Abstract
CYP2D6 is of great importance for the metabolism of clinically used drugs and about 20–25% of those are metabolised by this enzyme. In addition, the enzyme utilises hydroxytryptamines as endogenous substrates. The polymorphism of the enzyme results in poor, intermediate, efficient or ultrarapid metabolisers (UMs) of CYP2D6 drugs. It is plausible that the UM genotype, where more than one active gene on one allele occurs, is the outcome of selective dietary selection in certain populations in North East Africa. The UM phenotype affects 5.5% of the population in Western Europe. A hypothesis for the evolutionary basis behind selection for CYP2D6 gene duplications is presented in relation to selection for Cyp6 variants in insecticide resistant Drosophila strains. The polymorphism of CYP2D6 significantly affects the pharmacokinetics of about 50% of the drugs in clinical use, which are CYP2D6 substrates. The consequences of the polymorphism at ordinary drug doses can be either adverse drug reactions or no drug response. Examples are presented where CYP2D6 polymorphism affects the efficacy and costs of drug treatment. Predictive CYP2D6 genotyping is estimated by the author to be beneficial for treatment of about 30–40% of CYP2D6 drug substrates, that is, for about 7–10% of all drugs clinically used, although prospective clinical studies are necessary to evaluate the exact benefit of drug selection and dosage based on the CYP2D6 genotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Evans WE, Relling MV . Moving towards individualized medicine with pharmacogenomics. Nature 2004; 429: 464–468.
Ingelman-Sundberg M . Pharmacogenetics: an opportunity for a safer and more efficient pharmacotherapy. J Intern Med 2001; 250: 186–200.
Meyer UA . Pharmacogenetics and adverse drug reactions. Lancet 2000; 356: 1667–1671.
Goldstein DB, Tate SK, Sisodiya SM . Pharmacogenetics goes genomic. Nat Rev Genet 2003; 4: 937–947.
Ingelman-Sundberg M . Pharmacogenetics of cytochrome P450 and its applications in drug therapy: the past, present and future. Trends Pharmacol Sci 2004; 25: 193–200.
Ingelman-Sundberg M . Human drug metabolising cytochrome P450 enzymes: properties and polymorphisms. Naunyn Schmiedebergs Arch Pharmacol 2004; 369: 89–104.
Weinshilboum R . Inheritance and drug response. New Engl J Med 2003; 348: 529–537.
Zanger UM, Raimundo S, Eichelbaum M . Cytochrome P450 2D6: overview and update on pharmacology, genetics, biochemistry. Naunyn Schmiedebergs Arch Pharmacol 2004; 369: 23–37.
de Groot MJ, Bijloo GJ, Martens BJ, van Acker FA, Vermeulen NP . A refined substrate model for human cytochrome P450 2D6. Chem Res Toxicol 1997; 10: 41–48.
Fonne-Pfister R, Meyer UA . Xenobiotic and endobiotic inhibitors of cytochrome P-450dbl function, the target of the debrisoquine/sparteine type polymorphism. Biochem Pharmacol 1988; 37: 3829–3835.
Yu AM, Idle JR, Herraiz T, Kupfer A, Gonzalez FJ . Screening for endogenous substrates reveals that CYP2D6 is a 5-methoxyindolethylamine O-demethylase. Pharmacogenetics 2003; 13: 307–319.
Yu AM, Idle JR, Byrd LG, Krausz KW, Kupfer A, Gonzalez FJ . Regeneration of serotonin from 5-methoxytryptamine by polymorphic human CYP2D6. Pharmacogenetics 2003; 13: 173–181.
Llerena A, Edman G, Cobaleda J, Benitez J, Schalling D, Bertilsson L . Relationship between personality and debrisoquine hydroxylation capacity. Suggestion of an endogenous neuroactive substrate or product of the cytochrome P 4502D6. Acta Psychiatr Scand 1993; 87: 23–28.
Roberts RL, Luty SE, Mulder RT, Joyce PR, Kennedy MA . Association between cytochrome P450 2D6 genotype and harm avoidance. Am J Med Gen Part B 2004; 127B: 90–93.
Pai HV, Kommaddi RP, Chinta SJ, Mori T, Boyd MR, Ravindranath V . A frame shift mutation and alternate splicing in human brain generates a functional form of the pseudogene, cytochrome P4502D7 that demethylates codeine to morphine. J Biol Chem 2004; 279: 27383–27389.
Hammer W, Sjoqvist F . Plasma levels of monomethylated tricyclic antidepressants during treatment with imipramine-like compounds. Life Sci 1967; 6: 1895–1903.
Mahgoub A, Idle JR, Dring LG, Lancaster R, Smith RL . Polymorphic hydroxylation of Debrisoquine in man. Lancet 1977; 2: 584–586.
Eichebaum M, Spannbrucker N, Dengler HJ . A probably genetic defect in the metabolism of sparteine in biological oxidation of nitrogen. In: Gorrow JW (ed). Elsevier North-Holland Biomedical Press: Amsterdam, 1978; pp 113–118.
Eichelbaum M, Spannbrucker N, Dengler HJ . Influence of the defective metabolism of sparteine on its pharmacokinetics. Eur J Clin Pharmacol 1979; 16: 189–194.
Gonzalez FJ, Skoda RC, Kimura S, Umeno M, Zanger UM, Nebert DW et al. Characterization of the common genetic defect in humans deficient in debrisoquine metabolism. Nature 1988; 331: 442–446.
Skoda RC, Gonzalez FJ, Demierre A, Meyer UA . Two mutant alleles of the human cytochrome P-450db1 gene (P450C2D1) associated with genetically deficient metabolism of debrisoquine and other drugs. Proc Natl Acad Sci USA 1988; 85: 5240–5243.
Heim M, Meyer UA . Genotyping of poor metabolisers of debrisoquine by allele-specific PCR amplification. Lancet 1990; 336: 529–532.
Yue QY, Bertilsson L, Dahl-Puustinen ML, Sawe J, Sjoqvist F, Johansson I et al. Disassociation between debrisoquine hydroxylation phenotype and genotype among Chinese. Lancet 1989; 2: 870.
Johansson I, Oscarson M, Yue QY, Bertilsson L, Sjoqvist F, Ingelman-Sundberg M . Genetic analysis of the Chinese cytochrome P4502D locus: characterization of variant CYP2D6 genes present in subjects with diminished capacity for debrisoquine hydroxylation. Mol Pharmacol 1994; 46: 452–459.
Ingelman-Sundberg M . Duplication, multiduplication, and amplification of genes encoding drug-metabolizing enzymes: evolutionary, toxicological, and clinical pharmacological aspects. Drug Metab Rev 1999; 31: 449–459.
Johansson I, Lundqvist E, Bertilsson L, Dahl ML, Sjoqvist F, Ingelman-Sundberg M . Inherited amplification of an active gene in the cytochrome P450 CYP2D locus as a cause of ultrarapid metabolism of debrisoquine. Proc Natl Acad Sci USA 1993; 90: 11825–11829.
Bertilsson L, Dahl ML, Sjoqvist F, Aberg-Wistedt A, Humble M, Johansson I et al. Molecular basis for rational megaprescribing in ultrarapid hydroxylators of debrisoquine. Lancet 1993; 341: 63.
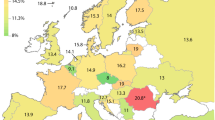
Aklillu E, Persson I, Bertilsson L, Johansson I, Rodrigues F, Ingelman-Sundberg M . Frequent distribution of ultrarapid metabolizers of debrisoquine in an Ethiopian population carrying duplicated and multiduplicated functional CYP2D6 alleles. J Pharmacol Exp Ther 1996; 278: 441–446.
McLellan RA, Oscarson M, Seidegard J, Evans DA, Ingelman-Sundberg M . Frequent occurrence of CYP2D6 gene duplication in Saudi Arabians. Pharmacogenetics 1997; 7: 187–191.
Heim MH, Meyer UA . Evolution of a highly polymorphic human cytochrome P450 gene cluster: CYP2D6. Genomics 1992; 14: 49–58.
Yu A, Kneller BM, Rettie AE, Haining RL . Expression, purification, biochemical characterization, and comparative function of human cytochrome P450 2D6.1, 2D6.2, 2D6.10, and 2D6.17 allelic isoforms. J Pharmacol Exp Ther 2002; 303: 1291–1300.
Masimirembwa C, Persson I, Bertilsson L, Hasler J, Ingelman-Sundberg M . A novel mutant variant of the CYP2D6 gene (CYP2D6*17) common in a black African population: association with diminished debrisoquine hydroxylase activity. Br J Clin Pharmacol 1996; 42: 713–719.
Oscarson M, Hidestrand M, Johansson I, Ingelman-Sundberg M . A combination of mutations in the CYP2D6*17 (CYP2D6Z) allele causes alterations in enzyme function. Mol Pharmacol 1997; 52: 1034–1040.
Wennerholm A, Dandara C, Sayi J, Svensson JO, Abdi YA, Ingelman-Sundberg M et al. The African-specific CYP2D617 allele encodes an enzyme with changed substrate specificity. Clin Pharmacol Ther 2002; 71: 77–88.
Wennerholm A, Johansson I, Hidestrand M, Bertilsson L, Gustafsson LL, Ingelman-Sundberg M . Characterization of the CYP2D6*29 allele commonly present in a black Tanzanian population causing reduced catalytic activity. Pharmacogenetics 2001; 11: 417–427.
Zanger UM, Raimundo S, Eichelbaum M . Cytochrome P450 2D6: overview and update on pharmacology, genetics, biochemistry. Naunyn Schmiedebergs Arch Pharmacol 2004; 369: 23–37.
Nelson DR, Zeldin DC, Hoffman SMG, Maltais LJ, Wain HM, Nebert DW . Comparison of cytochrome P450 (CYP) genes from the mouse and human genomes, including nomenclature recommendations for genes, pseudogenes, and alternative-splice variants. Pharmacogenetics 2004; 14: 1–18.
Danielson PB, MacIntyre RJ, Fogleman JC . Molecular cloning of a family of xenobiotic-inducible drosophilid cytochrome P450s: evidence for involvement in host-plant allelochemical resistance. Proc Natl Acad Sci USA 1997; 94: 10797–10802.
Daborn PJ, Yen JL, Bogwitz MR, Le Goff G, Feil E, Jeffers S et al. A single p450 allele associated with insecticide resistance in Drosophila. Science 2002; 297: 2253–2256.
Amichot M, Tarès S, Brun-Barale A, Arthaud L, Bride J-M, Bergé J-P . Point mutations associated with insecticide resistance in the Drosophila cytochrome P450 Cyp6a2 enable DDT metabolism. Eur J Biochem 2004; 271: 1250–1257.
Aklillu E, Herrlin K, Gustafsson LL, Bertilsson L, Ingelman-Sundberg M . Evidence for environmental influence on CYP2D6-catalysed debrisoquine hydroxylation as demonstrated by phenotyping and genotyping of Ethiopians living in Ethiopia or in Sweden. Pharmacogenetics 2002; 12: 375–383.
Ingelman-Sundberg M, Oscarson M, McLellan RA . Polymorphic human cytochrome P450 enzymes: an opportunity for individualized drug treatment. Trends Pharmacol Sci 1999; 20: 342–349.
Stearns V, Johnson MD, Rae JM, Morocho A, Novielli A, Bhargava P et al. Active tamoxifen metabolite plasma concentrations after coadministration of tamoxifen and the selective serotonin reuptake inhibitor paroxetine. J Natl Cancer Inst 2003; 95: 1758–1764.
Kaiser R, Sezer O, Papies A, Bauer S, Schelenz C, Tremblay PB et al. Patient-tailored antiemetic treatment with 5-hydroxytryptamine type 3 receptor antagonists according to cytochrome P-450 2D6 genotypes. J Clin Oncol 2002; 20: 2805–2811.
Kirchheiner J, Nickchen K, Bauer M, Wong ML, Licinio J, Roots I et al. Pharmacogenetics of antidepressants and antipsychotics: the contribution of allelic variations to the phenotype of drug response. Mol Psychiatry 2004; 9: 442–473.
Chou WH, Yan FX, de Leon J, Barnhill J, Rogers T, Cronin M et al. Extension of a pilot study: impact from the cytochrome P450 2D6 polymorphism on outcome and costs associated with severe mental illness. J Clin Psychopharmacol 2000; 20: 246–251.
Dahl ML . Cytochrome P450 phenotyping/genotyping in patients receiving antipsychotics: useful aid to prescribing? Clin Pharmacokinet 2002; 41: 453–470.
Schillevoort I, de Boer A, van der Weide J, Steijns LS, Roos RA, Jansen PA et al. Antipsychotic-induced extrapyramidal syndromes and cytochrome P450 2D6 genotype: a case–control study. Pharmacogenetics 2002; 12: 235–240.
Tamminga WJ, Wemer J, Oosterhuis B, de Boer A, Vranckx S, Drenth BF et al. Polymorphic drug metabolism (CYP2D6) and utilisation of psychotropic drugs in hospitalised psychiatric patients: a retrospective study. Eur J Clin Pharmacol 2003; 59: 57–64.
Brockmoller J, Kirchheiner J, Schmider J, Walter S, Sachse C, Muller-Oerlinghausen B et al. The impact of the CYP2D6 polymorphism on haloperidol pharmacokinetics and on the outcome of haloperidol treatment. Clin Pharmacol Ther 2002; 72: 438–452.
Kawanishi C, Lundgren S, Agren H, Bertilsson L . Increased incidence of CYP2D6 gene duplication in patients with persistent mood disorders: ultrarapid metabolism of antidepressants as a cause of nonresponse. A pilot study. Eur J Clin Pharmacol 2004; 59: 803–807.
Bradford LD . CYP2D6 allele frequency in European Caucasians, Asians, Africans and their descendants. Pharmacogenomics 2002; 3: 229–243.
Sorensen LB, Sorensen RN, Miners JO, Somogyi AA, Grgurinovich N, Birkett DJ . Polymorphic hydroxylation of perhexiline in vitro. Br J Clin Pharmacol 2003; 55: 635–638.
Barclay ML, Sawyers SM, Begg EJ, Zhang M, Roberts RL, Kennedy MA et al. Correlation of CYP2D6 genotype with perhexiline phenotypic metabolizer status. Pharmacogenetics 2003; 13: 627–632.
Wuttke H, Rau T, Heide R, Bergmann K, Bohm M, Weil J et al. Increased frequency of cytochrome P450 2D6 poor metabolizers among patients with metoprolol-associated adverse effects. Clin Pharmacol Ther 2002; 72: 429–437.
Rendic S . Summary of information on human CYP enzymes: human P450 metabolism data. Drug Metab Rev 2002; 34: 83–448.
Acknowledgements
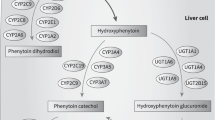
I am indebted to Maria Karlgren for the construction of Figure 1 and to Dr Inger Johansson for critical reading of the manuscript. The research in the author's laboratory is supported by grants from the Swedish Research Council and from NIH (NIGMS 1-R01 GM60548-01A2).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ingelman-Sundberg, M. Genetic polymorphisms of cytochrome P450 2D6 (CYP2D6): clinical consequences, evolutionary aspects and functional diversity. Pharmacogenomics J 5, 6–13 (2005). https://doi.org/10.1038/sj.tpj.6500285
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.tpj.6500285
Keywords
This article is cited by
-
Solanidine is a sensitive and specific dietary biomarker for CYP2D6 activity
Human Genomics (2024)
-
CYP1A2 expression rather than genotype is associated with olanzapine concentration in psychiatric patients
Scientific Reports (2023)
-
CYP2D6 Substrate Dispensing Among Patients Dispensed Mirabegron: An Administrative Claims Analysis
Drugs - Real World Outcomes (2023)
-
Effects of CYP2D6 and CYP2C19 genetic polymorphisms and cigarette smoking on the pharmacokinetics of tolperisone
Archives of Pharmacal Research (2023)
-
Association Between the Post-Mortem and Poisoning Concentration of Tramadol and Its Main Metabolite and Genotype of CYP2D6
Pharmaceutical Chemistry Journal (2023)