Abstract
Aim:
To test the effect of orexin-A (hypocretin-1), a neuropeptide synthesized in the lateral hypothalamus and the perifornical area, on the glycinergic inputs and the GABAergic inputs of cardiac vagal neurons (CVN).
Methods:
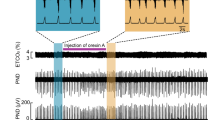
The effects of orexin-A at three concentrations (20 nmol/L, 100 nmol/L, 500 nmol/L) on the glycinergic inputs and the GABAergic inputs were investigated by using retrograde fluorescent labeling of cardiac neurons (CVN) in the nucleus ambiguus (NA) and the voltage patch-clamp technique.
Results:
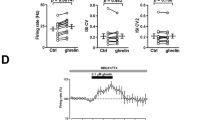
Orexin-A dose-dependently increased the frequency of both the glycinergic and the GABAergic spontaneous inhibitory postsynaptic currents (sIPSC). However, at a lower concentration (20 nmol/L) of orexin-A, although the frequency of the glycinergic sIPSC was significantly increased, the frequency of the GABAergic sIPSC was not significantly changed.
Conclusion:
The glycinergic inputs and the GABAergic inputs have different sensitivities to orexin-A, which suggests that the two kinds of inhibitory inputs might play different roles in the synaptic control of cardiac vagal functions.
Similar content being viewed by others
Article PDF
References
de Lecea L, Kilduff T, Peyron C, Gao X, Foye PE, Danielson PE, et al. The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc Natl Acad Sci USA 1998; 95: 322–7.
Sakurai T, Amemiya A, Ishii M, Matsuzaki I, Chemelli RM, Tanaka H, et al. Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 1998; 92: 573–85.
Burdakov D, Liss B, Ashcroft FM . Orexin excites GABAergic neurons of the arcuate nucleus by activating the sodium—calcium exchanger. J Neurosci 2003; 23: 4951–7.
van den Pol, AN, Gao XB, Obrietan K, Kilduff TS, Belousov AB . Presynaptic and postsynaptic actions and modulation of neuroendocrine neurons by a new hypothalamic peptide, hypocretin/ orexin. J Neurosci 1998; 18: 7962–71.
Wu M, Zhang Z, Leranth C, Xu, C, van den Pol, AN, Alreja M . Hypocretin increases impulse flow in the septohippocampal GABAergic pathway: implications for arousal via a mechanism of hippocampal disinhibition. J Neurosci 2002; 22: 7754–65.
Matsumura K, Tsuchihashi T, Abe I . Central orexin-A augments sympathoadrenal outflow in conscious rabbits. Hypertension 2001; 37: 1382–7.
Samson WK, Gosnell B, Chang JK, Resch ZT, Murphy TC . Cardiovascular regulatory actions of the hypocretins in brain. Brain Res 1999; 831: 248–53.
Shirasaka T, Nakazato M, Matsukura S, Takasaki M, Kannan H . Sympathetic and cardiovascular actions of orexins in conscious rats. Am J Physiol 1999; 277: R1780–5.
Antunes VR, Brailoiu GC, Kwok EH, Scruggs P, Dun NJ . Orexins/ hypocretins excite rat sympathetic preganglionic neurons in vivo and in vitro. Am J Physiol Regul Integr Comp Physiol 2001; 281: R1801–7.
Chen CT, Hwang LL, Chang JK, Dun NJ . Pressor effects of orexins injected intracisternally and to rostral ventrolateral medulla of anesthetized rats. Am J Physiol Regul Integr Comp Physiol 2000; 278: R692–7.
Machado BH, Bonagamba LG, Dun SL, Kwok EH, Dun NJ . Pressor response to microinjection of orexin/hypocretin into rostral ventrolateral medulla of awake rats. Regul Pept 2002; 104: 75–81.
Cheng Z, Powley TL . Nucleus ambiguus projections to cardiac ganglia of rat atria: an anterograde tracing study. J Comp Neurol 2000; 424: 588–606.
Mendelowitz D . Advances in parasympathetic control of heart rate and cardiac function. News Physiol Sci 1999; 14: 155–61.
Wang J, Irnaten M, Mendelowitz D . Characteristics of spontaneous and evoked GABAergic synaptic currents in cardiac vagal neurons in rats. Brain Res 2001; 889: 78–83.
Wang J, Irnaten M, Neff RA, Venkatesan P, Evans C, Loewy AD, et al. Synaptic and neurotransmitter activation of cardiac vagal neurons in the nucleus ambiguus. Ann N Y Acad Sci 2001; 940: 237–46.
Neff RA, Mihalevich M, Mendelowitz D . Stimulation of NTS activates NMDA and non-NMDA receptors in rat cardiac vagal neurons in the nucleus ambiguus. Brain Res 1998; 792: 277–82.
Neff RA, Wang J, Baxi S, Evans C, Mendelowitz D . Respiratory sinus arrhythmia: endogenous activation of nicotinic receptors mediates respiratory modulation of brainstem cardioinhibitory parasympathetic neurons. Circ Res 2003; 93: 565–72.
Wang J, Wang X, Irnaten M, Venkatesan P, Evans C, Baxi S, et al. Endogenous acetylcholine and nicotine activation enhances GABAergic and glycinergic inputs to cardiac vagal neurons. J Neurophysiol 2003; 89: 2473–81.
Ciriello J, de Oliveira CV . Cardiac effects of hypocretin-1 in nucleus ambiguus. Am J Physiol Regul Integr Comp Physiol 2003; 284: R1611–20.
de Oliveira CV, Rosas-Arellano MP, Solano-Flores LP, Ciriello J . Cardiovascular effects of hypocretin-1 in nucleus of the solitary tract. Am J Physiol Heart Circ Physiol 2003; 284: H1369–77.
Smith PM, Connolly BC, Ferguson AV . Microinjection of orexin into the rat nucleus tractus solitarius causes increases in blood pressure. Brain Res 2002; 950: 261–7.
Dergacheva O, Wang X, Huang ZG, Bouairi E, Stephens C, Gorini C, et al. Hypocretin 1 (orexin A) facilitates inhibitory and diminishes excitatory synaptic pathways to cardiac vagal neurons in the nucleus ambiguus. J Pharmacol Exp Ther 2005; 314( 3): 1322–7.
Fujiki N, Yoshida Y, Ripley B, Honda K, Mignot E, Nishino SCA . Changes in CSF hypocretin-1 (orexin A) levels in rats across 24 hours and in response to food deprivation. Neuroreport 2001; 12: 993–7.
John J, Wu MF, Maidment NT, Lam HA, Boehmer LN, Patton M, et al. Developmental changes in CSF hypocretin-1 (orexin-A) levels in normal and genetically narcoleptic Doberman pinschers. J Physiol 2004; 560: 587–92.
Kiyashchenko LI, Mileykovskiy BY, Maidment N, Lam HA, Wu MF, John J, et al. Release of hypocretin (orexin) during waking and sleep states. J Neurosci 2002; 22: 5282–6.
Irnaten M, Neff RA, Wang J, Loewy AD, Mettenleiter TC, Mendelowitz D . Activity of cardiorespiratory networks revealed by transsynaptic virus expressing GFP. J Neurophysiol 2001; 85: 435–8.
Mendelowitz D, Kunze DL . Identification and dissociation of cardiovascular neurons from the medulla for patch clamp analysis. Neurosci Lett 1991; 132: 217–21.
Wang X, Dergacheva O, Griffioen KJ, Huang ZG, Evans C, Gold A, et al. Action of kappa and delta opioid agonists on premotor cardiac vagal neurons in the nucleus ambiguus. Neuroscience 2004; 129: 235–41.
Wang, Y, Ramage, AG . The role of central 5-HT (1A) receptors in the control of B-fibre cardiac and bronchoconstrictor vagal preganglionic neurones in anaesthetized cats. J Physiol (London) 2001; 536: 753–67.
Author information
Authors and Affiliations
Corresponding author
Additional information
Project supported by grants from Shanghai Education and Development Foundation (03SG06) and National Natural Science Foundation of China (No 30470690).
Rights and permissions
About this article
Cite this article
Wang, Jj., Chen, Yh., Li, Ky. et al. Differential sensitivity of GABAergic and glycinergic inputs to orexin-A in preganglionic cardiac vagal neurons of newborn rats. Acta Pharmacol Sin 26, 1442–1447 (2005). https://doi.org/10.1111/j.1745-7254.2005.00231.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00231.x