Abstract
Background/Objective:
One of the speculated mechanisms underlying fetal origin hypothesis of breast cancer is the possible influence of maternal environment on epigenetic regulation, such as changes in DNA methylation of the insulin-like growth factor-2 (IGF2) gene. The aim of the study is to investigate the relationship between folate, vitamin B12 and methylation of the IGF2 gene in maternal and cord blood.
Subjects/Methods:
We conducted a cross-sectional study to measure methylation patterns of IGF2 in promoters 2 (P2) and promoters 3 (P3).
Results:
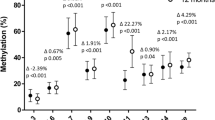
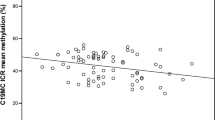
The percentage of methylation in IGF2 P3 was higher in maternal blood than in cord blood (P<0.0001), whereas the methylation in P2 was higher in cord blood than in maternal blood (P=0.016). P3 methylation was correlated between maternal and cord blood (P<0.0001), but not P2 (P=0.06). The multivariate linear regression model showed that methylation patterns of both promoters in cord blood were not associated with serum folate levels in either cord or maternal blood, whereas the P3 methylation patterns were associated with serum levels of vitamin B12 in mother's blood (mean change (MC)=−0.22, P=0.0014). Methylation patterns in P2 of maternal blood were associated with serum levels of vitamin B12 in mother's blood (MC=−0.23, P=0.012), exposure to passive smoking (MC=0.46, P=0.034) and mother's weight gain during pregnancy (MC=0.23, P=0.019).
Conclusions:
The study suggests that environment influences methylation patterns in maternal blood, and then the maternal patterns influence the methylation status and levels of folate and vitamin B12 in cord blood.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bates P, Fisher R, Ward A, Richardson L, Hill DJ, Graham CF (1995). Mammary cancer in transgenic mice expressing insulin-like growth factor II (IGF-II). Br J Cancer 72, 1189–1193.
Beeghly AC, Katsaros D, Wiley AL, Rigault de la Longrais IA, Prescott AT, Chen H et al. (2007). IGF-II promoter methylation and ovarian cancer prognosis. J Cancer Res Clin Oncol 133, 713–723.
Cox GF, Burger J, Lip V, Mau UA, Sperling K, Wu BL et al. (2002). Intracytoplasmic sperm injection may increase the risk of imprinting defects. Am J Hum Genet 71, 162–164.
DeBaun MR, Niemitz EL, Feinberg AP (2003). Association of in vitro fertilization with Beckwith-Wiedemann syndrome and epigenetic alterations of LIT1 and H19. Am J Hum Genet 72, 156–160.
Engstrom W, Shokrai A, Otte K, Granerus M, Gessbo A, Bierke P et al. (1998). Transcriptional regulation and biological significance of the insulin like growth factor II gene. Cell Prolif 31, 173–189.
Holthuizen P, Van Dijk MA, Rodenburg RJ, Koonen-Reemst AM, Sussenbach JS (1993). Transcriptional regulation of the major promoters of the human IGF-II gene. Mol Reprod Dev 35, 391–393.
Issa JP, Vertino PM, Boehm CD, Newsham IF, Baylin SB (1996). Switch from monoallelic to biallelic human IGF2 promoter methylation during aging and carcinogenesis. Proc Natl Acad Sci USA 93, 11757–11762.
Lehti KK (1989). Iron, folic acid and zinc intakes and status of low socio-economic pregnant and lactating Amazonian women. Eur J Clin Nutr 43, 505–513.
Lillycrop KA, Phillips ES, Jackson AA, Hanson MA, Burdge GC (2005). Dietary protein restriction of pregnant rats induces and folic acid supplementation prevents epigenetic modification of hepatic gene expression in the offspring. J Nutr 135, 1382–1386.
McCann AH, Miller N, O’Meara A, Pedersen I, Keogh K, Gorey T et al. (1996). Biallelic expression of the IGF2 gene in human breast disease. Hum Mol Genet 5, 1123–1127.
Michels KB, Xue F (2006). Role of birthweight in the etiology of breast cancer. Int J Cancer 119, 2007–2025.
Moverare-Skrtic S, Mellstrom D, Vandenput L, Ehrich M, Ohlsson C (2009). Peripheral blood leukocyte distribution and body mass index are associated with the methylation pattern of the androgen receptor promoter. Endocrine 35, 204–210.
Nielsen FC, Christiansen J (1992). Endonucleolysis in the turnover of insulin-like growth factor II mRNA. J Biol Chem 267, 19404–19411.
Nielsen FC, Christiansen J (1995). Posttranscriptional regulation of insulin-like growth factor II mRNA. Scand J Clin Lab Invest Suppl 220, 37–46.
Ong K, Kratzsch J, Kiess W, Costello M, Scott C, Dunger D (2000). Size at birth and cord blood levels of insulin, insulin-like growth factor I (IGF-I), IGF-II, IGF-binding protein-1 (IGFBP-1), IGFBP-3, and the soluble IGF-II/mannose-6-phosphate receptor in term human infants. The ALSPAC Study Team. Avon Longitudinal Study of Pregnancy and Childhood. J Clin Endocrinol Metab 85, 4266–4269.
Orstavik KH, Eiklid K, van der Hagen CB, Spetalen S, Kierulf K, Skjeldal O et al. (2003). Another case of imprinting defect in a girl with Angelman syndrome who was conceived by intracytoplasmic semen injection. Am J Hum Genet 72, 218–219.
Reik W, Walter J (2001). Genomic imprinting: parental influence on the genome. Nat Rev Genet 2, 21–32.
Sun FL, Dean WL, Kelsey G, Allen ND, Reik W (1997). Transactivation of Igf2 in a mouse model of Beckwith-Wiedemann syndrome. Nature 389, 809–815.
Thoradeniya T, Wickremasinghe R, Ramanayake R, Atukorala S (2006). Low folic acid status and its association with anaemia in urban adolescent girls and women of childbearing age in Sri Lanka. Br J Nutr 95, 511–516.
Trichopoulos D (1990). Hypothesis: does breast cancer originate in utero? Lancet 335, 939–940.
Van den Veyver IB (2002). Genetic effects of methylation diets. Annu Rev Nutr 22, 255–282.
van Dijk MA, van Schaik FM, Bootsma HJ, Holthuizen P, Sussenbach JS (1991). Initial characterization of the four promoters of the human insulin-like growth factor II gene. Mol Cell Endocrinol 81, 81–94.
van Roozendaal CE, Gillis AJ, Klijn JG, van Ooijen B, Claassen CJ, Eggermont AM et al. (1998). Loss of imprinting of IGF2 and not H19 in breast cancer, adjacent normal tissue and derived fibroblast cultures. FEBS Lett 437, 107–111.
Vobecky JS, Vobecky J, Shapcott D, Demers PP, Blanchard R, Fisch C (1985). The vitamin status of infants in a free living population. Int J Vitam Nutr Res 55, 205–216.
Waterland RA, Jirtle RL (2003). Transposable elements: targets for early nutritional effects on epigenetic gene regulation. Mol Cell Biol 23, 5293–5300.
Wolff GL, Kodell RL, Moore SR, Cooney CA (1998). Maternal epigenetics and methyl supplements affect agouti gene expression in Avy/a mice. FASEB J 12, 949–957.
Wu G, Bazer FW, Cudd TA, Meininger CJ, Spencer TE (2004). Maternal nutrition and fetal development. J Nutr 134, 2169–2172.
Wu HK, Squire JA, Catzavelos CG, Weksberg R (1997). Relaxation of imprinting of human insulin-like growth factor II gene, IGF2, in sporadic breast carcinomas. Biochem Biophys Res Commun 235, 123–129.
Acknowledgements
We thank all the participants in this study and all the doctors and nurses of Houzhai Center Hospital. This publication was made possible by CTSA Grant number UL1 RR024139 from the National Center for Research Resources (NCRR), a component of the NIH and NHL roadmap for medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of NCRR. This research is also supported by Fogarty training Grants 1D43TW008323-01 and 1D43TW007864-01 from the NIH, and by the Grant 2009A330005 from the Education Department of Henan Province, China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Contributors: YB, HY, CZ, TZ, SM, and YZ designed the study; YB, HY, FL, XG, QZ, GW, ZL, and YZ collected the data; YB, HY, FL, GW, ZL, and YZ performed the analyses; YB and YZ wrote the first draft of the manuscript; and all authors reviewed and contributed to the final draft.
Rights and permissions
About this article
Cite this article
Ba, Y., Yu, H., Liu, F. et al. Relationship of folate, vitamin B12 and methylation of insulin-like growth factor-II in maternal and cord blood. Eur J Clin Nutr 65, 480–485 (2011). https://doi.org/10.1038/ejcn.2010.294
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2010.294
Keywords
This article is cited by
-
Associations of circulating folate, vitamin B12 and homocysteine concentrations in early pregnancy and cord blood with epigenetic gestational age: the Generation R Study
Clinical Epigenetics (2021)
-
Association of leukocyte DNA methylation changes with dietary folate and alcohol intake in the EPIC study
Clinical Epigenetics (2019)
-
Identifying and correcting epigenetics measurements for systematic sources of variation
Clinical Epigenetics (2018)
-
Maternal vitamin D, DNA methylation at imprint regulatory regions and offspring weight at birth, 1 year and 3 years
International Journal of Obesity (2018)