Abstract
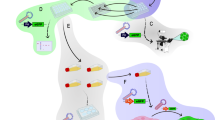
In spite of advances in conventional four-plasmid transient transfection methods and development of inducible stable production cell lines, production of replication-defective lentiviral vectors in clinical scale has been challenging. Baculovirus technology offers an alternative to scalable virus production as a result of fast and easy production of baculoviruses, efficient transduction of mammalian cells and safety of the baculoviruses. As a first step toward scalable lentiviral production system, we have constructed four recombinant baculoviruses: the BAC-transfer virus expresses green fluorescent protein (GFP) as a transgene and BAC-gag-pol, BAC-vesicular stomatitis virus glycoprotein G and BAC-rev express all elements required for a safe lentivirus vector generation. Following 293T cell transduction with recombinant baculoviruses functional lentiviruses were produced. Different baculovirus concentrations, mediums and transduction times were used to find optimal conditions for lentivirus production. The unconcentrated lentiviral titers in cell culture mediums were on average 2.5 × 106 TU ml−1, which are comparable to titers of the lentiviruses produced by conventional four-plasmid methods. Lentiviruses produced by baculovirus method transduced HeLa cells and showed sustained GFP expression. No evidence of the formation of replication competent lentiviruses was detected by p24 enzyme-linked immunosorbent assay. Our results show that baculoviruses are an attractive alternative for the production of lentiviruses in mammalian cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lu X, Humeau L, Slepushkin V, Binder G, Yu Q, Slepushkina T et al. Safe two-plasmid production for the first clinical lentivirus vector that achieves >99% transduction in primary cells using a one-step protocol. J Gene Med 2004; 6: 963–973.
Follenzi A, Naldini L . Generation of HIV-1 derived lentiviral vectors. Methods Enzymol 2002; 346: 454–465.
Tiscornia G, Singer O, Verma IM . Production and purification of lentiviral vectors. Nat Protoc 2006; 1: 241–245.
Geraerts M, Michiels M, Baekelandt V, Debyser Z, Gijsbers R . Upscaling of lentiviral vector production by tangential flow filtration. J Gene Med 2005; 7: 1299–1310.
Segura MM, Garnier A, Durocher Y, Coelho H, Kamen A . Production of lentiviral vectors by large-scale transient transfection of suspension cultures and affinity chromatography purification. Biotechnol Bioeng 2007; 98: 789–799.
Kafri T, van PH, Ouyang L, Gage FH, Verma IM . A packaging cell line for lentivirus vectors. J Virol 1999; 73: 576–584.
Farson D, Witt R, McGuinness R, Dull T, Kelly M, Song J et al. A new-generation stable inducible packaging cell line for lentiviral vectors. Hum Gene Ther 2001; 12: 981–997.
Pacchia AL, Adelson ME, Kaul M, Ron Y, Dougherty JP . An inducible packaging cell system for safe, efficient lentiviral vector production in the absence of HIV-1 accessory proteins. Virology 2001; 282: 77–86.
Xu K, Ma H, McCown TJ, Verma IM, Kafri T . Generation of a stable cell line producing high-titer self-inactivating lentiviral vectors. Mol Ther 2001; 3: 97–104.
Haselhorst D, Kaye JF, Lever AM . Development of cell lines stably expressing human immunodeficiency virus type 1 proteins for studies in encapsidation and gene transfer. J Gen Virol 1998; 79 (Part 2): 231–237.
Cronin J, Zhang XY, Reiser J . Altering the tropism of lentiviral vectors through pseudotyping. Curr Gene Ther 2005; 5: 387–398.
Burns JC, Friedmann T, Driever W, Burrascano M, Yee JK . Vesicular stomatitis virus G glycoprotein pseudotyped retroviral vectors: concentration to very high titer and efficient gene transfer into mammalian and nonmammalian cells. Proc Natl Acad Sci USA 1993; 90: 8033–8037.
Ni Y, Sun S, Oparaocha I, Humeau L, Davis B, Cohen R et al. Generation of a packaging cell line for prolonged large-scale production of high-titer HIV-1-based lentiviral vector. J Gene Med 2005; 7: 818–834.
Strang BL, Ikeda Y, Cosset FL, Collins MK, Takeuchi Y . Characterization of HIV-1 vectors with gammaretrovirus envelope glycoproteins produced from stable packaging cells. Gene Therapy 2004; 11: 591–598.
Strang BL, Takeuchi Y, Relander T, Richter J, Bailey R, Sanders DA et al. Human immunodeficiency virus type 1 vectors with alphavirus envelope glycoproteins produced from stable packaging cells. J Virol 2005; 79: 1765–1771.
Sena-Esteves M, Tebbets JC, Steffens S, Crombleholme T, Flake AW . Optimized large-scale production of high titer lentivirus vector pseudotypes. J Virol Methods 2004; 122: 131–139.
Kumar M, Bradow BP, Zimmerberg J . Large-scale production of pseudotyped lentiviral vectors using baculovirus GP64. Hum Gene Ther 2003; 14: 67–77.
Kotsopoulou E, Kim VN, Kingsman AJ, Kingsman SM, Mitrophanous KA . A Rev-independent human immunodeficiency virus type 1 (HIV-1)-based vector that exploits a codon-optimized HIV-1 gag-pol gene. J Virol 2000; 74: 4839–4852.
Koldej R, Cmielewski P, Stocker A, Parsons DW, Anson DS . Optimisation of a multipartite human immunodeficiency virus based vector system; control of virus infectivity and large-scale production. J Gene Med 2005; 7: 1390–1399.
Wu X, Wakefield JK, Liu H, Xiao H, Kralovics R, Prchal JT et al. Development of a novel trans-lentiviral vector that affords predictable safety. Mol Ther 2000; 2: 47–55.
O'reilly DR, Miller LK, Luckov VA . Baculovirus Expression Vectors. A laboratory Manual. Oxford University Press: New York, 2004.
Airenne KJ, Mahonen AJ, Laitinen OH, Yla-Herttuala S . Baculovirus-mediated gene transfer: an evolving new concept. In: Templeton NS (ed). Gene and Cell Therapy. Marcel Dekker Inc.: New York, NY, 2004, pp 181–197.
Poomputsa K, Kittel C, Egorov A, Ernst W, Grabherr R . Generation of recombinant influenza virus using baculovirus delivery vector. J Virol Methods 2003; 110: 111–114.
Cheshenko N, Krougliak N, Eisensmith RC, Krougliak VA . A novel system for the production of fully deleted adenovirus vectors that does not require helper adenovirus. Gene Therapy 2001; 8: 846–854.
Sollerbrant K, Elmen J, Wahlestedt C, Acker J, Leblois-Prehaud H, Latta-Mahieu M et al. A novel method using baculovirus-mediated gene transfer for production of recombinant adeno-associated virus vectors. J Gen Virol 2001; 82: 2051–2060.
Huang KS, Lo WH, Chung YC, Lai YK, Chen CY, Chou ST et al. Combination of baculovirus-mediated gene delivery and packed-bed reactor for scalable production of adeno-associated virus. Hum Gene Ther 2007; 18: 1161–1170.
Chen YH, Wu JC, Wang KC, Chiang YW, Lai CW, Chung YC et al. Baculovirus-mediated production of HDV-like particles in BHK cells using a novel oscillating bioreactor. J Biotechnol 2005; 118: 135–147.
Naldini L, Blomer U, Gallay P, Ory D, Mulligan R, Gage FH et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector. Science 1996; 272: 263–267.
Sastry L, Xu Y, Johnson T, Desai K, Rissing D, Marsh J et al. Certification assays for HIV-1-based vectors: frequent passage of gag sequences without evidence of replication-competent viruses. Mol Ther 2003; 8: 830–839.
Scott MJ, Modha SS, Rhodes AD, Broadway NM, Hardwicke PI, Zhao HJ et al. Efficient expression of secreted proteases via recombinant BacMam virus. Protein Expr Purif 2007; 52: 104–116.
Hofmann C, Sandig V, Jennings G, Rudolph M, Schlag P, Strauss M . Efficient gene transfer into human hepatocytes by baculovirus vectors. Proc Natl Acad Sci USA 1995; 92: 10099–10103.
Burges HD, Croizier G, Huger J . A review of safety tests on baculoviruses. Entomophaga 1980; 25: 329–340.
Urabe M, Ding C, Kotin RM . Insect cells as a factory to produce adeno-associated virus type 2 vectors. Hum Gene Ther 2002; 13: 1935–1943.
Mahonen AJ, Airenne KJ, Purola S, Peltomaa E, Kaikkonen MU, Riekkinen MS et al. Post-transcriptional regulatory element boosts baculovirus-mediated gene expression in vertebrate cells. J Biotechnol 2007; 131: 1–8.
Hsu CS, Ho YC, Wang KC, Hu YC . Investigation of optimal transduction conditions for baculovirus-mediated gene delivery into mammalian cells. Biotechnol Bioeng 2004; 88: 42–51.
Rodrigues T, Carrondo MJ, Alves PM, Cruz PE . Purification of retroviral vectors for clinical application: biological implications and technological challenges. J Biotechnol 2007; 127: 520–541.
Dull T, Zufferey R, Kelly M, Mandel RJ, Nguyen M, Trono D et al. A third-generation lentivirus vector with a conditional packaging system. J Virol 1998; 72: 8463–8471.
Miyoshi H, Blomer U, Takahashi M, Gage FH, Verma IM . Development of a self-inactivating lentivirus vector. J Virol 1998; 72: 8150–8157.
Sinn PL, Sauter SL, McCray Jr PB . Gene therapy progress and prospects: development of improved lentiviral and retroviral vectors—design, biosafety, and production. Gene Therapy 2005; 12: 1089–1098.
Gheysen D, Jacobs E, de FF, Thiriart C, Francotte M, Thines D et al. Assembly and release of HIV-1 precursor Pr55gag virus-like particles from recombinant baculovirus-infected insect cells. Cell 1989; 59: 103–112.
Nermut MV, Hockley DJ, Jowett JB, Jones IM, Garreau M, Thomas D . Fullerene-like organization of HIV gag-protein shell in virus-like particles produced by recombinant baculovirus. Virology 1994; 198: 288–296.
Jones IM, Morikawa Y . The molecular basis of HIV capsid assembly. Rev Med Virol 1998; 8: 87–95.
Makinen PI, Koponen JK, Karkkainen AM, Malm TM, Pulkkinen KH, Koistinaho J et al. Stable RNA interference: comparison of U6 and H1 promoters in endothelial cells and in mouse brain. J Gene Med 2006; 8: 433–441.
Airenne KJ, Hiltunen MO, Turunen MP, Turunen AM, Laitinen OH, Kulomaa MS et al. Baculovirus-mediated periadventitial gene transfer to rabbit carotid artery. Gene Therapy 2000; 7: 1499–1504.
Airenne KJ, Peltomaa E, Hytonen VP, Laitinen OH, Yla-Herttuala S . Improved generation of recombinant baculovirus genomes in Escherichia coli. Nucleic Acids Res 2003; 31: e101.
Cha HJ, Gotoh T, Bentley WE . Simplification of titer determination for recombinant baculovirus by green fluorescent protein marker. Biotechniques 1997; 23: 782, 784, 786.
Acknowledgements
We thank Miia Roschier, Tarja Taskinen, Erik Peltomaa, Riikka Eisto, Joonas Malinen, Anne Martikainen and Anneli Miettinen for technical assistance, Dr Olli Laitinen, Minna Kaikkonen and Petri Mäkinen for their invaluable discussions, and Dr Jani Räty and Dr Roseanne Girnary for reviewing the manuscript. This study was supported by Ark Therapeutics Group Plc and EU Clinigene (LSHB-CT-2006-018933) Consortium Flexibility Funds.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lesch, H., Turpeinen, S., Niskanen, E. et al. Generation of lentivirus vectors using recombinant baculoviruses. Gene Ther 15, 1280–1286 (2008). https://doi.org/10.1038/gt.2008.76
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2008.76
Keywords
This article is cited by
-
Baculovirus: an Insect-derived Vector for Diverse Gene Transfer Applications
Molecular Therapy (2013)
-
Enhanced pseudotyping efficiency of HIV-1 lentiviral vectors by a rabies/vesicular stomatitis virus chimeric envelope glycoprotein
Gene Therapy (2012)
-
Production and purification of lentiviral vectors generated in 293T suspension cells with baculoviral vectors
Gene Therapy (2011)
-
Development of a Hybrid Baculoviral Vector for Sustained Transgene Expression
Molecular Therapy (2009)
-
(Strept)avidin-displaying lentiviruses as versatile tools for targeting and dual imaging of gene delivery
Gene Therapy (2009)