol. Cell. Biol. 19: 5800–5810
Lehmann JM, Kliewer SA, Moore LB, Smith-Oliver TA, Oliver BB, Su JL, Sunseth SS, Winegard DA, Blanchard DE, Spencer TA and Wilson TM . 1997 Activation of the nuclear receptor LXR by oxysterols defines a new hormone response pathway J. Biol. Chem. 272: 3137–3140
Peet DJ, Janowski BA and Mangeldorf DJ . 1998 The LXRs: a new class of oxysterol receptors Curr. Opin. Genet. Dev. 8: 571–575
Pai JT, Brown MS and Goldstein JL . 1996 Purification and cDNA cloning of a second apoptosis-related cysteine protease that cleaves and activates sterol regulatory element binding proteins Proc. Natl. Acad. Sci. USA 93: 5437–5442
Lund EG, Kerr TA, Sakai J, Li WP and Russell DW . 1998 cDNA cloning of mouse and human cholesterol 25-hydroxylases, polytopic membrane proteins that synthetize a potent oxysterol regulator of lipid metabolism J. Biol. Chem. 343: 16–27
Folch J, Lees M and Sloane Stanley GH . 1957 A simple method for the isolation and purification of total lipids from animal tissues J. Biol. Chem. 226: 497–509
Gu M, Kervin JL, Watts JD and Aebersold R . 1997 Ceramides profiling of complex lipid mixtures by electrospray ionization mass spectrometry Anal. Biochem. 244: 347–356
Chen LB . 1988 Mitochondrial membrane potential in living cells Annu. Rev. Cell. Biol. 4: 155–181
Smith PK, Krohn RI, Hermanson GT, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ and Klenk DC . 1985 Measurement of protein using bicinchoninic acid Anal. Biochem. 150: 76–85
Acknowledgements
This work was supported by the Conseil Régional de Bourgogne, the Institut National de la Santé et de la Recherche Médicale (Inserm), the Fondation pour la Recherche Médicale (FRM), and the Fondation de France. We thank Dr Serge Gueldry (Inserm U 498) for his expert statistical analysis, as well as Ms Maryvonne Moisant and Colette Bernard (Laboratoire de Microscopie Préparative Appliquée à la Biologie et à la Médecine) for their technical supports in electron microscopy. We also acknowledge the Centre de Microscopie Appliquée à la Biologie (Université de Bourgogne, Dijon, France) for the observations by transmission electron microscopy. The authors are also indebted to Mr John Ewing for reviewing the English version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by RA Lockshin
Rights and permissions
About this article
Cite this article
Miguet, C., Monier, S., Bettaieb, A. et al. Ceramide generation occurring during 7β-hydroxycholesterol- and 7-ketocholesterol-induced apoptosis is caspase independent and is not required to trigger cell death. Cell Death Differ 8, 83–99 (2001). https://doi.org/10.1038/sj.cdd.4400792
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cdd.4400792
Keywords
This article is cited by
-
Current knowledge on the mechanism of atherosclerosis and pro-atherosclerotic properties of oxysterols
Lipids in Health and Disease (2017)
-
Interactions between cell death induced by statins and 7‐ketocholesterol in rabbit aorta smooth muscle cells
British Journal of Pharmacology (2008)
-
Cytotoxic oxysterols induce caspase-independent myelin figure formation and caspase-dependent polar lipid accumulation
Histochemistry and Cell Biology (2007)
-
Different apoptotic mechanisms are involved in the antiproliferative effects of 7β-hydroxysitosterol and 7β-hydroxycholesterol in human colon cancer cells
Cell Death & Differentiation (2005)
-
Comparison of the cytotoxic, pro-oxidant and pro-inflammatory characteristics of different oxysterols
Cell Biology and Toxicology (2005)