Abstract
Aim:
To develop a novel, in situ gel system for nasal delivery of scopolamine hydrobromide (SCOP) and study its efficacy on motion sickness.
Methods:
SCOP in situ gels at 0.2%, 0.5%, and 1.0% gellan gum concentration (w/v) were prepared, respectively, and characterized in terms of viscosity, in vitro release, and nasal ciliotoxicity. Single photon emission computing tomography technique was used to evaluate the nasal residence time of gel containing 99mTc tracer. The antimotion sickness efficacy produced by the in situ gel formulation was investigated in rats and compared with those achieved after subcutaneous and oral administration.
Results:
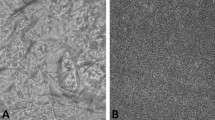
The viscosity of the gellan gum formulations either in solution or in gel increased with increasing concentrations of gellan gum. Its release in vitro was moderate in artificial nasal fluid. The micrographic results showed that in situ gels were safe, without nasal ciliotoxicity. In comparison with phosphate buffer saline, a prolonged radioactivity of 99mTc in the rabbit nasal cavity was observed after administration of the gellan gum formulation. Intranasal SCOP in situ gel at a dose of 100 μg/kg decreased symptoms of motion sickness significantly in comparison with subcutaneous and oral administration (P<0.01).
Conclusion:
SCOP nasal in situ gel is a safe and promising therapeutic alternative to existing medications for motion sickness.
Similar content being viewed by others
Article PDF
References
Putcha, L, Cintron, NM, Tsui, J, Vanderploeg, JM, Kramer, WG, Pharmacokinetics and oral bioavailability of scopolamine in normal subjects. Pharm Res 1989; 6: 481–5.
Shaw, J, Urquhart, J, Programmed systemic delivery by the transdermal route. Trends Pharmacol Sic 1980; 2: 208–11.
Cintron, NM, Chen, Y, A sensitive radioreceptor assay for determining scopolamine concentrations in plasma and urine. J Pharm Sci 1987; 76: 328–32.
Hyde, RW, Tonndorf, J, Chinn, HE, Absorption from the nasal mucous membrane. Ann Otol Rhinol Laryngol 1953; 62: 957–68.
Illum, L, Jorgensen, H, Bisgaard, H, Krogsgaard, O, Rossing, N, Bioadhesive microspheres as a potential nasal drug delivery system. Int J Pharm 1987; 39: 189–99.
Gizurason, S, The relevance of nasal physiology to the design of drug absorption studies. Adv Drug Del Rev 1993; 11: 329–47.
Gurny, R, Boye, T, Ibrahim, H, Ocular therapy with nanoparticulate systems for controlled drug delivery. J Control Release 1985; 2: 353–61.
Miller, SC, Donovan, MD, Effect of Poloxamer 407 gel on the miotic activity of pilocarpine nitrate in rabbits. Int J Pharm 1982; 12: 147–52.
Edsman, K, Carlfors, J, Petersson, R, Rheological evaluation of poloxamer as an in situ gel for ophthalmic use. Eur J Pharm Sci 1998; 6: 105–12.
Rozier, A, Mazuel, C, Grove, J, Plazonnet, B, Functionality testing of gellan gum, a polymeric excipient material for ophthalmic dosage forms. Int J Pharm 1997; 153: 191–8.
Grasdalen, H, Smidsroed, O, Gelation of gellan gum. Carbohydr Polym 1987; 7: 371–93.
Chanrasekaran, R, Puigjaner, LC, Joyce, KL, Arnott, S, Cation interaction in gellan: an X-ray study of the potassium salt. Carbohydr Res 1988; 181: 23–40.
Chanrasekaran, R, Thailambal, VG, The influence of calcium ions, acetate and l-glycerate groups on the gellan double helix. Carbohydr Polym 1990; 12: 431–2.
Sanzgiri, YD, Maschi, S, Crescenzi, V, Callegaro, L, Topp, EM, Stella, VJ, Gellan-based systems for ophthalmic sustained delivery of methylprednisolone. J Control Release 1993; 26: 195–201.
Lorin, MI, Gaerlan, PF, Mandel, ID, Quantitative composition of nasal secretions in normal subjects. J Lab Clin Med 1972; 2: 275–81.
Jiang, XG, Cui, JB, Fang, XL, Wei, Y, Xi, NZ, Toxicity of drugs on nasal mucocilia and the method of its evaluation. Acta Pharm Sin 1995; 30: 848–53.
Gupta, YK, Chaudhary, G, Effect of antiemetic drugs on decrease in gastric emptying in experimental model of motion sickness in rats. Acta Pharmacol Sin 2003; 24: 296–300.
Higuchi, WI, The analysis of data on the medicament release from ointments. J Pharm Sci 1962; 51: 802–4.
Money, K, Motion sickness. Physiol Rev 1970; 50: 1–39.
Klöcker, N, Hanschke, W, Toussaint, S, Verse, T, Scopolamine nasal spray in motion sickness: a randomized, controlled, and crossover study for the comparison of two scopolamine nasal sprays with oral dimenhydrinate and placebo. Eur J Pharm Sci 2001; 13: 227–32.
Nickerson, MT, Paulson, AT, Rheological properties of gellan, k-carrageenan and alginate polysaccharides: effect of potassium and calcium ions on macrostructure assemblages. Carbohydr Polym 2004; 58: 15–24.
Ugwoke, MI, Agu, RU, Jorissen, M, Augustijns, P, Sciot, R, Verbeke, N, et al. Nasal toxicological investigations of Carbopol 971P formulation of apomorphine: effects on ciliary beat frequency of human nasal primary cell culture and in vivo on rabbit nasal mucosa. Eur J Pharm Sci 2000; 9: 387–96.
Marttin, E, Schipper, NGM, Verhoef, JC, Merkus, FWHM, Nasal mucociliary clearance as a factor in nasal drug delivery. Adv Drug Deliv Rev 1998; 29: 13–38.
Puchelle, E, Aug, F, Pham, QT, Bertrand, A, Comparison of three methods for measuring nasal mucociliary clearance in man. Acta Otolaryngol 1981; 91: 297–303.
Schipper, NGM, Verhoef, JC, Merkus, FWHM, The nasal mucociliary clearance: Relevance to nasal drug delivery. Pharm Res 1991; 8: 807–14.
Ingels, K, Van Hoom, V, Obrie, E, Osmanagaoglu, K, A modified technetium-99m isotope test to measure nasal mucocillary transport: comparison with the saccharine-dye test. Eur Arch Otorhinolaryngol 1995; 252: 340–3.
Author information
Authors and Affiliations
Corresponding author
Additional information
Work supported by the National Natural Science Foundation of China (No 30171112).
Rights and permissions
About this article
Cite this article
Cao, Sl., Zhang, Qz. & Jiang, Xg. Preparation of ion-activated in situ gel systems of scopolamine hydrobromide and evaluation of its antimotion sickness efficacy. Acta Pharmacol Sin 28, 584–590 (2007). https://doi.org/10.1111/j.1745-7254.2007.00540.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2007.00540.x
Keywords
This article is cited by
-
Formulation and Development of In Situ Nasal Gelling Systems for Quetiapine Fumarate-Loaded Mucoadhesive Microemulsion
Journal of Pharmaceutical Innovation (2015)