Abstract
OBJECTIVE: To determine the relative frequencies of complications in severe early intrauterine growth-restricted (IUGR) infants.
METHODS: All infants 32 weeks gestation or less with birth weight less than the fifth percentile admitted from January 1991 to December 1998 were identified retrospectively. Two infants were identified for each IUGR case: the subsequent admission with birth weight ±100 g of the case, and the subsequent admission with the same gestational age. Infants with multiple congenital anomalies, congenital infections or admission after 48 hours of age were excluded. Maternal and neonatal demographic data, neonatal morbidity and mortality until discharge were gathered by chart review.
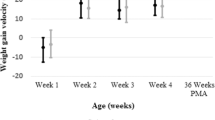
RESULTS: A total of 39 IUGR identified infants met criteria, with 41 gestational age infants and 33 birth weight infants. Mean birth weights and gestational ages for the IUGR group, gestational age group, and birth weight group were 744 g and 29.6 weeks, 1370 g and 29.7 weeks, and 781 g and 25.5 weeks respectively. Mortality was higher for IUGR infants than gestational age infants (20.5 vs 0%), but less than the birth weight infants (30%). In surviving infants, total ventilator days, total oxygen days, days to full feeds, and patent ductus arteriosis, were higher for IUGR infants than gestational age infants, but less than birth weight infants. Hypoglycemia, direct hyperbilirubinemia, necrotizing enterocolitis (NEC), thrombo-cytopenia, chronic lung disease and feeding difficulties occurred more frequently in IUGR infants than in both other groups. Length of stay for survivors and incidence of retinopathy of prematurity (ROP) was similar for the IUGR and birth weight infants.
CONCLUSIONS: Infants born prematurely who are also severely IUGR have higher neonatal morbidity and mortality when compared to infants of similar gestational age. The surviving IUGR infants had less intraventricular hemorrhage and periventricular leukomalacia than less mature infants of comparable birth weight, but a similar incidence of ROP and length of stay. They had a higher incidence of NEC, direct hyperbilirubinemia and chronic lung disease, probably due to end-organ damage in utero from chronic placental insufficiency. These findings highlight the unique pattern of mortality and morbidity seen in infants with severe early IUGR.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Lapillone A, Peretti N, Ho PS, Claris O, Salle BL . Aetiology, morphology and body composition of infants born small for gestational age. Acta Paediatr Suppl 1997;423:173–176.
Dincsoy MY, Kim YM, Siddiq F, Garcia MG, Williams H . Intracranial hemorrhage in small-for gestational-age neonates: comparison with weight-matched appropriate-for-gestational-age infants. Clin Pediatr 1988;27:21–26.
Amin H, Singhal N, Sauve RS . Impact of intrauterine growth restriction on neurodevelopmental and growth outcomes in very low birth weight infants. Acta Paediatr 1997;86:306–314.
Tyson JE, Kennedy K, Broyles S, Rosenfeld CR . The small for gestational age infant: accelerated or delayed pulmonary maturation? Increased or decreased survival? Pediatrics 1995;95:534–538.
Palo P, Erkkola R . Risk factors and deliveries associated with preterm, severely small for gestational age fetuses. Am J Perinatal 1993;10:88–91.
Ley D, Wide-Swensson D, Lindroth M, Svenningsen N, Marsal K . Respiratory distress syndrome in infants with impaired intrauterine growth. Acta Pediatr 1997;86:1090–1096.
Gortner L, Wauer RR, Stock GJ . et al. Neonatal outcome in small for gestational age infants: do they really better? J Perinatal Med 1999;27:484–489.
Ruys-Dudok van Heel I, de Leeuw R . Clinical outcome of small for gestational age preterm infants. J Perinatal Med 1989;17:77–83.
Bernstein IM, Horbar JD, Badger GJ, Ohlsson A, Golan A . Morbidity and mortality among very-low-birth-weight neonates with intrauterine growth restriction. Am J Obstet Gynecol 2000;182:198–206.
Chen SJ, Vohr BR, Oh W . Effects of birth order, gender and intrauterine growth retardation on the outcome of very low birth weight in twins. J Pediatr 1993;123:132–136.
Bardin C, Zelkowitz P, Papageorgiou A . Outcome of small-for-gestational age and appropriate-for-gestational age infants born before 27 weeks of gestation. Pediatrics 1997;100:e4.
Malcolm G, Ellwood D, Devonald K, Beilby R, Henderson-Smart D . Absent or reversed end diastolic flow velocity in the umbilical artery and necrotising enterocolitis. Arch Dis Child 1991;66:805–807.
Newell SJ . Enteral feeding of the micropremie. Clin Perinatol 2000;27:221–234.
Robel-Tillig E, Vogtmann C, Faber R . Postnatal intestinal disturbances in small-for-gestational-age premature infants after prenatal haemodynamic disturbances. Acta Pediatr 2000;89:324–330.
Ewer AK, McHugo JM, Chapman S, Newell SJ . Fetal echogenic gut: a marker of intrauterine gut ischaemia? Arch Dis Child 1993;69:510–513.
Alexander GR, Himes JH, Kaufman RB, Mor J, Kogan M . A United States national reference for fetal growth. Obstet Gynecol 1996;87:163–168.
Stringer MD, Thornton JG, Mason GC . Hyperechogenic fetal bowel. Arch Dis Child Fetal Neonatal Ed 1996;74:F1–2.
Shanklin DR, Cooke RJ . Effects of intrauterine growth on intestinal length in the human fetus. Biol Neonate 1993;64:76–81.
Xu RJ, Mellor DJ, Birtles MJ, Reynolds GW, Simpson HV . Impact of intrauterine growth retardation on the gastrointestinal tract and the pancreas in newborn pigs. J Pediatr Gastroenterol Nutr 1994;18:231–240.
Flecknell PA, Wootton R, John M, Royston JP . Pathologic features of intra-uterine growth retardation in the piglet: differential effects on organ weights. Diagn Histopathol 1981;4:295–298.
Tanaka M, Natori M, Ishimoto H, Miyazaki T, Kobayashi T, Nozawa S . Experimental growth retardation produced by transient period of uteroplacental ischemia in pregnant Sprague–Dawley rats. Am J Obstet Gynecol 1994;171:1231–1234.
Ernst LM, Salafia CM, Carter AM, Pezzullo JC . Hepatic histology in intrauterine growth retardation following uterine artery ligation in the guinea pig. Pediatr Pathol 1993;13:763–772.
Detmer A, Carter AM, Thomas CR . The metabolism of free fatty acids and tricylglycerols by the fetal liver in a guinea pig model of intrauterine growth retardation. Pediatr Res 1992;32:441–446.
Boehm G, Muller DM, Teichmann B, Krumbiegel P . Influence of intrauterine growth retardation on parameters of liver function in low birth weight infants. Eur J Pediatr 1990;149:396–398.
Boehm G, Teichmann B, Krumbiegel P . Hepatic biotransformation capacity in low-birth-weight infants as measured with the [15N]methacetin urine test: influences of gestational age, postnatal age, and intrauterine growth retardation. Biol Neonate 1995;68:19–25.
Boehm G, Senger H, Braun W, Beyreiss K, Raiha NC . Metabolic differences between AGA- and SGA-infants of low birth weight. I. Relationship to intrauterine growth retardation. Acta Paediatr Scand 1988;77:19–23.
Lane RH, Flozak AS, Ogata ES, Bell GI, Simmons RA . Altered hepatic gene expression of enzymes involved in energy metabolism in the growth-retarded fetal rat. Pediatr Res 1996;39:390–394.
Bisquera JA, Cooper TR, Berseth CL . Impact of necrotizing enterocolitis on length of stay and hospital charges in very low birth weight infants. Pediatrics 2002;109:423–428.
Akre O, Lipworth L, Cnattingius S, Sparen P, Ekbom A . Risk factor patterns for cryptorchidism and hypospadius. Epidemiology 1999;10:364–369.
Weidner IS, Moller H, Jensen TK, Skakkebach NE . Risk factors for cryptorchidism and hypospadius. J Urol 1999;161:1606–1609.
Fredell L, Lichtenstein P, Pedersen NL, Svensson J, Nordenskjold A . Hypospadius is related to birth weight in discordant monozygotic twins. J Urol 1998;160:2197–2199.
Hussain N, Chaghti A, Herndon A . et al. Hypospadius and early gestation growth restriction in infants. Pediatrics 2002;109:473–478.
Barker DJ . Intrauterine programming of coronary heart disease and strokes. Acta Paediatr Suppl 1997;143:178–182.
Leeson CPM, Kattenhorn M, Morely R, Lucas A, Deanfield JE . Impact of low birth weight and cardiovascular risk factors on endothelial function in early adult life. Circulation 2001;103:1264–1268.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aucott, S., Donohue, P. & Northington, F. Increased Morbidity in Severe Early Intrauterine Growth Restriction. J Perinatol 24, 435–440 (2004). https://doi.org/10.1038/sj.jp.7211116
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7211116
This article is cited by
-
Exosomes secreted by chemoresistant ovarian cancer cells promote angiogenesis
Journal of Ovarian Research (2021)
-
The frequency of pulmonary hypertension in newborn with intrauterine growth restriction
Scientific Reports (2020)
-
Variants of the OLIG2 Gene are Associated with Cerebral Palsy in Chinese Han Infants with Hypoxic–Ischemic Encephalopathy
NeuroMolecular Medicine (2019)
-
Detection and assessment of brain injury in the growth-restricted fetus and neonate
Pediatric Research (2017)