Abstract
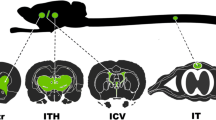
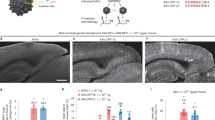
We recently demonstrated that axonal transport of adeno-associated virus (AAV) is serotype-dependent. Thus, AAV serotype 2 (AAV2) is anterogradely transported (e.g., from cell bodies to nerve terminals) in both rat and non-human primate (NHP) brain. In contrast, AAV serotype 6 (AAV6) is retrogradely transported from terminals to neuronal cell bodies in the rat brain. However, the directionality of axonal transport of AAV6 in the NHP brain has not been determined. In this study, two Cynomolgus macaques received an infusion of AAV6 harboring green fluorescent protein (GFP) into the striatum (caudate and putamen) by magnetic resonance (MR)-guided convection-enhanced delivery. One month after infusion, immunohistochemical staining of brain sections revealed a striatal GFP expression that corresponded well with MR signal observed during gene delivery. As shown previously in rats, GFP expression was detected throughout the prefrontal, frontal and parietal cortex, as well as the substantia nigra pars compacta and thalamus, indicating retrograde transport of the vector in NHP. AAV6-GFP preferentially transduced neurons, although a few astrocytes were also transduced. Transduction of non-neuronal cells in the brain was associated with the upregulation of the major histocompatibility complex-II and lymphocytic infiltration as previously observed with AAV1 and AAV9. This contrasts with highly specific neuronal transduction in the rat brain. Retrograde axonal transport of AAV6 from a single striatal infusion permits efficient transduction of cortical neurons in significant tissue volumes that otherwise would be difficult to achieve.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hennig AK, Levy B, Ogilvie JM, Vogler CA, Galvin N, Bassnett S et al. Intravitreal gene therapy reduces lysosomal storage in specific areas of the CNS in mucopolysaccharidosis VII mice. J Neurosci 2003; 23: 3302–3307.
Ciesielska A, Mittermeyer G, Hadaczek P, Kells AP, Forsayeth J, Bankiewicz KS . Anterograde axonal transport of AAV2-GDNF in rat basal ganglia. Mol Ther 2011; 19: 922–927.
Kells AP, Hadaczek P, Yin D, Bringas J, Varenika V, Forsayeth J et al. Efficient gene therapy-based method for the delivery of therapeutics to primate cortex. Proc Natl Acad Sci USA 2009; 106: 2407–2411.
Hadaczek P, Kohutnicka M, Krauze MT, Bringas J, Pivirotto P, Cunningham J et al. Convection-enhanced delivery of adeno-associated virus type 2 (AAV2) into the striatum and transport of AAV2 within monkey brain. Hum Gene Ther 2006; 17: 291–302.
Salegio EA, Samaranch L, Kells AP, Mittermeyer G, San Sebastian W, Zhou S et al. Axonal transport of adeno-associated viral vectors is serotype-dependent. Gene Therapy 2013; 20: 348–352.
Hadaczek P, Forsayeth J, Mirek H, Munson K, Bringas J, Pivirotto P et al. Transduction of nonhuman primate brain with adeno-associated virus serotype 1: vector trafficking and immune response. Hum Gene Ther 2009; 20: 225–237.
Bobo R, Laske D, Akbasak A, Morrison P, Dedrick R, Oldfield E . Convection-enhanced delivery of macromolecules in the brain. Proc Natl Acad Sci USA 1994; 91: 2076–2080.
Hadaczek P, Kohutnicka M, Krauze MT, Bringas J, Pivirotto P, Cunningham J et al. Convection-enhanced delivery of adeno-associated virus type 2 (AAV2) into the striatum and transport of AAV2 within monkey brain. Hum Gene Ther 2006; 17: 291–302.
Fiandaca M, Forsayeth J, Dickinson P, Bankiewicz K . Image-guided convection-enhanced delivery platform in the treatment of neurological diseases. Neurotherapeutics 2008; 5: 123–127.
Salegio EA, Samaranch L, Kells AP, Forsayeth J, Bankiewicz K . Guided delivery of adeno-associated viral vectors into the primate brain. Adv Drug Deliv Rev 2012; 64: 598–604.
Hadaczek P, Yamashita Y, Mirek H, Tamas L, Bohn MC, Noble C et al. The ‘perivascular pump’ driven by arterial pulsation is a powerful mechanism for the distribution of therapeutic molecules within the brain. Mol Ther 2006; 14: 69–78.
Chamberlin NL, Du B, de Lacalle S, Saper CB . Recombinant adeno-associated virus vector: use for transgene expression and anterograde tract tracing in the CNS. Brain Res 1998; 793: 169–175.
Salegio EA, Samaranch L, Kells AP, Mittermeyer G, San Sebastian W, Zhou S et al. Axonal transport of adeno-associated viral vectors is serotype-dependent. Gene Therapy 2012; 20: 348–352.
Ciesielska A, Hadaczek P, Mittermeyer G, Zhou S, Wright JF, Bankiewicz KS et al. Cerebral infusion of AAV9 vector-encoding non-self proteins can elicit cell-mediated immune responses. Mol Ther 2013; 21: 158–166.
The Huntington's Disease Collaborative Research Group. A novel gene containing a trinucleotide repeat that is expanded and unstable on Huntington’s disease chromosomes. Cell 1993; 72: 971–983.
Walker FO . Huntington’s disease. Lancet 2007; 369: 218–228.
Ross CA, Tabrizi SJ . Huntington’s disease: from molecular pathogenesis to clinical treatment. Lancet Neurol 2011; 10: 83–98.
Vagner T, Young D, Mouravlev A . Nucleic acid-based therapy approaches for Huntington’s disease. Neurol Res Int 2012; 2012: 358370.
Kemp JM, Powell TP . The cortico-striate projection in the monkey. Brain 1970; 93: 525–546.
Selemon LD, Goldman-Rakic PS . Longitudinal topography and interdigitation of corticostriatal projections in the rhesus monkey. J Neurosci 1985; 5: 776–794.
Matsushita T, Elliger S, Elliiger C, Podsakoff G, Villarreal L, Kurtzman G et al. Adeno-associated virus vectors can be efficiently produced without helper virus. Gene Therapy 1998; 5: 938–945.
Wright J, Qu G, Tang C, Sommer J . Recombinant adeno-associated virus: formulation challenges and strategies for a gene therapy vector. Curr Opin Drug Discov Dev 2003; 6: 174–178.
Krauze MT, Saito R, Noble C, Tamas M, Bringas J, Park JW et al. Reflux-free cannula for convection-enhanced high-speed delivery of therapeutic agents. J Neurosurg 2005; 103: 923–929.
Richardson RM, Kells AP, Rosenbluth KH, Salegio EA, Fiandaca MS, Larson PS et al. Interventional MRI-guided putaminal delivery of AAV2-GDNF for a planned clinical trial in Parkinson’s disease. Mol Ther 2011; 19: 1048–1057.
Acknowledgements
This study was supported by a grant to KSB from NIH-NINDS (R01NS073940-01).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
San Sebastian, W., Samaranch, L., Heller, G. et al. Adeno-associated virus type 6 is retrogradely transported in the non-human primate brain. Gene Ther 20, 1178–1183 (2013). https://doi.org/10.1038/gt.2013.48
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2013.48
Keywords
This article is cited by
-
Brain-wide TVA compensation allows rabies virus to retrograde target cell-type-specific projection neurons
Molecular Brain (2022)
-
Dorsal striatal dopamine induces fronto-cortical hypoactivity and attenuates anxiety and compulsive behaviors in rats
Neuropsychopharmacology (2022)
-
Systemic AAV6-synapsin-GFP administration results in lower liver biodistribution, compared to AAV1&2 and AAV9, with neuronal expression following ultrasound-mediated brain delivery
Scientific Reports (2021)
-
AAV9-Retro mediates efficient transduction with axon terminal absorption and blood–brain barrier transportation
Molecular Brain (2020)
-
Gene Therapy for Neurodegenerative Diseases
Neurotherapeutics (2019)