Abstract
Purpose
To determine the surgical outcome after initial surgery in children with primary developmental glaucoma (PDG).
Methods
Six hundred and twenty-four eyes of 360 consecutive patients who underwent primary combined trabeculotomy–trabeculectomy (CTT) for PDG from January 1990 to June 2004 were studied. The main outcome measures were pre- and postoperative intraocular pressures (IOPs), corneal clarity, visual acuities, refractive errors, success rate, time of surgical failure, complications, and factors associated with poor outcome.
Results
IOP reduced from 28.1±7.5 to 14.9±5.9 mmHg (P<0.0001). Probability of success (IOP<21 mmHg) was 85.2, 80.4, 77.2, 72.6, 66.2, and 57.5% at first, second, third, fourth, fifth, and sixth years, respectively (Kaplan–Meier analysis). The mean follow-up period was 20.3±25.6 months (median, 6 months). Preoperatively, 243 eyes (67.5%) had significant corneal oedema. Postoperatively, normal corneal transparency was achieved in 162 eyes (46.0%). Data on Snellen visual acuity were available in 100 patients (27.8%). At the final follow-up visit, 42 patients (42.0%) had normal visual acuity (≥20/60). Myopia (mean spherical equivalent, 6.1 D) was the most common (75.0%) refractive error. In multivariate analyses, failure increased by three-fold in the presence of preoperative IOP>35 mmHg (hazards ratio (HR)=3.12; 95% confidence interval (CI), 1.4–6.7) and two-fold in cases with a history of prior glaucoma surgery (HR=2.57; 95% CI, 1.1–6.0). There were no major intraoperative complications, bleb-related infection, or endophthalmitis.
Conclusions
This series shows that prolonged IOP control can be achieved in patients with PDG and 42% of the patients gained normal visual acuity.
Similar content being viewed by others
Main
Primary developmental glaucoma (PDG) is a potentially blinding disease, which is refractive to medical treatment.1 Early diagnosis and prompt microsurgical intervention has been advocated by several investigators.1, 2, 3, 4, 5 Goniotomy has traditionally been regarded as the procedure of choice.2, 4, 6, 7, 8 However, the best approach to the surgical treatment of PCG is a subject of considerable debate.9, 10, 11, 12, 13 The developmental glaucoma, which occurs in India,14, 15 Middle East,16 and in several other parts of the world, is more often familial than it is in the West17 and responds less favourably to goniotomy. In such cases, trabeculotomy ab externo is advocated as the initial surgical procedure of choice by several authors.10, 12, 13, 18, 19, 20, 21, 22, 23 Management of PDG is further complicated, as multiple surgical interventions are often required for the control of intraocular pressure (IOP).2, 4, 8, 24, 25, 26 In the search for the best surgical treatment of developmental glaucoma, combined trabeculotomy–trabeculectomy (CTT) owing to its obvious advantages over conventional procedures has emerged as the new treatment modality.27, 28, 29 Over the last decade, the good success rate of CTT reported by Elder28 has been substantiated by other institutions and paediatric ophthalmologists.30, 31, 32, 33, 34, 35, 36
The functional outcome of patients with PDG is poor; no more than 40–50% of these children achieve a vision of 20/50 or better.24, 25 However, only a few reports exist on the long-term surgical outcome in developmental glaucoma.2, 4, 23, 24, 25, 32, 34, 35, 36 The primary objective of the present study was to evaluate the surgical outcome after CTT for PDG in 360 consecutive children (624 eyes) who were operated upon by a single surgeon over a 14.5-year period in a tertiary eye care centre in India. The secondary objective was to identify clinically relevant factors that were associated with a poor outcome.
Methods
Study design
We conducted a retrospective chart review of all records of children who underwent CTT for PDG at the LV Prasad Eye Institute (LVPEI), Hyderabad, India from 1 January 1990 to 30 June 2004. In case of bilateral affliction, the choice of simultaneous bilateral surgery was offered to all patients after thoroughly discussing the risks and benefits with the parents. All surgeries were performed by a single surgeon (AKM) who was also present at almost all follow-up examinations. Before surgery, an informed consent was obtained from the parents of all patients. We excluded patients with a follow-up of less than 3 months from the present study. The use of primary CTT as the main therapeutic modality was approved by the Ethics Committee for Human Research at LVPEI.
Examination procedures
All patients underwent ophthalmological examinations, including applanation tonometry using Perkin's hand-held applanation tonometer in children who were examined under anaesthesia and using Goldmann applanation tonometer in children old enough for slit-lamp examination. Various suitable age-appropriate procedures were used to assess visual acuity. These included grating acuity assessment using Teller Acuity Cards37 (TAC) (Vistech Consultants Inc., Dayton, OH, USA) for infants and toddlers, using a method similar to that described by Trueb et al38 The grating acuity was, however, converted to Snellen equivalent for purposes of analysis and comparison, and acuities within the 99% prediction limits for the child's age were recorded as normal, based on the norms provided by Mayer et al39 Other assessment procedures included a 3-m folding chart containing Lea symbols40 for preschoolers and the logMAR chart for school-aged children and adolescents.41 For children to whom these procedures could not be applied, the ability to fix and follow light using a pen torch was recorded. Although the ability to fix and follow a light does not provide a measure of visual acuity, it can help identify children who are totally blind.42 Retinoscopy was performed manually using a streak retinoscope (Heine, Beta 2000) under cycloplegia (1% cyclopentolate hydrochloride eye drops) in the postoperative period after 6 weeks in eyes with clear corneas. It was performed in the office on cooperative children and during examination under anaesthesia (EUA) for uncooperative children. In such children, the refractive correction was prescribed based on the objective value after appropriate adjustments for cycloplegia. In cooperative children in whom the retinoscopic reflex was irregular and dull owing to the presence of a corneal scar, subjective procedures such as axial length measurements and keratometry were used to obtain a rough estimate, which was then used as a baseline value for further refinements in the subjective refraction.43, 44 Any child whose best-corrected spectacle acuity was below 20/60 was referred to the vision rehabilitation centres for further management. These results are not reported as they are outside the scope of the present study.
Visual acuity was reported according to the World Health Organization classification of visual impairment.45 These categories have been defined based on the best-corrected visual acuity in the better eye and consist of (a) low vision (<20/60–20/400) and (b) blindness (<20/400).
Data analysis and recording
All preoperative, surgical, and postoperative parameters were entered into a Microsoft excel database. Any intra- and perioperative surgical complications and re-treatments were also recorded.
Outcome measures
The outcome measures included preoperative and postoperative IOPs, corneal clarity and diameter, visual acuity, success rate, bleb characteristics, time of surgical failure, and complications. The incidence of endophthalmitis (if any) and anaesthetic morbidity and mortality were analysed.
Surgical procedure
The surgical technique employed in all cases was primary CTT. This technique has previously been described by us.32, 46, 47 In brief, the Schlemm's canal was dissected under a partial thickness limbal-based triangular scleral flap, and trabeculotomy ab externo was performed on both sides of the radial incision. Trabeculectomy was then performed in the usual manner. In cases of bilateral affliction, after completion of surgery on the first eye, the second eye was operated using a similar technique but with a new set of instruments, drapes, drops, gown, gloves, etc, simulating a surgical procedure on a different patient.
Postoperative regimen
A standard postoperative care regimen was followed for all patients. In the immediate postoperative period, all the patients received topical 1% cyclopentolate hydrochloride combined with a corticosteroid-antibiotic preparation that was tapered gradually over a 6-week period. All patients were examined 1, 2, and 3 days after surgery, followed by examination in the office at the end of 1, 3, and 6 weeks and in the office every 3 months thereafter. At each visit, patients were examined fully with a slit-lamp biomicroscope, and the anterior chamber depth, corneal appearance, IOP, bleb appearance, and fundus picture were documented. Patients who could not be examined in the office were scheduled for EUA at the end of 1, 3, and 6 weeks and every 3 months thereafter.
Success criteria
Surgical success and failure were defined before data analysis. The surgery was considered a complete success when the IOP was less than 16 mmHg in patients examined under general anaesthesia or less than 21 mmHg in patients who were old enough to be examined with the slit lamp and when there was no progression of disc cupping or corneal diameter. Success was deemed qualified success if ocular antihypertensive agents were required to maintain these criteria. Failure was defined as uncontrolled IOP despite additional medical therapy, developing hypotony-related maculopathy or sight-threatening complications, or requiring additional surgery.
Statistical analysis
To avoid bias, we evaluated the results in terms of patients rather than eyes.48 For the purpose of statistical analysis, only one randomly chosen eye was included for patients with a bilateral affliction. In cases of unilateral affliction, the affected eye was included. Descriptive statistics are given as mean and standard deviation (SD). Cumulative success probability was determined using Kaplan–Meier survival analysis. Spherical equivalent (SE) was used to analyse the refractive errors. SE was calculated by adding half the cylindrical value to the spherical value of the refractive error. Subjects with SE higher than +0.50 D were considered as having hyperopia and SE≥−0.50 D was considered for analysis of myopia.
The effect of individual clinical and demographic variables on the outcome of treatment was analysed by a series of univariate Cox proportional hazards models.49 Any factor with P<0.15 from the univariate analysis was subjected to a multivariate analysis using the forward stepwise method. The effect of each category of a two-category variable was assessed by keeping either of the two categories as the reference. Results are expressed as hazards ratio (HR) with 95% confidence intervals. P<0.05 was considered as statistically significant.
Using the SPSS (SPSS for Windows, version 10.0, SPSS, Chicago, IL, USA) statistical package, analyses of continuous data were made using the paired ‘t’ test. Ordinal data were analysed by the Wilcoxon signed rank test.
Results
Demographic data
The study group included 360 consecutive patients (624 eyes) who underwent CTT. Three hundred and thirty patients (91.7%) underwent CTT as an initial glaucoma surgery, whereas 30 patients (8.3%) underwent CTT as the second surgical intervention, in whom the primary glaucoma surgery was performed elsewhere. Table 1 summarizes the demographics of our study population. The mean age at surgery was 31.9±54.6 months (range, 0–353.13 months; median, 6.5 months). The study population primarily had PDG (360, 100%). Two hundred and sixty-four (73.3%) patients were affected in both eyes. Follow-up averaged 20.3±25.6 months (range, 3–120 months; median, 6 months).
Intraocular pressure
Preoperatively, the mean IOP was 28.1±7.5 mmHg (range, 10–56 mmHg) with patients using an average of 0.6±0.7 medications. On the last follow-up visit, the IOP averaged 14.9±5.9 mmHg (range, 1–42 mmHg) (P<0.0001, paired t-test) with a mean of 0.2±0.4 medications (P<0.0001, Wilcoxon signed rank test). Preoperatively, 170 of 360 eyes (47.2%) were on antiglaucoma medication, whereas at the last follow-up, 63 of 360 eyes (17.5%) required antiglaucoma medication. Although some of the patients had normal IOP preoperatively, they were operated upon because they were on antiglaucoma medication and the diagnosis of glaucoma was confirmed based on a comprehensive ocular examination that included corneal diameters.
Corneal diameter and clarity
At presentation, mean (±SD) horizontal corneal diameters were 13.0±1.4 mm (range, 6.5–18.0 mm). Preoperatively, approximately one-fifth of the eyes (75, 22.3%) had normal corneal transparency and the decision for surgery in these eyes was based on high IOP, megalocornea, and gonioanamoly suggestive of developmental glaucoma. Two hundred and forty-three eyes (67.9%) had varying degrees of corneal oedema. Thirty-four eyes (9.5%) had corneal scarring. At the final follow-up visit, corneal clarity data were not available for 7 eyes. One hundred and sixty two (46.0 %) had normal corneal transparency, 118 eyes (33.5 %) corneal oedema, and 72 eyes (20.5%) corneal scarring. The improvement in corneal transparency was statistically significant (P<0.0001). The preoperative and postoperative clinical photographs of congenital, infantile, and juvenile-onset developmental glaucomas are depicted in Figures 1a–f.
(a) Clinical appearance of a 1-week-old child showing bilateral corneal oedema resulting from congenital glaucoma. (b) Six-month postoperative photograph of the child seen in (a). (c) Clinical appearance of a 6-month-old child showing megalocornea and corneal oedema of the left eye resulting from infantile glaucoma. (d) Five-year postoperative photograph of the child seen in (c). (e) Clinical appearance of a 3-year-old child showing bilateral buphthalmos resulting from juvenile-onset developmental glaucoma. (f) Ten-year postoperative photograph of the child seen in (e).
Visual acuity
Visual acuity data were available for approximately two-third of the patients (229, 63.6%). Of these, best-corrected Snellen visual acuity could be recorded for 100 patients (27.8%). In the remaining patients, grating acuity using TAC was recorded. However, for the purpose of reporting, we analysed Snellen acuity data only. At the final postoperative visit, 42 patients (42.0%) achieved normal visual acuity (≥20/60). Thirty-one (31.0%) patients had low vision and 27 (27.0%) were blind as defined by WHO criteria of vision loss.
Refractive errors
Data regarding refractive status were available for 202 (56.1%) patients. Approximately three-quarter of the eyes (149, 73.8%) were myopic with mean SE of 6.0±4.9 D (range, 0.75–32.0 D). Twenty-seven eyes (13.4%) were emmetropic and the remaining 26 eyes (12.9%) were hypermetropic with mean SE of 3.9±4.7 D (range, 0.75–21.0 D).
Success rate
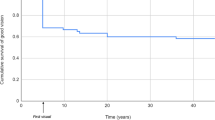
Figure 2 demonstrates Kaplan–Meier survival curve of the entire group. Complete success as defined in the present study was obtained in 279 eyes (77.5%). Kaplan–Meier survival analysis revealed 12-, 24- 36-, 48-, 60-, 72-, and 84-month success rates of 85.2±2.3, 80.4±2.9, 77.2±3.3, 72.6±4.1, 66.2±5.2, and 57.5±6.6%, respectively, that was maintained for 8 years of follow-up. Qualified success was obtained in 19 eyes (5.3%), whereas 62 eyes (17.2%) were considered to be failures.
Factors associated with poor functional outcome
Table 2 presents univariate and multivariate comparisons of final surgical outcome according to several patient characteristics. In univariate analysis, the failure rate was higher in patients who were older than 6 months at the time of surgery than in those who were younger than 6 months of age (Figure 3) at the time of surgery (21.4 vs 12.8%; HR=1.98). Patients who presented with IOP>35 mmHg performed significantly worse (Figure 4) than those with IOP≤35 mmHg (31.9 vs 15.1%; HR=2.43). Patients who underwent prior glaucoma surgery had greater chances of failure with CTT as the second procedure (43.3 vs 14.8%; HR=3.17). No significant associations (P<0.05) were found with the other factors evaluated. In multivariate analyses, the significant factors were the preoperative IOP>35 mmHg and a history of prior glaucoma surgery (Table 2). Patients who presented with IOP>35 mmHg had twice the risk of failure (HR=3.12) as compared with those who presented with IOP≤35 mmHg. Patients who underwent prior glaucoma surgery had thrice the risk of failure (HR=2.57) as compared with those who underwent CTT as an initial surgical procedure. There were five patients who presented with a history of prior glaucoma surgery and uncontrolled glaucoma with IOP>35 mmHg; four (80%) of these patients failed and the risk of failure doubled (HR=6.42) when the two factors (IOP>35 mmHg and prior surgery) co-existed as opposed to when they contributed individually.
Complications
Operative complications
Operative complications were encountered in 64 of 360 eyes (17.8%) and are listed in Table 3. There was no sight-threatening intraoperative complication. Of the minor postoperative complications, the most common one was shallow anterior chamber, which occurred in 51 (14.2%) eyes in the first postoperative week; however surgical reformation was required in only eight (15.7%) of these eyes and in the remainder (43 eyes, 84.3%) the anterior chamber deepened spontaneously. One eye (0.3%) developed choroidal detachment that was successfully managed conservatively. Four eyes (1.1%) required enucleation because of painful blind eye secondary to absolute glaucoma. Rhegmatogenous retinal detachment (RD) occurred in eight (2.2%) eyes; four eyes underwent successful vitreo-retinal procedures and these eyes regained the baseline visual acuity. Four eyes were deemed inoperable owing to total RD with proliferative vitreoretinopathy. There was no incidence of bleb leakage, bleb-related infection, or endophthalmitis.
Anaesthetic complications
Apnea occurred in 21 (5.8%) patients, who were successfully resuscitated. Anaesthetic complication in the form of bleeding from trachea occurred in one patient. This was observed during the recovery phase from anaesthesia and was successfully managed.
The most serious anaesthetic complication was cardio-pulmonary arrest that occurred 5 h postoperatively following aspiration during feeding in one child, who could not be resuscitated. Two children had delayed recovery (2 and 4 h, respectively) because of prematurity (weight at surgery was 2.2 and 2.3 kg, respectively). Both children were shifted to the paediatric intensive care unit. The child with 2 h delayed recovery responded well and has completed 5 years of follow-up; the other child expired 48 h later.
Discussion
The primary objective in the management of PDG is to normalize and permanently control the IOP, thereby preventing loss of visual function and preserving the ocular integrity. Developmental glaucoma is a worldwide disease and the impact on the visual function is extreme. The only effective and definitive form of treatment of most of the PDGs is surgical. Traditionally, goniotomy2, 4, 6, 7, 8 or trabeculotomy10, 12, 13, 18, 19, 20, 21, 22, 23 has been the procedure of choice amongst the glaucoma specialists. However, goniotomy should never be performed without a view of the angle.50 On the other hand, the disadvantage of trabeculotomy is that the Schlemm's canal may not be found in 11–15% of the procedures.19, 28 In view of these limitations, some surgeons have resorted to primary trabeculectomy in the management of PDG because primary trabeculectomy is easier than goniotomy or trabeculotomy.51, 52, 53, 54, 55 Unfortunately, many of the patients with PDG require multiple surgeries to achieve a successful outcome.2, 4, 8, 24, 25, 26
Over the last decade, 360° trabeculotomy55, 56 and CTT have emerged as alternatives to the conventional modes of surgery.27, 28, 29 Of the two, many surgeons have preferred CTT as an initial procedure for several ethnic population groups such as Middle East,30, 31 Jerusalem,28 and India.32, 33, 34, 35, 36 The most compelling argument in favour of primary CTT in our Indian patient population was the higher incidence of successful IOP control with a single operative procedure, as has already been reported by us.32, 33, 34, 35, 36 The aim of the present study was to evaluate the outcome of PDG operated by a single surgeon over a 14.5-year period. Kaplan–Meier survival analysis demonstrated the success probabilities of 85.2, 80.4, 77.2, 72.6, 66.2, and 57.5% at the first, second, third, fourth, fifth, and sixth years, respectively. Our success rate with initial surgery is significantly better than most of the reported results of initial goniotomy, trabeculotomy ab externo, or primary trabeculectomy. The results of the present study are at least as effective as multiple goniotomies and are comparable to the results of 360° trabeculotomy.55, 56 Recently, Mendicino et al56 compared the long-term surgical results of 360° trabeculotomy and goniotomy and reported significantly better IOP control with the former technique, as they believed that it is more standardized than the latter technique. We do not have any experience in performing 360° trabeculotomy, but excellent IOP control and an efficacy equal to that of multiple standard procedures make it an attractive choice among the available angle procedures. Filous and Brunova57 reported the results of modified trabeculotomy in the treatment of primary congenital glaucoma, employing trabeculotomy probes more closely corresponding to the variable course of Schlemm's canal. They reported a success rate of 87% over a mean follow-up of 38.4±22.5 months and concluded that probing with the innovative instrument was easier and safer compared to standard trabeculotomy probes. We speculate that our success rate of CTT could be further enhanced with the use of a modified trabeculotomy probe.57
Mullaney et al31 and Al-Hazmi et al30 used mitomycin-C (MMC) in primary CTT and reported a higher success rate. Our results are comparable to Mullaney et al31 but we do not believe in using MMC in primary surgery. However, in cases of failed primary CTT, we perform repeat trabeculectomy with MMC as a secondary procedure.58, 59 Whether primary CTT is superior to trabeculotomy or trabeculectomy alone is a subject of considerable debate. In a small series of seven Arab Bedouin infants, Biender and Rothkoff60 found no difference between trabeculotomy and CTT in patients with congenital glaucoma. Dietlein et al61 investigated the outcome of trabeculotomy, trabeculectomy, and a combined procedure as initial surgical treatment in primary congenital glaucoma. Although combined procedure seems to have a favourable outcome, the advantages of this procedure over trabeculotomy or trabeculectomy were not statistically significant after 2 years. Elder28 compared primary trabeculectomy with CTT and found the latter to be superior to the former. In the present study, all patients underwent CTT and our results are comparable to those reported by Elder28 and better than the results of goniotomy, trabeculotomy, or trabeculectomy alone. The superior results of the combined procedure may be because of the dual outflow pathway as explained by Elder,28 Mandal,33 and Mandal et al.32, 35, 36, 46, 47 Based on our results, we believe that CTT may be considered as an initial procedure in PDG.
In the present series, 243 (67.9%) patients presented with variable degrees of corneal oedema and 34 (9.5%) patients presented with corneal scarring that precluded the possibility of goniotomy. Postoperatively at the last follow-up visit, 162 (46%) patients had clear corneas. Although corneal oedema cleared in about half of the patients, its persistence and the development of corneal scarring in the remaining patients was the major reason for visual impairment (best-corrected visual acuity <20/60 in the better eye). Nevertheless, IOP was under control in these patients.
The outcome of surgery for PDG is generally reported by IOP control and by life-table analysis of success rates.10, 13, 21, 22, 27, 51, 54 The primary purpose of our study was to determine the functional outcome of the treated patients. However, at the final postoperative visit, 42 patients (42.0%) achieved normal visual acuity (≥20/60). Thirty-one patients (31.0%) had low vision. Only 27 patients (27.0%) were blind as defined by WHO criteria of vision loss.45 Literature review revealed that the visual outcomes in PDG have not been consistent.2, 3 and 4, 8, 23, 24, 25, and 26, 32, 34, 35 and 36, 56, 57, 62, 63 Although our results of visual outcome are not as good as those reported by some of these investigators,23, 56, 57 the results are still encouraging because most of our patients had earlier onset of the disease and approximately 80% patients presented with corneal oedema/scarring. Early clearance of corneal oedema is essential for an infant's visual development, and several investigators have suggested that surgery should be performed early – within a few days to a few weeks of life.1, 2, 3, 4, 5 In the present study, we performed surgery as soon as the patients presented to us. Given the high percentage of persistent corneal oedema despite lowering of IOP in our cohort, the benefits of penetrating keratoplasty for improved visual outcome were discussed with the parents.
One of the objectives of the present study was to identify clinically relevant factors that were associated with a poor outcome. This objective was achieved by evaluating the role of various possible risk factors on the outcome of treatment. The intent was to provide predictive information on the risk of failure according to specific patient characteristics at baseline. In univariate analysis, several factors were associated with failure. These included age of the patient at surgery, preoperative IOP, and a history of prior glaucoma surgery (Table 2). Patients who were older than 6 months of age at the time of surgery, with preoperative IOP>35 mmHg and who had undergone previous glaucoma surgery had approximately twice the risk of failure leading to a poor outcome. The outcome was closely linked to the magnitude of the preoperative IOP and to the history of a prior glaucoma surgery. Preoperative IOP>35 mmHg was strongly associated with failure, as also the history of a prior glaucoma surgery. However, when these two factors coexisted in a small subset of patients (n=5), the risk of failure was six times (HR=6.42). Clearly, increased IOP at surgery and a repeat surgery are the risk factors associated with a poor outcome. Hence, the present study substantiates the fact that the chances of success are highest with the first surgical procedure.3, 21 Previous studies have shown another important factor responsible for a poor outcome, the earlier onset of disease.1, 2, 3, 4, 5, 9, 10, 11 However, in our study, we cannot comment on this factor, as we did not have accurate information regarding the age of onset of disease in approximately two-third of our patients (61.4%). This lack of information was related to the retrospective nature of our study and poor recall ability of most of the parents with regard to the exact time of onset of symptoms.
In addition to early surgery, appropriate refractive correction coupled with aggressive amblyopia therapy is essential for improved visual outcome in patients with PDG.1, 3, 5 Approximately, three-quarter of our patients (73.8%) were myopic and the mean refraction was −6.0 D. Previous investigators have also reported the occurrence of myopia in children with PDG.1, 2, 4, 25 Interestingly, we also observed emmetropia in 27 eyes (13.4%) and hypermetropia in 26 eyes (12.9%). When we compared the present series to our previously reported series on CTT performed within 1 month35 and 6 months of age,36 we observed that the incidence of myopia is higher (73.8%) in the current series as opposed to 53.8 and 57.3%, respectively. We attribute this higher incidence of myopia to the late presentation and advanced nature of PDG in a significantly greater proportion of patients in the present study. We speculate that the myopia in the present study was axial in nature. The attendant complications of axial myopia ought to be discussed with the parents of children with PDG.
In the present study, there were no serious intraoperative complications. Of the minor postoperative complications, shallow anterior chamber occurred in 51 (14.2%) eyes in the first postoperative week; however, surgical reformation was required in only eight (15.7%) eyes. In the remainder (43 eyes, 84.3%), the anterior chamber deepened spontaneously.
One eye (0.3%) developed choroidal detachment that was successfully managed conservatively. Four eyes (1.1%) required enucleation because of painful blind eye secondary to absolute glaucoma. Rhegmatogenous RD occurred in eight (2.2%) eyes; four eyes underwent successful vitreo-retinal procedures and these eyes regained the baseline visual acuity. Four eyes were deemed inoperable owing to total RD with proliferative vitreoretinopathy. The long-term complications of the present series are comparable to those reported by Litinsky et al,64 Shaffer,2 and Shaffer and Hoskins.4 The treating ophthalmologist ought to bear in mind the long-term complications as well as the long-term drift in IOP control61, 65, 66 when counselling the parents before surgery.
In the present study, 264 (73.3%) patients underwent simultaneous bilateral surgery to avoid another long anaesthesia despite the theoretical risk of endophthalmitis. There was no incidence of bleb leakage, bleb-related infection, or endophthalmitis. Based on these results, we believe that simultaneous bilateral surgery can be undertaken safely in these tiny patients.34 However, strict intraoperative asepsis is mandatory and the second eye should be operated as if performing a surgical procedure on a different patient. Nevertheless, the decision-making process must involve the treating surgeon, the parents, and the anaesthesiologists.34
The limitations of the present study are its non-randomized, retrospective design and the variability in the timing of the surgery that depended on the clearance for anaesthesia and variability in the age at presentation. In addition, Snellen acuity data were available only for 100 (29.6%) patients.
In conclusion, primary CTT is safe and effective in patients with all types of PDG. Our study with PDG achieved prolonged IOP control and experienced good visual outcomes; 42% of the patients achieved normal visual acuity. Preoperative IOP>35 mmHg and a history of prior failed glaucoma surgery were significant risk factors for poor outcome. The results of this study provide useful information relating to the functional outcome and long-term complications. Furthermore, because this study reflects the experience of a single surgeon over a 14.5-year period, the data may be regarded as the minimum level of achievable outcomes for PDG. Such data will help in parental counselling and improve compliance.
References
deLuise VP, Anderson DR . Primary infantile glaucoma.(Congenital glaucoma). Surv Ophthalmol 1983; 28: 1–19.
Shaffer RN . Prognosis of goniotomy in primary infantile glaucoma (trabeculodysgenesis). Trans Am Ophthalmol Soc 1982; 80: 321–325.
Haas J . Principles and problems of therapy in congenital glaucoma. Invest Ophthalmol 1968; 7: 140–146.
Shaffer RN, Hoskins HD . Montgomery lecture. Goniotomy in the treatment of isolated trabeculodysgenesis (primary congenital [infantile] developmental glaucoma). Trans Ophthalmol Soc UK 1983; 103: 581–585.
Stamper RL, Lieberman MF, Drake MV (eds). Becker–Shaffer's Diagnosis and Therapy of the Glaucomas. 7th edn. CV Mosby Co.: St Louis, 1999, pp 386, 668.
Barkan O . Technique of goniotomy. Arch Ophthalmol 1938; 19: 217–223.
Barkan O . Operation for congenital glaucoma. Am J Ophthalmol 1942; 25: 552–568.
Broughton WL, Parks MM . An analysis of treatment of congenital glaucoma by goniotomy. Am J Ophthalmol 1981; 91: 566–572.
Hoskins Jr HD, Shaffer RN, Hetherington J . Goniotomy vs trabeculotomy. J Pediatr Ophthalmol Strabismus 1984; 21: 153–158.
Anderson DR . Trabeculotomy compared to goniotomy for glaucoma in children. Ophthalmology 1983; 90: 805–806.
Khaw PT . What is the best primary surgical treatment for the infantile glaucomas? [editorial). Br J Ophthalmol 1996; 80: 495–496.
Luntz MH . The advantages of trabeculotomy over goniotomy. J Pediatr Ophthalmol Strabismus 1984; 21: 150–153.
McPherson Jr SD, Berry DP . Goniotomy vs external trabeculotomy for developmental glaucoma. Am J Ophthalmol 1983; 95: 427–431.
Panicker SG, Reddy ABM, Mandal AK, Ahmed N, Nagarajaram HA, Hasnain SE et al. Identification of novel mutations causing familial primary congenital glaucoma in Indian pedigrees. Invest Ophthalmol Vis Sci 2002; 43: 1358–1366.
Reddy ABM, Panicker SG, Mandal AK, Hasnain SE, Balasubramanian D . Identification of R368H as a predominant CYP1B1 allele causing primary congenital glaucoma in Indian patients. Invest Ophthalmol Vis Sci 2003; 44: 4200–4203.
Elder MJ, Cock RD . Childhood blindness in the West Bank and Gaza strip: prevalence, aetiology and hereditary factors. Eye 1993; 7: 580–583.
Jaffar MS . Care of the Infantile Glaucoma Patient. Ophthalmology Annual, Vol. 7. Raven Press: New York, 1988, pp 15–37.
Harms H, Danheim R . Trabeculotomy – results and problems. Adv Ophthalmol 1965; 22: 121–131.
Harms H, Danheim R . Epicritical consideration of 300 cases of trabeculotomy ‘ab externo’. Trans Ophthalmol Soc UK 1970; 89: 491–499.
McPherson Jr SD . Results of external trabeculotomy. Am J Ophthalmol 1973; 76: 18–20.
McPherson Jr SD, McFarland D . External trabeculotomy for developmental glaucoma. Ophthalmology 1980; 87: 302–305.
Quigley HA . Childhood glaucoma: results with trabeculotomy and study of reversible cupping. Ophthalmology 1982; 89: 219–226.
Akimoto M, Tanihara H, Negi A, Nagata M . Surgical results of trabeculotomy ab Externo for developmental glaucoma. Arch Ophthalmol 1994; 112: 1540–1544.
Richardson KT, Ferguson WJ, Shaffer RN . Long-term functional results in infantile glaucoma. Trans Am Acad Opthalmol Otolaryngol 1967; 71: 833–837.
Biglan AW, Hiles DA . The visual results following infantile glaucoma surgery. J Pediatr Ophthalmol Strabismus 1979; 16: 377–381.
Morgan KS, Black B, Ellis FD, Helveston EM . Treatment of congenital glaucoma. Am J Ophthalmol 1981; 92: 799–803.
Luntz MH . Congenital, infantile, and juvenile glaucoma. Ophthalmology 1979; 86: 793–802.
Elder MJ . Combined trabeculotomy–trabeculectomy compared with primary trabeculectomy for congenital glaucoma. Br J Ophthalmol 1994; 78: 745–748.
O’Connor G . Combined trabeculotomy–trabeculectomy for congenital glaucoma. Br J Ophthalmol 1994; 78: 735.
Al-Hazmi A, Zwaan J, Awad A, Al-Mesfer S, Mullaney PB, Wheeler DT et al. Effectiveness and complications of mitomycin-C use during pediatric glaucoma surgery. Ophthalmology 1998; 105: 1915–1920.
Mullaney PB, Selleck, Al-Award A, Al-Mesfer S, Zwaan J . Combined trabeculotomy and trabeculectomy as initial procedure in uncomplicated congenital glaucoma. Arch Ophthalmol 1999; 117: 457–460.
Mandal AK, Naduvilath TJ, Jayagandhan A . Surgical results of combined trabeculotomy–trabeculectomy for developmental glaucoma. Ophthalmology 1998; 105: 974–982.
Mandal AK . Primary combined trabeculotomy–trabeculectomy for early onset glaucoma in Sturge–Weber syndrome. Ophthalmology 1999; 106: 1621–1627.
Mandal AK, Bhatia PG, Gothwal VK, Reddy VM, Sriramulu P, Prasad MS et al. Safety and efficacy of simultaneous bilateral primary combined trabeculotomy–trabeculectomy for developmental glaucoma in India. Indian J Ophthalmol 2002; 50: 13–19.
Mandal AK, Gothwal V, Bagga H, Nutheti R, Mansoori T . Outcome of surgery on infants younger than 1 month with congenital glaucoma. Ophthalmology 2003; 110: 1909–1915.
Mandal AK, Bhatia PG, Bhaskar A, Nutheti R . Long-term surgical and visual outcomes in Indian children with developmental glaucoma operated on within 6 months of birth. Ophthalmology 2004; 111: 283–290.
Teller DY, McDonald MA, Preston K, Sebris SL, Dobson V . Assessment of visual acuity in infants and children: the acuity card procedure. Dev Med Child Neurol 1986; 28: 779–789.
Trueb L, Evans J, Hammel A, Bartholomew P, Dobson V . Assessing visual acuity of visually impaired children using the Teller Acuity Card procedure. Am Orthopt J 1992; 42: 149–154.
Mayer DL, Beiser AS, Warner AF, Pratt EM, Raye KN, Lang JM et al. Monocular acuity norms for the Teller Acuity Cards between ages one month and four years. Invest Ophthalmol Vis Sci 1995; 36: 671–685.
Hyvarinen L, Nasanen R, Laurinen P . New visual acuity test for preshool children. Acta Ophthalmol (Kbh) 1980; 58: 507–511.
Bailey IL, Lovie-Kitchin JE . New design principles for visual acuity letter charts. Am J Optom Physiol Opt 1976; 53 (11): 740–745.
Hatton DD . Model registry of early childhood visual impairment: first-year results. J Vis Imp Blindness 2001; 95 (7): 418–433.
Belkin M, Ticho U, Susal A, Levinson A . Ultrasonography in refraction of aphakic infants. Br J Ophthalmol 1973; 57: 845–848.
Gordon RA, Doniz BB . Refractive development of the human eye. Arch Ophthalmol 1985; 103: 785–789.
International Statistical Classification of Diseases and Related Health Problems (ICD). 10th revision. Vol. 1. WHO: Geneva, 1992, pp 456–457.
Mandal AK . Current concepts in the diagnosis and management of developmental glaucomas. Indian J Ophthalmol 1993; 41: 51–70.
Mandal AK . Microsurgical technique combines trabeculotomy and trabeculectomy to treat developmental glaucoma. Ocular Surg News Int Ed 1994; 5 (8): 38–43.
Newcombe RG, Duff GR . Eyes or patients? Traps for the unwary in the statistical analysis of ophthalmological studies. Br J Ophthalmol 1987; 71: 645–646.
Cox DR . Regression models and life-tables (with discussion). J R Stat Assoc 1972; 34: 187–220.
Russell-Eggitt I . In defence of goniotomy [letter]. Br J Ophthalmol 1995; 79: 709.
Burke JP, Bowell R . Primary trabeculectomy in congenital glaucoma. Br J Ophthalmol 1989; 73: 186–190.
Fulcher T, Chan J, Lanigan B, Bowell R, O’Keefe M . Long-term follow-up of primary trabeculectomy for primary infantile glaucoma. Br J Ophthalmol 1996; 80: 499–502.
Debnath SC, Teichmann KD, Salamah K . Trabeculectomy vs trabeculotomy in congenital glaucoma. Br J Ophthalmol 1989; 73: 608–611.
Rao KV, Sai CM, Babu BVN . Trabeculectomy in congenital glaucoma. Indian J Ophthalmol 1984; 32: 439–440.
Beck AD, Lynch MG . 360° trabeculotomy for primary congenital glaucoma. Arch Ophthalmol 1995; 113: 1200–1202.
Mendicino ME, Lynch MG, Drack A, Beck AD, Harbin T, Pollard Z et al. Long-term surgical and visual outcomes in primary congenital glaucoma: 360° trabeculotomy vs goniotomy. J AAPOS 2000; 4: 205–210.
Filous A, Brunova B . Results of the modified trabeculotomy in the treatment of primary congenital glaucoma. J AAPOS 2002; 6: 182–186.
Mandal AK, Walton DS, John T, Jayagandhan A . Mitomycin-C augmented trabeculectomy in refractory congenital glaucoma. Ophthalmology 1997; 104: 996–1001.
Mandal AK, Prasad K, Naduvilath TJ . Surgical results and complications of Mitomycin-C augmented trabeculectomy in refractory developmental glaucoma. Ophthalmic Surg Lasers 1999; 30: 473–480.
Biedner BZ, Rothkoff L . Combined trabeculotomy–trabeculectomy compared with primary trabeculotomy for congenital glaucoma. J Pediatr Ophthalmol Strabismus 1998; 35: 49–50.
Dietlein TS, Jacob PC, Krieglstein GK . Prognosis of primary ab externo surgery for primary congenital glaucoma. Br J Ophthalmol 1999; 83: 317–322.
Scheie Harold G . The management of infantile glaucoma. Arch Ophthalmol 1959; 62: 35–54.
Robin AL, Quigley HA, Pollack IP, Maumenee AE, Maumenee IH . An analysis of visual acuity, visual fields, and disc cupping in childhood glaucoma. Am J Ophthalmol 1979; 88: 847–858.
Litinsky SM, Shaffer RN, Hetherington J, Hoskins HD . Operative complications of goniotomy. Trans Am Ophthalmol Oto 1977; 83: 78–79.
Russell-Eggitt IM, Rice NS, Jay B, Wyse RK . Relapse following goniotomy for congenital glaucoma due to trabecular dysgenesis. Eye 1992; 6: 197–200.
Rice NSC . The surgical management of the congenital glaucomas. Aust J Ophthalmol 1977; 5: 174–179.
Acknowledgements
Supported in part by the Hyderabad Eye Research Foundation, LV Prasad Eye Institute, Hyderabad, India.
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented as a poster at the Association for Research in Vision and Ophthalmology meeting Fort Lauderdale, Florida, May 1–5, 2005
The authors have no proprietary interest in any of the materials or techniques used in this study.
Rights and permissions
About this article
Cite this article
Mandal, A., Gothwal, V. & Nutheti, R. Surgical outcome of primary developmental glaucoma: a single surgeon's long-term experience from a tertiary eye care centre in India. Eye 21, 764–774 (2007). https://doi.org/10.1038/sj.eye.6702324
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.eye.6702324
Keywords
This article is cited by
-
Primary congenital glaucoma surgery: outcomes and visual function
International Ophthalmology (2021)
-
Quality of life following surgery for congenital glaucoma: findings of the LVPEI congenital glaucoma registry
Eye (2019)
-
Long-term outcome of combined trabeculotomy–trabeculectomy by a single surgeon in patients with primary congenital glaucoma
Eye (2018)
-
Early-onset glaucoma in Axenfeld–Rieger anomaly: long-term surgical results and visual outcome
Eye (2016)
-
Childhood glaucoma surgery in the 21st Century
Eye (2014)