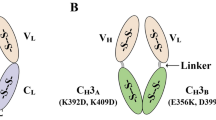
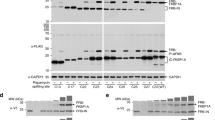
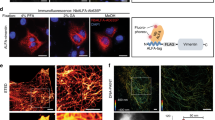
Abstract
The wild-type human Fas-associated death domain (FADD) protein was expressed as a His-tag fusion protein in Escherichia coli. Recombinant FADD proteins were purified under the denatured condition. After denatured protein purification, it was refolded and obtained at a yield of about 23 mg/L. Purified FADD exhibited as a homogenous band corresponding to the molecular weight of 31 kDa. Immunization of rabbits against the refolded FADD protein was allowed the production of high titre polyclonal antiserum. This new polyclonal antibody could recognize recombinant FADD protein in Western blot. Immunoreactivity was also observed in immunofluorescence assay. The low cost polyclonal antiserum was applicable to extensive detection of FADD in various immunoassays.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marikar, F., Ma, D., Ye, J. et al. Expression of Recombinant Human FADD, Preparation of Its Polyclonal Antiserum and the Application in Immunoassays. Cell Mol Immunol 5, 471–474 (2008). https://doi.org/10.1038/cmi.2008.59
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2008.59
Keywords
This article is cited by
-
Identification of S-nitrosylation of proteins of Helicobacter pylori in response to nitric oxide stress
The Journal of Microbiology (2011)