Abstract
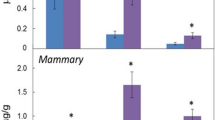
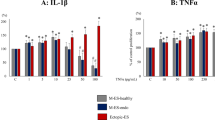
Malignant human breast tumours contain high levels of prostaglandin E2 (PGE2). However, the mechanisms controlling PGE2 production in breast cancer are unknown. This in vitro study investigates the capacity for PGE2 synthesis and metabolism in several human breast cancer cell lines and early passage human breast fibroblasts and seeks to identify potential regulatory factors which may control these pathways. Basal PGE2 production rose up to 30-fold in breast fibroblast lines on addition of exogenous arachidonic acid (10 microM), whereas no such changes were observed in six out of seven cancer cell lines, with the exception of modest increases in MDA-MB-231 cells. Interleukin 1 beta (IL-1 beta) also induced PGE2 production in breast fibroblasts in the presence of excess substrate, consistent with cyclo-oxygenase induction by the cytokine. Under these conditions only Hs578T cells and MDA-MB-231 cells demonstrated large increases in PGE2 in response to IL-1 beta or phorbol ester; no such responses were seen in MCF-7, T47-D, ZR-75-1, BT-20 or CLF-90-1 cells. In the absence of added arachidonate, bradykinin (BK) and endothelin-1 (ET-1), potentiated PGE2 production in IL-1 beta-treated fibroblasts, possibly by mobilising endogenous substrate. PGE2 also stimulated ET-1 production by breast cancer cells. In co-cultures with T47-D cells both basal and stimulated PGE2 production by breast fibroblasts was greatly reduced. This appeared to be due to metabolic inactivation by the cancer cell since T47-D cells readily converted PGE2 to 15-keto-PGE2. This apparent 15-hydroxy-PG dehydrogenase activity was stimulated by TPA and inhibited by cycloheximide. In conclusion, breast fibroblasts, particularly under the influence of inflammatory mediators, provide a potentially rich source for PGE2 production in breast tumours, whereas significant contributions from the epithelial tumour component may be restricted to cancer cells exhibiting an invasive phenotype. Metabolic inactivation by the cancer cells may also play an important role in the regulation of breast tumour PGE2 levels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 24 print issues and online access
$259.00 per year
only $10.79 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schrey, M., Patel, K. Prostaglandin E2 production and metabolism in human breast cancer cells and breast fibroblasts. Regulation by inflammatory mediators. Br J Cancer 72, 1412–1419 (1995). https://doi.org/10.1038/bjc.1995.523
Issue Date:
DOI: https://doi.org/10.1038/bjc.1995.523
This article is cited by
-
Antiproliferative and antiinflammatory coxib–combretastatin hybrids suppress cell cycle progression and induce apoptosis of MCF7 breast cancer cells
Molecular Diversity (2021)
-
Prostaglandin E2 stimulates COX-2 expression via mitogen-activated protein kinase p38 but not ERK in human follicular dendritic cell-like cells
BMC Immunology (2020)
-
The Multifaceted Roles Neutrophils Play in the Tumor Microenvironment
Cancer Microenvironment (2015)
-
Dormancy and growth of metastatic breast cancer cells in a bone-like microenvironment
Clinical & Experimental Metastasis (2015)
-
Tumor-derived TGF-β and prostaglandin E2 attenuate anti-tumor immune responses in head and neck squamous cell carcinoma treated with EGFR inhibitor
Journal of Translational Medicine (2014)