Abstract
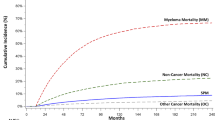
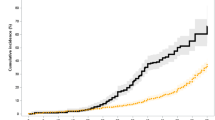
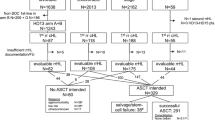
Population-based evidence on second cancer risk following autologous haematopoietic SCT (HCT) is lacking. We quantified second cancer risk for a national, population-based cohort of adult Australians receiving autologous HCT for cancer and notified to the Australasian Bone Marrow Transplant Recipient Registry 1992–2007 (n=7765). Cancer diagnoses and deaths were ascertained by linkage with the Australian Cancer Database and National Death Index. Standardized incidence ratios (SIRs) were calculated and Cox regression models were used to estimate within-cohort risk factors treating death as a competing risk. During a median 2.5 years follow-up, second cancer risk was modestly increased compared with the general population (SIR 1.4, 95% confidence interval 1.2–1.6); significantly elevated risk was also observed for AML/myelodysplastic syndrome (SIR=20.6), melanoma (SIR=2.6) and non-Hodgkin lymphoma (SIR=3.3). Recipients at elevated risk of any second cancer included males, and those transplanted at a younger age, in an earlier HCT era, or for lymphoma or testicular cancer. Male sex, older age (>45 years) and history of relapse after HCT predicted melanoma risk. Transplantation for Hodgkin lymphoma and older age were associated with lung cancer risk. Second malignancies are an important late effect and these results inform and emphasize the need for cancer surveillance in autologous HCT survivors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Nivison-Smith I, Bradstock KF, Dodds AJ, Hawkins PA, Ma DDF, Moore JJ et al. Hematopoietic stem cell transplantation in Australia and New Zealand, 1992-2004. Biol Blood Marrow Trans 2007; 13: 905–912.
Gratwohl A, Baldomero H, Aljurf M, Pasquini MC, Bouzas LF, Yoshimi A et al. Hematopoietic stem cell transplantation. JAMA 2010; 303: 1617–1624.
Martin PJ, Counts GW, Appelbaum FR, Lee SJ, Sanders JE, Deeg HJ et al. Life expectancy in patients surviving more than 5 years after hematopoietic cell transplantation. J Clin Oncol 2010; 28: 1011–1016.
Witherspoon RP, Fisher LD, Schoch G, Martin P, Sullivan KM, Sanders J et al. Secondary cancers after bone marrow transplantation for leukemia or aplastic anemia. N Engl J Med 1989; 321: 784–789.
Lowsky R, Lipton J, Fyles G, Minden M, Meharchand J, Tejpar I et al. Secondary malignancies after bone marrow transplantation in adults. J Clin Oncol 1994; 12: 2187–2192.
Bhatia S, Ramsay N, Steinbuch M, Dusenbery K, Shapiro R, Weisdorf D et al. Malignant neoplasms following bone marrow transplantation. Blood 1996; 87: 3633–3639.
Bhatia S, Louie AD, Bhatia R, O’Donnell MR, Fung H, Kashyap A et al. Solid cancers after bone marrow transplantation. J Clin Oncol 2001; 19: 464–471.
Baker KS, DeFor TE, Burns LJ, Ramsay NKC, Neglia JP, Robison LL . New malignancies after blood or marrow stem-cell transplantation in children and adults: incidence and risk factors. J Clin Oncol 2003; 21: 1352–1358.
Rizzo JD, Curtis RE, Socié G, Sobocinski KA, Gilbert E, Landgren O et al. Solid cancers after allogeneic hematopoietic cell transplantation. Blood 2009; 113: 1175–1183.
Metayer C, Curtis RE, Vose J, Sobocinski KA, Horowitz MM, Bhatia S et al. Myelodysplastic syndrome and acute myeloid leukemia after autotransplantation for lymphoma: a multicenter case-control study. Blood 2003; 101: 2015–2023.
Brown JR, Yeckes H, Friedberg JW, Neuberg D, Kim H, Nadler LM et al. Increasing incidence of late second malignancies after conditioning with cyclophosphamide and total-body irradiation and autologous bone marrow transplantation for non-Hodgkin’s lymphoma. J Clin Oncol 2005; 23: 2208–2214.
Forrest DL, Nevill TJ, Naiman SC, Le A, Brockington DA, Barnett MJ et al. Second malignancy following high-dose therapy and autologous stem cell transplantation: incidence and risk factor analysis. Bone Marrow Transplant 2003; 32: 915–923.
Cohen A, Rovelli A, Merlo DF, van Lint MT, Lanino E, Bresters D et al. Risk for secondary thyroid carcinoma after hematopoietic stem-cell transplantation: an EBMT Late Effects Working Party study. J Clin Oncol 2007; 25: 2449–2454.
Seshadri T, Pintilie M, Kuruvilla J, Keating A, Tsang R, Zadeh S et al. Incidence and risk factors for second cancers after autologous hematopoietic cell transplantation for aggressive non-Hodgkin lymphoma. Leuk Lymphoma 2009; 50: 380–386.
Tarella C, Passera R, Magni M, Benedetti F, Rossi A, Gueli A et al. Risk factors for the development of secondary malignancy after high-dose chemotherapy and autograft, with or without rituximab: a 20-year retrospective follow-up study in patients with lymphoma. J Clin Oncol 2011; 29: 814–824.
Nivison-Smith I, Bradstock KF, Dodds AJ, Hawkins PA, Szer J . Haemopoietic stem cell transplantation in Australia and New Zealand, 1992-2001: progress report from the Australasian Bone Marrow Transplant Recipient Registry. Int Med J 2005; 35: 18–27.
Shimada K, Yokozawa T, Atsuta Y, Kohno A, Maruyama F, Yano K et al. Solid tumors after hematopoietic stem cell transplantation in Japan: incidence, risk factors and prognosis. Bone Marrow Transplant 2005; 36: 115–121.
Krishnan AY, Mei M, Sun C-L, Thomas SH, Teh JB, Kang T et al. Second primary malignancies after autologous hematopoietic cell transplantation for multiple myeloma. Biol Blood Marrow Trans 2012; 19: 260–265.
Fenk R, Neubauer F, Bruns I, Schröder T, Germing U, Haas R et al. Secondary primary malignancies in patients with multiple myeloma treated with high-dose chemotherapy and autologous blood stem cell transplantation. Br J Haem 2012; 156: 683–686.
Danner-Koptik KE, Majhail NS, Brazauskas R, Wang Z, Buchbinder D, Cahn JY et al. Second malignancies after autologous hematopoietic cell transplantation in children. Bone Marrow Transplant 2013; 48: 363–368.
Travis LB, Fosså SD, Schonfeld SJ, McMaster ML, Lynch CF, Storm H et al. Second cancers among 40 576 testicular cancer patients: focus on long-term survivors. J Natl Cancer Inst 2005; 97: 1354–1365.
Kollmannsberger C, Beyer J, Droz JP, Harstrick A, Hartmann JT, Biron P et al. Secondary leukemia following high cumulative doses of etoposide in patients treated for advanced germ cell tumors. J Clin Oncol 1998; 16: 3386–3391.
Sacchi S, Marcheselli L, Bari A, Marcheselli R, Pozzi S, Luminari S et al. Secondary malignancies after treatment for indolent non-Hodgkin’s lymphoma: a 16-year follow-up study. Haematologica 2008; 93: 398–404.
Lens MB, Newton-Bishop JA . An association between cutaneous melanoma and non-Hodgkin's lymphoma: pooled analysis of published data with a review. Ann Oncol 2005; 16: 460–465.
Lenz G, Dreyling M, Schiegnitz E, Haferlach T, Hasford J, Unterhalt M et al. Moderate increase of secondary hematologic malignancies after myeloablative radiochemotherapy and autologous stem-cell transplantation in patients with indolent lymphoma: results of a prospective randomized trial of the German Low Grade Lymphoma Study Group. J Clin Oncol 2004; 22: 4926–4933.
Grulich AE, van Leeuwen MT, Falster MO, Vajdic CM . Incidence of cancers in people with HIV/AIDS compared with immunosuppressed transplant recipients: a meta-analysis. Lancet 2007; 370: 59–67.
Vajdic CM, van Leeuwen MT, Webster AC, McCredie MRE, Stewart JH, Chapman JR et al. Cutaneous melanoma is related to immune suppression in kidney transplant recipients. Cancer Epidemiol Biomarkers Prev 2009; 18: 2297–2303.
Little EG, Eide MJ . Update on the current state of melanoma incidence. Dermatol Clin 2012; 30: 355–361.
Hughes BR, Cunliffe WJ, Bailey CC . Excess benign melanocytic naevi after chemotherapy for malignancy in childhood. BMJ 1989; 299: 88–91.
McGregor JM, JNWN Barker, Macdonald DM . The development of excess numbers of melanocytic naevi in an immunosuppressed identical twin. Clin Exp Dermatol 1991; 16: 131–132.
Pedersen-Bjergaard J, Andersen MK, Christiansen DH . Therapy-related acute myeloid leukemia and myelodysplasia after high-dose chemotherapy and autologous stem cell transplantation. Blood 2000; 95: 3273–3279.
Dong C, Hemminki K . Second primary neoplasms among 53 159 haematolymphoproliferative malignancy patients in Sweden, 1958-1996: a search for common mechanisms. Br J Cancer 2001; 85: 997–1005.
Curtis RE, Freedman DM, Ron E, Ries LAG, Hacker DG, Edwards BK et al. New malignancies among cancer survivors. In:. SEER Cancer Registries, 1973-2000. National Cancer Institute: Bethesda, MD, USA, 2006.
Mailankody S, Pfeiffer RM, Kristinsson SY, Korde N, Bjorkholm M, Goldin LR et al. Risk of acute myeloid leukemia and myelodysplastic syndromes after multiple myeloma and its precursor disease (MGUS). Blood 2011; 118: 4086–4092.
Attal M, Lauwers-Cances V, Marit G, Caillot D, Moreau P, Facon T et al. Lenalidomide maintenance after stem-cell transplantation for multiple myeloma. N Engl J Med 2012; 366: 1782–1791.
McCarthy PL, Owzar K, Hofmeister CC, Hurd DD, Hassoun H, Richardson PG et al. Lenalidomide after stem-cell transplantation for multiple myeloma. N Engl J Med 2012; 366: 1770–1781.
Lorigan P, Radford J, Howell A, Thatcher N . Lung cancer after treatment for Hodgkin's lymphoma: a systematic review. Lancet Oncol 2005; 6: 773–779.
Friedman DL, Rovo A, Leisenring W, Locasciulli A, Flowers MED, Tichelli A et al. Increased risk of breast cancer among survivors of allogeneic hematopoietic cell transplantation: a report from the FHCRC and the EBMT-Late Effect Working Party. Blood 2008; 111: 939–944.
Bishop MM, Lee SJ, Beaumont JL, Andrykowski MA, Rizzo JD, Sobocinski KA et al. The preventive health behaviors of long-term survivors of cancer and hematopoietic stem cell transplantation compared with matched controls. Biol Blood Marrow Trans 2010; 16: 207–214.
Majhail NS, Rizzo JD, Lee SJ, Aljurf M, Atsuta Y, Bonfim C et al. Recommended screening and preventive practices for long-term survivors after hematopoietic cell transplantation. Biol Blood Marrow Trans 2012; 18: 348–371.
Acknowledgements
This work was supported by the National Health and Medical Research Council (APP568775); the Arrow Bone Marrow Transplant Foundation Australia; and support to the ABMTRR from the Commonwealth Department of Health and Ageing. These agencies had no role in the design of the study, collection and analysis of data, or decision to publish. We thank the staff of the hospital SCT units, the state and territory cancer registries and health departments for the use of their data, and the Australian Institute of Health and Welfare for conducting the data linkage. The ABMTRR is grateful to the BMT Network NSW, the Arrow Bone Marrow Transplant Foundation, St Vincent’s Hospital Darlinghurst and the Australian Bone Marrow Donor Registry for their support.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
L Ashton, R Le Marsney (Children's Cancer Institute Australia, New South Wales); A Dodds, J Tan, I Bilmon (St Vincent's Hospital, New South Wales); L Wilcox, I Nivison-Smith, D Aarons, S Tran (ABMTRR); C Vajdic (University of New South Wales, New South Wales); J Gibson, A Johnston (Royal Prince Alfred Hospital, New South Wales); M Greenwood, M Forbes (Royal North Shore Hospital, New South Wales); M Hertzberg, G Huang (Westmead Hospital, New South Wales); A Spencer, J Muirhead (Alfred Hospital, Victoria); J Szer, K Mason, (Royal Melbourne Hospital, Victoria); I Lewis, C To (Royal Adelaide Hospital, South Australia); S Durrant, R Western (Royal Brisbane and Women's Hospital, Queensland); P Cannell, S Buffery (Royal Perth Hospital, Western Australia); T O'Brien, C Oswald, A Nelson (Sydney Children's Hospital, New South Wales); P Shaw, L Pearson (Children's Hospital at Westmead, New South Wales); K Tiedemann, M Scoyne (Royal Children's Hospital Melbourne, Victoria); C Fraser, J Seljak, (Royal Children's Hospital Brisbane, Queensland); C Cole, K Rowland, H Gough, (Princess Margaret Hospital for Children, Western Australia); H Tapp, N Green (Women and Children's Hospital, South Australia). Data collectors: A Moa, J McRae and M Jenkins (Children's Cancer Institute Australia, New South Wales); J Hicks (Royal Melbourne Hospital, Victoria); K Chaplin (Royal Adelaide Hospital, South Australia).
Rights and permissions
About this article
Cite this article
Bilmon, I., Ashton, L., Le Marsney, R. et al. Second cancer risk in adults receiving autologous haematopoietic SCT for cancer: a population-based cohort study. Bone Marrow Transplant 49, 691–698 (2014). https://doi.org/10.1038/bmt.2014.13
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2014.13
Keywords
This article is cited by
-
Male-specific late effects in adult hematopoietic cell transplantation recipients: a systematic review from the Late Effects and Quality of Life Working Committee of the Center for International Blood and Marrow Transplant Research and Transplant Complications Working Party of the European Society of Blood and Marrow Transplantation
Bone Marrow Transplantation (2022)
-
The risk of malignancies in patients receiving hematopoietic stem cell transplantation: a systematic review and meta-analysis
Clinical and Translational Oncology (2020)
-
The role of stem cell transplantation in the management of relapsed follicular lymphoma in the era of targeted therapies
Bone Marrow Transplantation (2019)
-
Long-term implications of autologous HCT for caregiver quality of life: how does the survivor’s health matter?
Supportive Care in Cancer (2019)
-
The Melanoma care study: protocol of a randomised controlled trial of a psycho-educational intervention for melanoma survivors at high risk of developing new primary disease
BMC Psychology (2015)