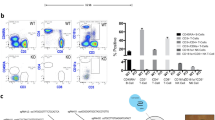
Abstract
Primary hyperoxaluria type 1 (PH1) is an inborn error of liver metabolism due to deficiency of the peroxisomal enzyme alanine:glyoxylate aminotransferase (AGT), which catalyzes conversion of glyoxylate into glycine. AGT deficiency results in overproduction of oxalate that ultimately leads to end-stage renal disease and death. Organ transplantation as either preemptive liver transplantation or combined liver/kidney transplantation is the only available therapy to prevent disease progression. Gene therapy is an attractive option to provide an alternative treatment for PH1. Toward this goal, we investigated helper-dependent adenoviral (HDAd) vectors for liver-directed gene therapy of PH1. Compared with saline controls, AGT-deficient mice injected with an HDAd encoding the AGT under the control of a liver-specific promoter showed a significant reduction of hyperoxaluria and less increase of urinary oxalate following challenge with ethylene glycol, a precursor of glyoxylate. These studies may thus pave the way to clinical application of HDAd for PH1 gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Piccolo P, Brunetti-Pierri N . Gene therapy for inherited diseases of liver metabolism. Hum Gene Ther 2015; 26: 186–192.
Leumann E, Hoppe B . The primary hyperoxalurias. J Am Soc Nephrol 2001; 12: 1986–1993.
Hoppe B, Langman CB . A United States survey on diagnosis, treatment, and outcome of primary hyperoxaluria. Pediatr Nephrol 2003; 18: 986–991.
Cochat P, Liutkus A, Fargue S, Basmaison O, Ranchin B, Rolland MO . Primary hyperoxaluria type 1: still challenging!. Pediatr Nephrol 2006; 21: 1075–1081.
Cochat P, Koch Nogueira PC, Mahmoud MA, Jamieson NV, Scheinman JI, Rolland MO . Primary hyperoxaluria in infants: medical, ethical, and economic issues. J Pediatr 1999; 135: 746–750.
Mandrile G, van Woerden CS, Berchialla P, Beck BB, Acquaviva Bourdain C, Hulton SA et al. Data from a large European study indicate that the outcome of primary hyperoxaluria type 1 correlates with the AGXT mutation type. Kidney Int 2014; 86: 1197–1204.
Cochat P, Faure JL, Divry P, Danpure CJ, Descos B, Wright C et al. Liver transplantation in primary hyperoxaluria type 1. Lancet 1989; 1: 1142–1143.
Watts RW, Calne RY, Rolles K, Danpure CJ, Morgan SH, Mansell MA et al. Successful treatment of primary hyperoxaluria type I by combined hepatic and renal transplantation. Lancet 1987; 2: 474–475.
Millan MT, Berquist WE, So SK, Sarwal MM, Wayman KI, Cox KL et al. One hundred percent patient and kidney allograft survival with simultaneous liver and kidney transplantation in infants with primary hyperoxaluria: a single-center experience. Transplantation 2003; 76: 1458–1463.
Danpure CJ . Primary hyperoxaluria: from gene defects to designer drugs? Nephrol Dial Transplant 2005; 20: 1525–1529.
Danpure CJ . Molecular etiology of primary hyperoxaluria type 1: new directions for treatment. Am J Nephrol 2005; 25: 303–310.
Sidhu H, Allison MJ, Chow JM, Clark A, Peck AB . Rapid reversal of hyperoxaluria in a rat model after probiotic administration of Oxalobacter formigenes. J Urol 2001; 166: 1487–1491.
Campieri C, Campieri M, Bertuzzi V, Swennen E, Matteuzzi D, Stefoni S et al. Reduction of oxaluria after an oral course of lactic acid bacteria at high concentration. Kidney Int 2001; 60: 1097–1105.
Lieske JC, Goldfarb DS, De Simone C, Regnier C . Use of a probiotic to decrease enteric hyperoxaluria. Kidney Int 2005; 68: 1244–1249.
Alonso MH, Ryckman FC . Current concepts in pediatric liver transplant. Semin Liver Dis 1998; 18: 295–307.
Newstead CG . Assessment of risk of cancer after renal transplantation. Lancet 1998; 351: 610–611.
Jiang J, Salido EC, Guha C, Wang X, Moitra R, Liu L et al. Correction of hyperoxaluria by liver repopulation with hepatocytes in a mouse model of primary hyperoxaluria type-1. Transplantation 2008; 85: 1253–1260.
Salido EC, Li XM, Lu Y, Wang X, Santana A, Roy-Chowdhury N et al. Alanine-glyoxylate aminotransferase-deficient mice, a model for primary hyperoxaluria that responds to adenoviral gene transfer. Proc Natl Acad Sci USA 2006; 103: 18249–18254.
Salido E, Rodriguez-Pena M, Santana A, Beattie SG, Petry H, Torres A . Phenotypic correction of a mouse model for primary hyperoxaluria with adeno-associated virus gene transfer. Mol Ther 2011; 19: 870–875.
Brunetti-Pierri N, Ng P . Progress towards the clinical application of helper-dependent adenoviral vectors for liver and lung gene therapy. Curr Opin Mol Ther 2006; 8: 446–454.
Brunetti-Pierri N, Ng P . Helper-dependent adenoviral vectors for liver-directed gene therapy. Hum Mol Genet 2011; 20: R7–R13.
Brunetti-Pierri N, Ng T, Iannitti D, Cioffi W, Stapleton G, Law M et al. Transgene expression up to 7 years in nonhuman primates following hepatic transduction with helper-dependent adenoviral vectors. Hum Gene Ther 2013; 24: 761–765.
Tao N, Gao GP, Parr M, Johnston J, Baradet T, Wilson JM et al. Sequestration of adenoviral vector by Kupffer cells leads to a nonlinear dose response of transduction in liver. Mol Ther 2001; 3: 28–35.
Schiedner G, Hertel S, Johnston M, Dries V, van Rooijen N, Kochanek S . Selective depletion or blockade of Kupffer cells leads to enhanced and prolonged hepatic transgene expression using high-capacity adenoviral vectors. Mol Ther 2003; 7: 35–43.
Sullivan DE, Dash S, Du H, Hiramatsu N, Aydin F, Kolls J et al. Liver-directed gene transfer in non-human primates. Hum Gene Ther 1997; 8: 1195–1206.
Nunes FA, Furth EE, Wilson JM, Raper SE . Gene transfer into the liver of nonhuman primates with E1-deleted recombinant adenoviral vectors: safety of readministration. Hum Gene Ther 1999; 10: 2515–2526.
Morral N, O'Neal WK, Rice K, Leland MM, Piedra PA, Aguilar-Cordova E et al. Lethal toxicity, severe endothelial injury, and a threshold effect with high doses of an adenoviral vector in baboons. Hum Gene Ther 2002; 13: 143–154.
Piccolo P, Vetrini F, Mithbaokar P, Grove NC, Bertin T, Palmer D et al. SR-A and SREC-I are kupffer and endothelial cell receptors for helper-dependent adenoviral vectors. Mol Ther 2013; 21: 767–774.
Raper SE, Chirmule N, Lee FS, Wivel NA, Bagg A, Gao GP et al. Fatal systemic inflammatory response syndrome in a ornithine transcarbamylase deficient patient following adenoviral gene transfer. Mol Genet Metab 2003; 80: 148–158.
Brunetti-Pierri N, Palmer DJ, Beaudet AL, Carey KD, Finegold M, Ng P . Acute toxicity after high-dose systemic injection of helper-dependent adenoviral vectors into nonhuman primates. Hum Gene Ther 2004; 15: 35–46.
Muruve DA, Cotter MJ, Zaiss AK, White LR, Liu Q, Chan T et al. Helper-dependent adenovirus vectors elicit intact innate but attenuated adaptive host immune responses in vivo. J Virol 2004; 78: 5966–5972.
Brunetti-Pierri N, Stapleton GE, Law M, Breinholt J, Palmer DJ, Zuo Y et al. Efficient, long-term hepatic gene transfer using clinically relevant HDAd doses by balloon occlusion catheter delivery in nonhuman primates. Mol Ther 2009; 17: 327–333.
Brunetti-Pierri N, Liou A, Patel P, Palmer D, Grove N, Finegold M et al. Balloon catheter delivery of helper-dependent adenoviral vector results in sustained, therapeutic hFIX expression in rhesus macaques. Mol Ther 2012; 20: 1863–1870.
Brunetti-Pierri N, Ng T, Iannitti DA, Palmer DJ, Beaudet AL, Finegold MJ et al. Improved hepatic transduction, reduced systemic vector dissemination, and long-term transgene expression by delivering helper-dependent adenoviral vectors into the surgically isolated liver of nonhuman primates. Hum Gene Ther 2006; 17: 391–404.
Kamoda N, Minatogawa Y, Nakamura M, Nakanishi J, Okuno E, Kido R . The organ distribution of human alanine-2-oxoglutarate aminotransferase and alanine-glyoxylate aminotransferase. Biochem Med 1980; 23: 25–34.
Danpure CJ . Primary Hyperoxaluria. McGraw Hill: New York, NY, USA, 2009.
Danpure CJ . Primary hyperoxaluria type 1: AGT mistargeting highlights the fundamental differences between the peroxisomal and mitochondrial protein import pathways. Biochim Biophys Acta 2006; 1763: 1776–1784.
Nathwani AC, Tuddenham EG, Rangarajan S, Rosales C, McIntosh J, Linch DC et al. Adenovirus-associated virus vector-mediated gene transfer in hemophilia B. N Engl J Med 2011; 365: 2357–2365.
Nathwani AC, Reiss UM, Tuddenham EG, Rosales C, Chowdary P, McIntosh J et al. Long-term safety and efficacy of factor IX gene therapy in hemophilia B. N Engl J Med 2014; 371: 1994–2004.
Manno CS, Pierce GF, Arruda VR, Glader B, Ragni M, Rasko JJ et al. Successful transduction of liver in hemophilia by AAV-Factor IX and limitations imposed by the host immune response. Nat Med 2006; 12: 342–347.
Donsante A, Miller DG, Li Y, Vogler C, Brunt EM, Russell DW et al. AAV vector integration sites in mouse hepatocellular carcinoma. Science 2007; 317: 477.
Chandler RJ, LaFave MC, Varshney GK, Trivedi NS, Carrillo-Carrasco N, Senac JS et al. Vector design influences hepatic genotoxicity after adeno-associated virus gene therapy. J Clin Invest 2015; 125: 870–880.
Nault JC, Datta S, Imbeaud S, Franconi A, Mallet M, Couchy G et al. Recurrent AAV2-related insertional mutagenesis in human hepatocellular carcinomas. Nat Genet 2015; 47: 1187–1193.
Jager L, Ehrhardt A . Persistence of high-capacity adenoviral vectors as replication-defective monomeric genomes in vitro and in murine liver. Hum Gene Ther 2009; 20: 883–896.
Ross PJ, Kennedy MA, Parks RJ . Host cell detection of noncoding stuffer DNA contained in helper-dependent adenovirus vectors leads to epigenetic repression of transgene expression. J Virol 2009; 83: 8409–8417.
Ross PJ, Kennedy MA, Christou C, Risco Quiroz M, Poulin KL, Parks RJ . Assembly of helper-dependent adenovirus DNA into chromatin promotes efficient gene expression. J Virol 2011; 85: 3950–3958.
Harui A, Suzuki S, Kochanek S, Mitani K . Frequency and stability of chromosomal integration of adenovirus vectors. J Virol 1999; 73: 6141–6146.
Stephen SL, Sivanandam VG, Kochanek S . Homologous and heterologous recombination between adenovirus vector DNA and chromosomal DNA. J Gene Med 2008; 10: 1176–1189.
Hillgenberg M, Tonnies H, Strauss M . Chromosomal integration pattern of a helper-dependent minimal adenovirus vector with a selectable marker inserted into a 27.4-kilobase genomic stuffer. J Virol 2001; 75: 9896–9908.
Ohbayashi F, Balamotis MA, Kishimoto A, Aizawa E, Diaz A, Hasty P et al. Correction of chromosomal mutation and random integration in embryonic stem cells with helper-dependent adenoviral vectors. Proc Natl Acad Sci USA 2005; 102: 13628–13633.
Suzuki K, Mitsui K, Aizawa E, Hasegawa K, Kawase E, Yamagishi T et al. Highly efficient transient gene expression and gene targeting in primate embryonic stem cells with helper-dependent adenoviral vectors. Proc Natl Acad Sci USA 2008; 105: 13781–13786.
Stephen SL, Montini E, Sivanandam VG, Al-Dhalimy M, Kestler HA, Finegold M et al. Chromosomal integration of adenoviral vector DNA in vivo. J Virol 2010; 84: 9987–9994.
Schnell MA, Zhang Y, Tazelaar J, Gao GP, Yu QC, Qian R et al. Activation of innate immunity in nonhuman primates following intraportal administration of adenoviral vectors. Mol Ther 2001; 3: 708–722.
Palmer D, Ng P . Improved system for helper-dependent adenoviral vector production. Mol Ther 2003; 8: 846–852.
Suzuki M, Cela R, Clarke C, Bertin TK, Mourino S, Lee B . Large-scale production of high-quality helper-dependent adenoviral vectors using adherent cells in cell factories. Hum Gene Ther 2010; 21: 120–126.
Jaffe M . Ueber den Niederschlag, welchen Pikrinsäure im normalen Harn erzeugt, und über eine neue Reaction des Kreatinins. Z Physiol Chem 1886; 10: 391–400.
Celis JE . Cell Biology. A Laboratory Handbook. Elsevier Academic Press: Burlington, MA, USA, 2006.
Wanders RJ, Ruiter J, van Roermund CW, Schutgens RB, Ofman R, Jurriaans S et al. Human liver L-alanine-glyoxylate aminotransferase: characteristics and activity in controls and hyperoxaluria type I patients using a simple spectrophotometric method. Clin Chim Acta 1990; 189: 139–144.
Yasue T . Histochemical identification of calcium oxalate. Acta Histochem Cytochem 1969; 2: 83–95.
Kalaitzis AA, Lawrence ND . A simple approach to ranking differentially expressed gene expression time courses through Gaussian process regression. BMC Bioinformatics 2011; 12: 180.
Acknowledgements
We thank Dr Roy-Chowdhury for providing us Agxt−/− mice and Dr Elisa Oppici for the anti-AGT antibody. We thank TIGEM Bioinformatic core for statistical analyses and the staff of TIGEM/IGB animal facility for assistance with mouse procedures. RC was supported by PONa3_00311. This work was supported by research grants of The Hyperoxaluria and Oxalosis Foundation to NB-P and Fondazione Telethon, Italy (TCBP37TELC and TCBMT3TELD to NB-P).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Castello, R., Borzone, R., D'Aria, S. et al. Helper-dependent adenoviral vectors for liver-directed gene therapy of primary hyperoxaluria type 1. Gene Ther 23, 129–134 (2016). https://doi.org/10.1038/gt.2015.107
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2015.107