Abstract
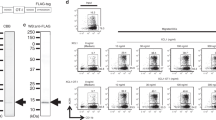
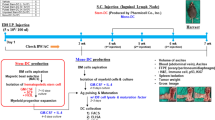
Secondary lymphoid-tissue chemokine (SLC/CCL21) is a CC chemokine that is constitutively expressed in various lymphoid tissues and binds to chemokine receptor CCR7 on mature dendritic cells (DCs) and distinct T-and B-cell sub-populations. In vivo, CCL21 regulates the encounters between DC and T cells and thus is a key regulator of adaptive immune responses. We asked whether CCL21 is able to augment immunogenicity of a DNA-based vaccine against Her2/neu in a Balb/c mouse model with syngeneic Her2/neu+ tumor cells (D2F2/E2). Mice were vaccinated intramuscularly with plasmid DNA (pDNA) on day 1 and boosted on day 15; tumor challenge was performed subcutaneously on day 25. Coexpression of CCL21 and Her-2/neu resulted in induction of a TH1-polarized immune response and substantial improvement of the protective effect of the DNA vaccine. Coexpression of tumor antigen pDNA(Her2/neu) with both pDNA(GM-CSF) and pDNA(CCL21) as adjuvants led to further improvement of protection by the vaccine (70% tumor-free mice on day 35 vs 40% with either adjuvant alone vs 5–10% with tumor antigen alone). Our results show that CCL21 is a potent adjuvant for DNA vaccination, particularly in combination with granulocyte-macrophage colony-stimulating factor (GM-CSF). Clinical use of a pDNA(Her2/neu/CCL21/GM-CSF) vaccine might be particularly promising in minimal residual Her2/neu+ breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Stewart TJ, Smyth MJ . Chemokine-chemokine receptors in cancer immunotherapy. Immunotherapy 2009; 1: 109–127.
Marsland BJ, Battig P, Bauer M, Ruedl C, Lassing U, Beerli R et al. CCL19 and CCL21 induce a potent proinflammatory differentiation program in licensed dendritic cells. Immunity 2005; 22: 493–505.
Friedman RS, Jacobelli J, Krummel MF . Surface-bound chemokines capture and prime T cells for synapse formation. Nat Immunol 2006; 7: 1101–1108.
Worbs T, Mempel TR, Bölter J, von Andrian UH, Förster R . CCR7 ligands stimulate the intranodal motility of T lymphocytes in vivo. J Exp Med 2007; 3: 489–495.
Gurunathan S, Klinman DM, Seder RA . DNA vaccines: immunology, application and optimization. Ann Rev Immunol 2000; 18: 927–974.
Rabinovich GA, Gabrilovich D, Sotomayor EM . Immunosuppressive strategies that are mediated by tumor cells. Annu Rev Immunol 2007; 25: 267–296.
Menard S, Pupa S, Campiglio M, Tagliabue E . Biologic and therapeutic role of HER2 in cancer. Oncogene 2003; 22: 6570–6578.
Cavallo F, Offringa R, van der Burg SH, Forni G, Melief CM . Vaccination for the treatment and prevention of cancer in animal models. Adv Immunol 2006; 90: 175–213.
Romond EH, Perez EA, Bryant J, Suma VJ, Geyer Jr CE, Davidson NE et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med 2005; 353: 1673–1680.
Curigliano G, Spitaleri G, Pietri E, Rescigno M, de Braud F, Cardillo A et al. Breast cancer vaccines: a clinical reality or fairy tale? Ann Oncol 2006; 17: 750–762.
Peoples GE, Holmes JP, Hueman MT, Mittendorf EA, Amin A, Khoo S et al. Combined clinical trial results of a Her2/neu (E75) vaccine for the prevention of recurrence in high-risk breast cancer patients: U.S. Military Cancer Institute Clinical Trials Group Study I-01 and I-02. Clin Cancer Res 2008; 14: 797–803.
Nagata Y, Furugen R, Hiasa A, Ikeda H, Ohta N, Furukawa K et al. Peptides derived from a wild-type murine proto-oncogene-erbB-2/HER2/neu can induce CTL and tumor suppression in syngeneic hosts. J Immunol 1997; 159: 1336–1343.
Piechoski MP, Pilon SA, Wei WZ . Quantitative measurement of anti-ErbB-2 antibody by flow cytometry and ELISA. J Immunol Meth 2002; 259: 33–42.
Stevenson FK, Zhu D, Rice J . New strategies for vaccination and imunomodulation in NHL. Ann Hematol 2001; 80 (Suppl 3): B132–B134.
Maraskovsky E, Brasel K, Teepe M, Roux ER, Lyman S, Shortman K et al. Dramatic increase in the numbers of functionally mature dendritic cells in Flt3 ligand-treated mice: multiple dendritic cell subpopulations identified. J Exp Med 1996; 184: 1953–1962.
Greenland JR, Letvin NL . Chemical adjuvants for plasmid DNA vaccines. Vaccine 2007; 25: 3731–3741.
DellÁgnola C, Biragyn A . Clinical utilisation of chemokines to combat cancer: the double-edged sword. Exp Rev Vaccines 2007; 6: 267–283.
Coscia M, Biragyn A . Cancer immunotherapy with chemoattractant peptides. Sem Cancer Biol 2004; 14: 209–218.
Scheerlinck JP . Genetic adjuvants for DNA vaccines. Vaccine 2001; 19: 2647–2656.
Gunn MD, Kyuwa S, Tam C, Kakiuchi T, Matsuzawa A, Williams LT et al. Mice lacking expression of secondary lymphoid organ chemokine have defects in lymphocyte homing and dendritic cell localization. J Exp Med 1999; 189: 451–460.
Ricart BG, John B, Lee D, Hunter AC, Hammer DA . Dendritic cells distinguish individual chemokine signals through CCR7 and CXCR4. J Immunol 2011; 186: 53–61.
Wei WZ, Shi WP, Galy A, Lichlyter D, Hernandez S, Groner B et al. Protection against mammary tumor growth by vaccination with full-length, modified human ErbB-2 DNA. Int J Cancer 1999; 81: 748–754.
Kiessling R, Wei WZ, Herrmann F, Lindencrona JA, Choudhury A, Kono K et al. Cellular immunity to the Her-2/neu protooncogene. Adv Cancer Res 2002; 85: 101–144.
Lindencrona JA, Preise S, Kammertöns T, Schüler T, Piechocki M, Wei WZ et al. CD4+ T cell-mediated Her-2/neu–specific tumor rejection in the absence of B cells. Int J Cancer 2004; 109: 259–264.
Pardoll DM . Paracrine cytokine adjuvants in cancer immunotherapy. Annu Rev Immunol 1995; 13: 399–415.
Dranoff G, Jaffee E, Lazenby A, Golumbek P, Levitsky H, Brose K et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci USA 1993; 90: 3539–3543.
Braun SE, Chen K, Foster RG, Kim CH, Hromas R, Kaplan MH et al. The CC chemokine CKb- 11/MIP-3b/ELC/Exodus 3 mediates tumor rejection of murine breast cancer cells through NK cells. J Immunol 2000; 164: 4025–4031.
Beauvillain C, Cunin P, Doni A, Scotet M, Jaillon S, Loiry ML et al. CCR7 is involved in the migration of neutrophils to lymph nodes. Blood 2011; 117: 1196–1204.
Gunn MD, Tangemann K, Tam C, Cyster JG, Rosen SD, Williams LT . A chemokine expressed in lymphoid high endothelial venules promotes the adhesion and chemotaxis of naive lymphocytes. Proc Natl Acad Sci USA 1998; 95: 258–263.
Martin-Fontecha AS, Sebastiani S, Höpken UE, Uguccioni M, Lipp M, Lanzavecchia A et al. Regulation of dendritic cell migration to the draining lymph node: impact on T lymphocyte traffic and priming. J Exp Med 2003; 198: 615–621.
Sallusto F, Palermo B, Lenig D, Miettinen M, Matikaine S, Julkunen I et al. Distinct patterns and kinetics of chemokine production regulate dendritic cell function. Eur J Immunol 1999; 29: 1617–1625.
Radstake TR, van der Voort R, ten Brummelhuis M, de Waal Melefijt M, Looman M, Figdor CG et al. Increased expression of CCL18, CCL19 and CCL17 by dendritic cells from patients with rheumatoid arthritis, and regulation by Fc gamma receptors. Ann Rheum Dis 2005; 64: 359–367.
Sharma S, Stolin M, Luo J, Strieter RM, Burdick M, Zhu LX et al. Secondary lymphoid tissue chemokine mediates T cell-dependent antitumor responses in vivo. J Immunol 2000; 164: 4558–4563.
Sharma S, Stolina M, Zhu L, Lin Y, Batra R, Huang M et al. Secondary lymphoid organ chemokine reduces pulmonary tumor burden in spontaneous murine bronchoalveolar cell carcinoma. Cancer Res 2001; 61: 6406–6412.
Yang SC, Batra RK, Hillinger S, Reckamp KL, Strieter RM, Dubinett SM et al. Intrapulmonary administration of CCL21 gene-modified dendritic cells reduces tumor burden in spontaneous murine bronchoalveolar cell carcinoma. Cancer Res 2006; 66: 3205–3213.
Yang SC, Hillinger S, Riedl K, Zhang L, Zhu L, Huang M et al. Intratumoral administration of dendritic cells overexpressing CCL21 generates systemic antitumor responses and confers tumor immunity. Cancer Res 2004; 10: 2891–2901.
Qin H, Zhou C, Wang D, Ma W, Liang X, Lin C et al. Enhancement of antitumour immunity by a novel chemotactic antigen DNA vaccine encoding chemokines and multiepitopes of prostate-tumour-associated antigens. Immunology 2006; 117: 419–430.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Nguyen-Hoai, T., Baldenhofer, G., Sayed Ahmed, M. et al. CCL21 (SLC) improves tumor protection by a DNA vaccine in a Her2/neu mouse tumor model. Cancer Gene Ther 19, 69–76 (2012). https://doi.org/10.1038/cgt.2011.69
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2011.69
Keywords
This article is cited by
-
Activated T cell therapy targeting glioblastoma cancer stem cells
Scientific Reports (2023)
-
A formulated poly (I:C)/CCL21 as an effective mucosal adjuvant for gamma-irradiated influenza vaccine
Virology Journal (2021)
-
HER2-Positive Breast Cancer Immunotherapy: A Focus on Vaccine Development
Archivum Immunologiae et Therapiae Experimentalis (2020)
-
Synergistic effect of programmed cell death protein 1 blockade and secondary lymphoid tissue chemokine in the induction of anti-tumor immunity by a therapeutic cancer vaccine
Archives of Virology (2017)
-
CCL4 as an adjuvant for DNA vaccination in a Her2/neu mouse tumor model
Cancer Gene Therapy (2016)