Abstract
The onset of cardiovascular diseases often shows time-of-day variation. Acute myocardial infarction or ventricular arrhythmia such as ventricular tachycardia occurs mainly in the early morning. Multiple biochemical and physiological parameters show circadian rhythm, which may account for the diurnal variation of cardiovascular events. These include the variations in blood pressure, activity of the autonomic nervous system and renin–angiotensin axis, coagulation cascade, vascular tone and the intracellular metabolism of cardiomyocytes. Importantly, the molecular clock system seems to underlie the circadian variation of these parameters. The center of the biological clock, also known as the central clock, exists in the suprachiasmatic nucleus. In contrast, the molecular clock system is also activated in each cell of the peripheral organs and constitute the peripheral clock. The biological clock system is currently considered to have a beneficial role in maintaining the homeostasis of each organ. Discoordination, however, between the peripheral clock and external environment could potentially underlie the development of cardiovascular events. Therefore, understanding the molecular and cellular pathways by which cardiovascular events occur in a diurnal oscillatory pattern will help the establishment of a novel therapeutic approach to the management of cardiovascular disorders.
Similar content being viewed by others
Introduction
Multiple systemic circulatory parameters, such as blood pressure (BP), heart rate (HR) and activity of the autonomic nervous system or renin–angiotensin cascade, exhibit time-of-day variation.1, 2, 3 BP mostly shows a circadian variation with a peak in the early morning, and dysregulation of systemic BP rhythm is independently associated with the poor prognosis of patients with acute myocardial infarction (AMI).4 The link between BP variation and the onset of cardiovascular diseases has been extensively studied.5 Moreover, the occurrence of cardiovascular events tends to show time-of-day variation.6, 7, 8 Therefore, elucidation of the link between the internal clock and cardiovascular function will help in the establishment of a novel therapeutic approach to cardiovascular diseases.
Circadian onset of cardiovascular diseases
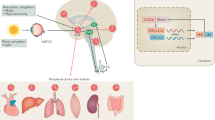
Circadian variation in the onset of cardiovascular diseases has been observed in stroke,9 unstable angina10 and sudden cardiac death.11 AMI has also been reported to mostly occur in the early morning.7 The rise in BP, HR, coagulation activity and vascular tone in the early morning underlies the frequent onset of AMI at this time of day (Figure 1). Ventricular arrhythmias such as ventricular tachycardia or ventricular fibrillation also tend to develop in the morning.6, 12, 13, 14 Meanwhile, the onset of takotsubo cardiomyopathy, an acute but reversible left ventricular dysfunction disorder that is usually triggered by stressful events, also exhibits circadian variation with a peak in the afternoon and nadir from 0000 to 0400 hours.15 The Mayo Clinic Percutaneous Coronary Intervention Registry investigated the medical records of patients (n=124) who were diagnosed with stent thrombosis and showed that the onset of stent thrombosis also exhibits circadian oscillation with a peak incidence around 0700 hours.16
In addition to thrombotic events, the occurrence of acute aortic dissection (AAD) also exhibits a time-of-day variation. The onset time of AAD has two peaks, consisting of a primary morning peak (0800–1100 hours) and a secondary evening peak (1700–1900 hours).17 Intriguingly, the diurnal BP pattern may affect the onset time of AAD, as illustrated by the observation that non-dipper or riser patients had a higher incidence of AAD during nighttime compared with dipper patients.18
It is important to note that the circadian variation of acute coronary syndrome (ACS) is not observed in patients receiving β-blocker treatment.7 The presence of chronic kidney disease (CKD) may also affect the onset time of ACS, as reflected by the finding that the onset of ACS showed circadian variation in non-CKD patients but not in CKD patients with an estimated glomerular filtration rate <60 ml min−1 1.73 m−2.19
Transition to or from daylight saving time occurs in spring and autumn, leading to the occurrence of a mismatch between the external and internal clocks during these transition periods. Compared with regular periods, there seems to be around a 30% increase in the incidence of ACS during the first 4 workdays of the transition period.20 These results imply that the trigger of cardiovascular events is strongly affected by the coordination between the internal clock and external environment.
Molecular clock in mammalian cells
In addition to the external environment, the internal clock also has a major role in physiological and biochemical functions. The center of the biological clock exists in the suprachiasmatic nucleus (SCN) of the hypothalamus and is referred to as the central clock. The intrinsic circadian rhythm of the central clock is elicited by the negative feedback loop formed by several core clock genes. Furthermore, the central clock receives external signals, such as light or physiological parameters, and adjusts the phase of its circadian clock accordingly, a phenomenon known as entrainment.21 The molecular processes of the biological clock have been summarized previously.5, 22 The circadian expression of clock genes can also be observed in each cell or tissue, which composes the peripheral clock. The peripheral clock, together with the rhythms of the systemic autonomic nervous system and endocrine system, are coordinated by the central clock. Although the peripheral clock seems to underlie the diurnal variation of these fluctuations, its precise molecular mechanisms are still unclear.
A transcriptional–translational negative feedback loop composed of the core clock genes constitutes the internal clock. Core clock genes include Clock, Npas2, Bmal1/2 and Period (Per)1/2/3, which belong to a group of helix-loop-helix/Per-Arnt-Sim domain-containing transcription factors. CLOCK/NPAS2 forms a heterodimer with BMAL1/2, and this CLOCK/BMAL heterodimer binds to the E-box upstream of Per1/2/3 and Cryptochrome1/2 (Cry). PER1/2/3 or CRY1/2 proteins are phosphorylated by casein kinase (CK)-1ɛ and degraded through an ubiquitin-mediated proteasomal pathway. Under normal circadian conditions, however, PER1/2/3 and CRY1/2 gradually accumulate in amount and inhibit CLOCK/BMAL-mediated transactivation processes, thereby establishing a negative feedback loop (Figure 2). This feedback loop takes approximately 24 h, thus accounting for the diurnal rhythm. The fundamental role of Bmal1 in the clock system is demonstrated by the fact that Bmal1-deficient mice (Bmal1-KO) exhibit arrhythmic behavior.23
The heterodimer of CLOCK and BMAL bind to the E-box upstream of per and cry genes and activates their transcription. PER and CRY proteins are phosphorylated by casein kinase 1 epsilon (CK-1ɛ) and degraded through the proteasomal pathway. PER and CRY proteins, however, gradually accumulate in amount and inhibit CLOCK/BMAL-mediated transcription of per and cry genes. A full color version of this figure is available at the Hypertension Research journal online.
The CLOCK/BMAL heterodimer also binds to the E-box and induces the circadian expression levels of the following genes, arginine vasopressin and Wee1. Genes induced by core clock genes are termed clock-controlled genes (CCGs). CLOCK/BMAL also binds to the promoter of proline-acid-rich (PAR)-domain basic leucine zipper transcription factors, D-element binding proteins (Dbp), hepatic leukemia factor (Hlf) and thyrotrophic embryonic factor (Tef). DBP, HLF, TEF and E4 promoter-binding Protein 4 (E4BP4) bind to D-box, which is also related to the molecular clock and involved in the diurnal expression of CCGs. In addition, the REV-ERB/ROR-binding element (RRE) acts to maintain the molecular clock system as well. The CLOCK/BMAL heterodimer induces the expression of Rev-erbα/β and Rorα genes, resulting in the circadian mobilization of REV-ERBα/β and RORα to the RRE upstream of other CCGs.
In addition to the central clock in the SCN, clock genes are also expressed in each organ with diurnal variation and make up the peripheral clock.24 With the exception of some specific differences, most of the molecular clock system of the peripheral clock seems to be similar to that of the central clock. We previously identified Clif/Bmal2 as one of the clock genes in vascular endothelial cells.25 CLIF/BMAL2 may function as a tissue-specific clock gene as circadian expression of Clif/Bmal2 is observed in the liver but not in the colon.26 Although the precise role of each peripheral clock still remains to be elucidated, the intrinsic clock in each organ is now considered to have a beneficial role in maintaining tissue homeostasis (Figure 3).27
The central clock exists in the suprachasmatic nucleus (SCN) in the hypothalamus. In addition, clock genes expressed in a circadian manner in each organ, thus called as peripheral clock. Coordination between the peripheral clock and autonomic nervous system or endocrine system has an integral role in maintaining the tissue homeostasis.
Circadian regulation of BP
It has been reported that BP in human individuals exhibits time-of-day variation with a rise in the morning and a decline at night time.28, 29 Fluctuations in the activities of the autonomic nervous system, renin–angiotensin–aldosterone axis and plasma cortisol level underlie the diurnal variation of BP. In addition, nighttime BP is inversely associated with urinary melatonin secretion in elderly population.30
Accumulating evidence has demonstrated that BP is also affected by the internal clock. Variation in Cry1 gene seems to be associated with arterial hypertension.31 In addition, circadian variation in BP is abolished in mice deficient in the Bmal1 gene or mutation in the Clock gene.32 Circadian variation of BP does not seem to depend on the internal clock in vascular endothelial cells, as its oscillation was not disrupted in endothelial cell-specific Bmal1-KO mice.33 Molecular clocks, however, in vascular smooth muscle cells, adrenal gland or kidney contribute to the variation in BP. The nuclear transcription factor, peroxisome proliferator-activated receptor γ (PPAR γ), directly induces Bmal1 mRNA expression. Notably, BP fluctuation was disrupted in mice deficient in PPARγ in vascular smooth muscle cells.34 Meanwhile, core clock genes Cry1 and Cry2 repressed the expression of the adrenal enzyme, type IV 3β-hydroxy-steroid dehydrogenase (Hsd3b6).35 Aldosterone synthesis is thus enhanced in Cry-1/2-deficient mice, resulting in the development of salt-sensitive hypertension. Moreover, renal collecting duct cells also have a critical role in BP fluctuation through the regulation of sodium channel expression.36 PER1 acts downstream of aldosterone and directly activates the expression of the α-subunit of the renal epithelial sodium channel (αENaC) in a circadian manner.37 In addition, endothelin-1 expression is increased in Per1-deficient mice, which suppresses the activity of sodium channel.36 Consistent with this hypothesis, systemic BP is decreased in Per1-deficient mice. The expression of αENaC is also decreased in Clock-knockout mice, thereby resulting in hypotension.38
Diurnal variation in the coagulation system
Of the several systemic parameters that affect the circadian onset of cardiovascular disorders, the coagulation cascade is also another factor that contributes to this diurnal variation. Systemic coagulation activity is determined by the balance between coagulation and fibrinolytic activities. In particular, serpin peptidase inhibitor/plasminogen activator inhibitor-1 (PAI-1) regulates the activity of tissue-type plasminogen activator (t-PA), thereby controlling systemic fibrinolytic activity. Importantly, PAI-1 mRNA and protein levels show a clear circadian variation.39, 40 Consistent with this result, plasma levels of the plasmin–plasmin inhibitor complex decreases in the morning.41 These results imply that the mismatch between coagulation and fibrinolytic activity may elicit the onset of hemostatic disorders. The coagulation cascade is further dysregulated in patients with obstructive sleep apnea syndrome (OSAS). In OSAS patients who suffer intermittent hypoxia, plasma PAI-1 activity was increased, whereas the activity of t-PA was decreased. Augmentation of the mismatch between coagulation and anticoagulation cascades may therefore account for the higher incidence of cardiovascular events in OSAS patients.42
Several studies have examined the molecular processes by which Pai-1 expression is induced in a circadian manner. Forced disruption of the behavioral cycle did not alter the diurnal oscillation of plasma PAI-1 levels in 12 healthy volunteers, suggesting that the morning peak in human plasma PAI-1 level is elicited by the internal clock.43 As an underlying mechanism, we and other groups identified that the heterodimer of CLOCK/BMAL1/2 binds to the E-box upstream of the Pai-1 gene, resulting in the circadian activation of its transcription.25, 44
Roles of the internal clock in vascular function
Consistent with the observation that BP or HR tends to increase in the early morning,45 vascular function also exhibits diurnal oscillation. In particular, endothelium-dependent vasodilatory activity shows time-of-day variation not only in in vitro cultured rat vascular endothelial cells but also in human subjects.46, 47 Importantly, vasodilatory function decreases in the early morning,48 resulting in a mismatch between oxygen consumption and supply during this time period.
Rho and its effector, Rho-associated protein kinase (ROCK), phosphorylates myosin light chain in vascular smooth muscle cells and thus acts as a major regulator of vascular contraction. Intriguingly, activation of ROCK2 shows diurnal variation, leading to the circadian phosphorylation of myosin light chain in cultured vascular smooth muscle cells.49 As an underlying molecular mechanism, RORα, a clock gene, enhances the expression of ROCK2 gene in a circadian manner. Notably, dysregulation of the Rho-kinase pathway has a major role in the pathogenesis of vasospastic angina (VSA), supported by the finding that inhibition of Rho-kinase seems to be beneficial in alleviating the VSA attack in both animal model and human VSA patients.50 Intriguingly, VSA patients exhibit enhanced Rho-kinase activity not only in vascular smooth muscle cells but also in circulating leukocytes.51 Moreover, Rho-kinase activity in circulating leukocytes also exhibits circadian variation.52 Although these results demonstrate the existence of a pathological process that activates Rho-kinase in a time-of-day-dependent manner, the molecular mechanisms by which Rho-kinase activity is enhanced in VSA patients still remain to be elucidated.
Similar to how it is in the central clock in the SCN, core clock genes are also expressed in vascular endothelial cells or smooth muscle cells.53, 54, 55 We previously identified thrombomodulin as one of the downstream CCGs in vascular endothelial cells, a factor that exerts anticoagulant activity on the membranes of vascular endothelial cells.55 In addition to the coagulation system, the expression levels of tissue inhibitor of metalloproteinase 1 and 3, collagen 3a1, transgelin1 (sm22α) and calponin1 also exhibit diurnal variation in vascular smooth muscle cells.56
Genetically engineered mice with mutations in core clock genes help to understand the roles of each peripheral clock in the pathogenesis of vascular disorders. Mouse carotid artery ligation was performed to induce vascular remodeling and thickening of the vessel walls. Intriguingly, carotid artery ligation of Bmal1-KO resulted in altered vascular remodeling with dilated vascular lumen and thrombus formation. Although Clockmut mice behave normally in a light/dark (L/D) cycle environment, it becomes arrhythmic in constantly dark conditions. The extent of vascular remodeling in Clockmut mice that have undergone carotid artery ligation was similar to control mice when the mice were kept in a L/D cycle. However, vascular remodeling was significantly augmented in Clockmut mice when they were kept in constant darkness, indicating the roles of intrinsic circadian rhythm in the development of vascular pathology. Importantly, the expression of Pai-1 gene in vascular endothelium or liver is increased in Bmal1-KO mice. Moreover, aortic ring prepared from Bmal1-KO mice showed reduced response to acetylcholine, highlighting the impaired endothelial function in Bmal1-KO mice. Supporting this hypothesis is the observation that endothelial Akt signaling is also blunted in Bmal1-KO mice.57
Although endothelial nitric oxide (NO) has an integral role in vascular integrity, its activity shows diurnal variation. The abundance of endothelial NO synthase (eNOS) mRNA or protein has time-of-day variation.58 Rat mesenteric artery exhibits time-of-day variation in its contractility. Intriguingly, treatment with nitro-L-arginine methyl ester, a NOS inhibitor, abolished its fluctuation. Core clock genes also induce the circadian expression of dihydrofolate reductase, which regulates the bioavailability of an eNOS cofactor, biopterin.59 Aorta from Bmal1-KO mice showed a reduction in tetrahydrobiopterin, and an increase in dihydrobiopterin, resulting in an impaired coupling of eNOS. Importantly, vascular signaling such as NO or angiotensin II could in turn affect the internal clock.54, 60 It has been reported that the amplitude of circadian clock decreases with age. Treatment with NO donor significantly upregulates the expression of Per2 gene, resulting in a restoration of the biological clock.60
Photochemical injury of the femoral artery was performed as a murine experimental thrombosis disease model. Time to thrombotic vascular occlusion (TTVO) after photochemical injury was measured at zeitgeber time (ZT) 2, 8, 14 and 20. Time-of-day variation in TTVO was observed in wild-type mice with the longest TTVO at ZT 8. Importantly, circadian oscillation of TTVO is disrupted when photochemical injury was performed in Clockmut mice, suggesting that the intrinsic molecular clock system contributes to the onset of vascular occlusion. Multiple factors contribute to thrombosis formation, including endothelial function, BP and coagulation activity. To elucidate the role of the peripheral clock in vascular endothelial cells, the photochemical injury model was carried out in endothelial cell-specific Bmal1-KO mice. Intriguingly, diurnal oscillation of TTVO disappeared in endothelial Bmal1-KO mice, whereas diurnal oscillation of BP or coagulation activity was not affected. These results thus showed that internal clock processes in vascular endothelial cells have an integral role in the development of vascular thrombosis.33
Biological clock system also regulates the tissue remodeling or angiogenesis. In response to a tissue injury, CD34+/CD133+ progenitor cells (PCs) or endothelial PCs (EPCs) were mobilized and promotes tissue repair or angiogenic processes. The number of human peripheral blood PCs shows diurnal oscillation with a peak at 2000 hours and nadir at 0800 hours.61 Intriguingly, Per2 is highly expressed in EPCs.62 EPC from Per2-deficient mice had reduced potential in proliferation, migration or tube formation. Supporting this hypothesis, mice transplanted with Per2-deficient EPC showed decreased left ventricular function after myocardial infarction compared with wild-type EPC-transplanted mice. Clock genes also have an integral role in angiogenic processes of zebrafish.63 Morpholino to Bmal1 reduced the expression of vascular endothelial growth factor, resulting in the impairment of developmental angiogenesis. Inhibition of Per2 in turn accelerates the angiogenesis, implying the link between clock genes and developmental angiogenesis.
The internal clock in smooth muscle cells also contributes to the time-of-day variations of BP. Deletion of Bmal1 in smooth muscle cells (SM-Bmal1-KO) showed decreased oscillation of clock genes in mesenteric arteries. Although the contractile responses of mesenteric arteries to phenylephrine or serotonin show time-of-day variation, it was significantly attenuated in SM-Bmal1-KO mice. As an underlying molecular mechanism, BMAL1 transactivates Rock2 gene expression and leads to the oscillation of myosin light chain phosphorylation. Consistent with this hypothesis, diurnal oscillation of BP is abolished in SM-Bmal1-KO mice, illuminating the roles of the internal clock in BP regulation.64
Peripheral clock in the vasculature also contributes to the homeostasis of transplanted vessels. Development of atherosclerosis and inflammatory cell accumulation were evaluated after isograft transplantation of the carotid artery. Compared with the isograft obtained from wild-type mice, those from Bmal1-KO or Per triple knockout mice showed severe atherosclerotic lesions.65
Molecular clock in the heart
In addition to the peripheral clock in the vasculature, cardiomyocytes also have an intrinsic molecular clock where the core clock genes exhibit diurnal variation.25 Similar to the serum shock of cultured fibroblasts, the phase of each peripheral clock in cardiomyocytes can be synchronized using norepinephrine.66 A comprehensive analysis of the heart or liver for genes that exhibit circadian expression revealed 8–10% of all genes in each organ to be CCGs.67 Intriguingly, most of the CCGs are organ specific, suggesting that each organ has its own specific peripheral clock. Several CCGs in the heart have been identified, including pyruvate dehydrogenase kinase isozyme-4 (Pdk-4), solute carrier family 2 (facilitated glucose transporter), member 1 (Slc2a1), also known as glucose transporter 1 (Glut-1), Slc2a4/Glut-4 and potassium channels Kv1.5 and Kv4.2.68, 69, 70
The peripheral clock in each organ may have a beneficial role in maintaining organ homeostasis. Natriuretic peptide A (NPPA/ANP) is known to act as a cardioprotective agent. Importantly, the expression of anp shows circadian oscillation. As BP rises in a time-of-day-dependent manner, simultaneous induction of ANP in cardiomyocytes may offset an increase in afterload on cardiomyocytes.
In addition to the circadian onset of AMI, myocardial infarction tolerance also exhibits diurnal oscillation. In a mice model of ischemia/reperfusion, hearts subjected to ischemia at ZT 12 showed significantly greater infarct size compared with hearts subjected to ischemia at ZT 0. Importantly, this time-of-day dependence of myocardial damage is abolished in cardiomyocyte-specific circadian Clock-mutant mice (CCM), thereby highlighting the roles of endogenous clock rhythm in ischemia tolerance.71 Ischemia tolerance also exhibits circadian oscillation in human subjects. A retrospective study investigated the medical records of patients with ST elevation myocardial infarction (STEMI). Using peak serum creatine kinase (CK) levels as a surrogate marker of infarct size, the study demonstrated a significant association between time of STEMI onset and peak CK level. Peak CK level was maximum when the STEMI developed at 0100 hours. Consistent with peak CK levels, the extent of decrease in left ventricular ejection fraction also depended on the time of STEMI onset.72
Importantly, the phase or amplitude of the peripheral clock in the heart seems to be affected when cardiac disease is present. In an animal model of pressure overload-induced cardiac hypertrophy, circadian expression levels of PAR transcription factors (Dbp, Hlf) and Anp were significantly dampened.69 As E4BP4 acts to antagonize the function of PAR transcription factors, attenuation of the peripheral clock of the heart is seen in myocardial ischemia/reperfusion owing to significant increase in E4BP4 expression under such conditions.73 Disruption of the peripheral clock in the heart is also observed in diabetic hearts.74
Desynchronization between the internal clock and external environment may elicit the onset of cardiovascular disorders. Continuous changes in the daily L/D cycle shortens the survival of cardiomyopathic hamsters.75 Transverse aortic constriction procedure is used to acutely increase the afterload of the left ventricle (LV), leading to LV hypertrophy. Rhythm disruption from a 24-h (12-h/12-h L/D) to a 20-h (10-h/10-h) cycle significantly impaired the LV systolic function of mice that were subjected to the transverse aortic constriction model.76 Moreover, even a short-term rhythm disruption augments the severity of LV remodeling after myocardial infarction.77
In contrast, adjusting the circadian rhythm with restrictive feeding may improve cardiac performance. Drosophila, a fruit fly, also has an internal circadian rhythm, and its cardiac performance can be evaluated by high-speed video imaging as fractional shortening. Time-restricted feeding (TRF) is a condition in which food access of a fly is limited to daytime 12 h every day. TRF did not affect the food consumption or locomotor activity of flies. Although fractional shortening in drosophila usually decreases with age, TRF significantly improved cardiac performance in aged flies, indicating the beneficial effect of TRF on cardiac function.78 Moreover, synchronization of the rhythm between the internal clock and external environment seems to be beneficial for shift-workers. The properties of human circadian rhythm, namely chronotypes, are highly individualistic and range from early, intermediate to late chronotypes. Intriguingly, both daily rhythm and sleep state were significantly improved when a work-shift schedule is designed based on the chronotype of each working individual.79 These findings also support the hypothesis that the peripheral clock in each organ has a beneficial role by promoting the coordination between the internal clock and the external environment.
To elucidate the roles of the peripheral clock in the heart, genetically engineered mice were used for the cardiovascular disease models. Although Bmal1-KO mice exhibit arrhythmic behavior, it also develops several organ dysfunctions such as infertility,80 structural alteration in skeletal muscle81 and dilated cardiomyopathy.82 To further elucidate the roles of clock genes in cardiomyocytes, the CCM transgenic mouse line was established in which mutant CLOCK protein is overexpressed specifically in cardiomyocytes. When the CCM mice were maintained in a repeated phase shift (12-h phase shift biweekly) environment, a significant rise in cardiac hypertrophy marker gene expression was observed.83 Studies using CCM mice also revealed that intracellular metabolism of cardiomyocytes, including triglycerides and glycogen stores, show circadian variation in a peripheral clock-dependent manner.84, 85 The roles of the peripheral clock in cardiomyocytes were also evaluated using cardiomyocyte-specific Bmal1-KO mice.82, 86 Although glucose metabolism was repressed, fatty acid oxidation was activated in cardiomyocyte-specific Bmal1-KO mice. Importantly, decreased ejection fraction and subsequent development of heart failure was observed in aged cardiomyocyte-specific Bmal1-KO mice. Phosphoinositide-3-kinase, regulatory subunit 1 (Pik3r1) is a subunit of PI3K and regulates its intracellular signaling. Intriguingly, the Pik3r1 gene is identified as one of the CCGs in cardiomyocytes. Taken together, these results indicate that the peripheral clock in cardiomyocytes is involved in growth factor signaling and fatty acid oxidation.
Circadian clock and the onset of cardiac arrhythmia
Diurnal variation of the autonomic nervous system and peripheral clock induces the time-of-day variation in the basic electrophysiological parameters of cardiomyocytes. Atrioventricular nodal function, QT interval and ventricular refractory period display circadian variation.87, 88, 89 Of the numerous CCGs identified, those involved in cellular electrophysiological function include two voltage gated K channels, Kv1.5 and Kv4.2,68 and sodium channel, voltage-gated, type V, alpha subunit (Scn5a).90 Intriguingly, BMAL1 directly binds and induces the circadian expression of Scn5a gene in cardiomyocytes, resulting in HR variability. Supporting this hypothesis, mice with a cardiomyocyte-specific deletion of the Bmal1 gene have slower HR and prolonged RR intervals, resulting in a higher susceptibility to the development of arrhythmia during electromechanical stimulations.
The onset of ventricular arrhythmias also shows time-of-day variation, with a first peak in the morning (0700–1100 hours) and followed by a second peak in the afternoon.6, 13 The short ventricular refractory period underlies the onset of ventricular tachycardia or ventricular fibrillation in the morning hours.14 Kv channel-interacting protein 2 (KChIP2) contributes to the transient outward potassium flow in cardiomyocytes, and its transcript level shows circadian variation. Intriguingly, BMAL1-mediated induction of a transcription factor, kruppel-like factor 15 (Klf15), elicits the diurnal variation in KChip2 expression,91 resulting in the time-of-day variation in QT interval and subsequent onset of ventricular arrhythmia.
It should be noted that the autonomic nervous system also seems to contribute to the time-of-day variation in ventricular arrhythmias. The Sudden Cardiac Death in Heart Failure Trial examined whether circadian patterns in ventricular arrhythmia is observed in patients with implantable cardioverter-defibrillator and found that the circadian onset of ventricular tachycardia/ventricular fibrillation disappeared in patients receiving beta-blocker therapy.92
Novel functions of core clock genes
It is important to be reminded that core clock genes may also have clock-independent functions. CRY, one of the core clock genes, seems to be involved in heart morphogenesis. Silencing Drosophila cryptochrome (dcry) slowed the HR and reduced heart size in flies, implicating the developmental functions of core clock genes.93
The core clock gene Bmal1 also has an anti-inflammatory effect. BMAL1 affects the epigenetic modification and suppresses the expression of chemokine (C-C motif) ligand 2 (Ccl2), a chemokine that mostly recruits pro-inflammatory monocytes (Ly6Chi) through the interaction with its receptor, CCR2.94 Supporting this hypothesis, the number of circulating Ly6Chi monocytes exhibits circadian oscillation, and the disease severity of Listeria monocytogenes infection was augmented when the pathogen was inoculated at ZT 8. In addition, REV-ERBα also suppresses the expression of Ccl2.95 Recruitment of pro-inflammatory monocytes/macrophages was increased in Rev-erbα-deficient mice, resulting in an augmentation of atheroma formation in a vascular injury model.96
Clock genes also directly affect intracellular metabolism. The nuclear transcription factor PPARγ has an essential role in adipocyte differentiation through its binding to the PPAR response elements (PPREs). PER2 directly inhibits the function of PPARγ by suppressing the binding of PPARγ to PPRE.97 Importantly, fatty acid oxidation was activated but triacylglycerol accumulation was decreased in Per2-deficient cells. Moreover, Per2-deficient fibroblasts exhibit higher differentiation potential to adipocytes. Intriguingly, a clinical study in diabetic patients showed that gene polymorphisms of Clif/Bmal2 are significantly associated with the presence of type 2 diabetes, suggesting a role for Clif/Bmal2 in intracellular metabolism.98
Signaling involving adenosine and the adenosine receptor, ADORA2B, has an essential role in ischemia preconditioning of the heart. Importantly, the adenosine signal stabilizes the glycolytic transcription factor, hypoxia inducible factor (HIF)-1α, through a PER2-dependent mechanism.99 HIF-1α subsequently activates the expression levels of glycolytic genes, resulting in the adaptation of cardiomyocytes to ischemia. HIF-1α also induces the expression of Per1 gene.100 Circadian oscillation of Per1 promoter activity is dampened in chronic hypoxia, illuminating an interplay between circadian rhythm and hypoxia signaling.
Conclusion
The internal clock system exists not only in the SCN as the central clock but also in each cell of the cardiovascular organs. The peripheral clock in each organ or cell seems to have a beneficial role by anticipating the changes in external environments. Therefore, disruption between the clock system in each organ and the external environment tends to augment the severity of the organ dysfunction. Elucidation of the precise roles of the molecular clock in each cardiovascular organ will help us establish a novel therapeutic approach for cardiovascular disorders.
References
Charloux A, Gronfier C, Lonsdorfer-Wolf E, Piquard F, Brandenberger G . Aldosterone release during the sleep-wake cycle in humans. Am J Physiol 1999; 276: E43–E49.
Hartikainen J, Tarkiainen I, Tahvanainen K, Mantysaari M, Lansimies E, Pyorala K . Circadian variation of cardiac autonomic regulation during 24-h bed rest. Clin Physiol 1993; 13: 185–196.
Stern N, Sowers JR, McGinty D, Beahm E, Littner M, Catania R, Eggena P . Circadian rhythm of plasma renin activity in older normal and essential hypertensive men: relation with inactive renin, aldosterone, cortisol and REM sleep. J Hypertens 1986; 4: 543–550.
Minamisawa M, Izawa A, Motoki H, Kashima Y, Hioki H, Abe N, Miura T, Ebisawa S, Miyashita Y, Koyama J, Ikeda U . Prognostic significance of neuroadrenergic dysfunction for cardiovascular events in patients with acute myocardial infarction. Circ J 2015; 79: 2238–2245.
Takeda N, Maemura K . Cardiovascular disease, chronopharmacotherapy, and the molecular clock. Adv Drug Deliv Rev 2010; 62: 956–966.
Behrens S, Galecka M, Bruggemann T, Ehlers C, Willich SN, Ziss W, Dissmann R, Andresen D . Circadian variation of sustained ventricular tachyarrhythmias terminated by appropriate shocks in patients with an implantable cardioverter defibrillator. Am Heart J 1995; 130: 79–84.
Muller JE, Stone PH, Turi ZG, Rutherford JD, Czeisler CA, Parker C, Poole WK, Passamani E, Roberts R, Robertson T, Sobel BE, Willerson JT, Braunwald E,, the MILIS Study Group. Circadian variation in the frequency of onset of acute myocardial infarction. N Engl J Med 1985; 313: 1315–1322.
Takeda N, Maemura K . Circadian clock and vascular disease. Hypertens Res 2010; 33: 645–651.
Elliott WJ . Circadian variation in the timing of stroke onset: a meta-analysis. Stroke 1998; 29: 992–996.
Cannon CP, McCabe CH, Stone PH, Schactman M, Thompson B, Theroux P, Gibson RS, Feldman T, Kleiman NS, Tofler GH, Muller JE, Chaitman BR, Braunwald E . Circadian variation in the onset of unstable angina and non-Q-wave acute myocardial infarction (the TIMI III Registry and TIMI IIIB). Am J Cardiol 1997; 79: 253–258.
Arntz HR, Willich SN, Oeff M, Bruggemann T, Stern R, Heinzmann A, Matenaer B, Schroder R . Circadian variation of sudden cardiac death reflects age-related variability in ventricular fibrillation. Circulation 1993; 88: 2284–2289.
Englund A, Behrens S, Wegscheider K, Rowland E . Circadian variation of malignant ventricular arrhythmias in patients with ischemic and nonischemic heart disease after cardioverter defibrillator implantation. European 7219 Jewel Investigators. J Am Coll Cardiol 1999; 34: 1560–1568.
Kozak M, Krivan L, Semrad B . Circadian variations in the occurrence of ventricular tachyarrhythmias in patients with implantable cardioverter defibrillators. Pacing Clin Electrophysiol 2003; 26: 731–735.
Willich SN, Maclure M, Mittleman M, Arntz HR, Muller JE . Sudden cardiac death. Support for a role of triggering in causation. Circulation 1993; 87: 1442–1450.
Sharkey SW, Lesser JR, Garberich RF, Pink VR, Maron MS, Maron BJ . Comparison of circadian rhythm patterns in Tako-tsubo cardiomyopathy versus ST-segment elevation myocardial infarction. Am J Cardiol 2012; 110: 795–799.
Mahmoud KD, Lennon RJ, Ting HH, Rihal CS, Holmes DR Jr . Circadian variation in coronary stent thrombosis. JACC Cardiovasc Interv 2011; 4: 183–190.
Sumiyoshi M, Kojima S, Arima M, Suwa S, Nakazato Y, Sakurai H, Kanoh T, Nakata Y, Daida H . Circadian, weekly, and seasonal variation at the onset of acute aortic dissection. Am J Cardiol 2002; 89: 619–623.
Seguchi M, Wada H, Sakakura K, Nakagawa T, Ibe T, Ikeda N, Sugawara Y, Ako J, Momomura S . Circadian variation of acute aortic dissection. Int Heart J 2015; 56: 324–328.
Itaya H, Takagi T, Sugi K, Nakamura M . Absence of circadian variation of acute coronary syndrome onset in chronic kidney disease patients. Cardiovasc Revasc Med 2011; 12: 362–366.
Culic V . Daylight saving time transitions and acute myocardial infarction. Chronobiol Int 2013; 30: 662–668.
Partch CL, Green CB, Takahashi JS . Molecular architecture of the mammalian circadian clock. Trends Cell Biol 2014; 24: 90–99.
Takeda N, Maemura K . The role of clock genes and circadian rhythm in the development of cardiovascular diseases. Cell Mol Life Sci 2015; 72: 3225–3234.
Bunger MK, Wilsbacher LD, Moran SM, Clendenin C, Radcliffe LA, Hogenesch JB, Simon MC, Takahashi JS, Bradfield CA . Mop3 is an essential component of the master circadian pacemaker in mammals. Cell 2000; 103: 1009–1017.
Balsalobre A, Damiola F, Schibler U . A serum shock induces circadian gene expression in mammalian tissue culture cells. Cell 1998; 93: 929–937.
Maemura K, de la Monte SM, Chin MT, Layne MD, Hsieh CM, Yet SF, Perrella MA, Lee ME . CLIF a novel cycle-like factor, regulates the circadian oscillation of plasminogen activator inhibitor-1 gene expression. J Biol Chem 2000; 275: 36847–36851.
Polidarova L, Sladek M, Novakova M, Parkanova D, Sumova A . Increased sensitivity of the circadian system to temporal changes in the feeding regime of spontaneously hypertensive rats - a potential role for Bmal2 in the liver. PLoS ONE 2013; 8: e75690.
Maemura K, Layne MD, Watanabe M, Perrell MA, Nagai R, Lee ME . Molecular mechanisms of morning onset of myocardial infarction. Ann NY Acad Sci 2001; 947: 398–402.
Hermida RC, Ayala DE, Portaluppi F . Circadian variation of blood pressure: the basis for the chronotherapy of hypertension. Adv Drug Deliv Rev 2007; 59: 904–922.
Millar-Craig MW, Bishop CN, Raftery EB . Circadian variation of blood-pressure. Lancet 1978; 1: 795–797.
Obayashi K, Saeki K, Tone N, Kurumatani N . Relationship between melatonin secretion and nighttime blood pressure in elderly individuals with and without antihypertensive treatment: a cross-sectional study of the HEIJO-KYO cohort. Hypertens Res 2014; 37: 908–913.
Kovanen L, Donner K, Kaunisto M, Partonen T . CRY1, CRY2 and PRKCDBP genetic variants in metabolic syndrome. Hypertens Res 2015; 38: 186–192.
Curtis AM, Cheng Y, Kapoor S, Reilly D, Price TS, Fitzgerald GA . Circadian variation of blood pressure and the vascular response to asynchronous stress. Proc Natl Acad Sci USA 2007; 104: 3450–3455.
Westgate EJ, Cheng Y, Reilly DF, Price TS, Walisser JA, Bradfield CA, FitzGerald GA . Genetic components of the circadian clock regulate thrombogenesis in vivo. Circulation 2008; 117: 2087–2095.
Wang N, Yang G, Jia Z, Zhang H, Aoyagi T, Soodvilai S, Symons JD, Schnermann JB, Gonzalez FJ, Litwin SE, Yang T . Vascular PPARgamma controls circadian variation in blood pressure and heart rate through Bmal1. Cell Metab 2008; 8: 482–491.
Doi M, Takahashi Y, Komatsu R, Yamazaki F, Yamada H, Haraguchi S, Emoto N, Okuno Y, Tsujimoto G, Kanematsu A, Ogawa O, Todo T, Tsutsui K, van der Horst GT, Okamura H . Salt-sensitive hypertension in circadian clock-deficient Cry-null mice involves dysregulated adrenal Hsd3b6. Nat Med 2010; 16: 67–74.
Stow LR, Richards J, Cheng KY, Lynch IJ, Jeffers LA, Greenlee MM, Cain BD, Wingo CS, Gumz ML . The circadian protein period 1 contributes to blood pressure control and coordinately regulates renal sodium transport genes. Hypertension 2012; 59: 1151–1156.
Gumz ML, Popp MP, Wingo CS, Cain BD . Early transcriptional effects of aldosterone in a mouse inner medullary collecting duct cell line. Am J Physiol Renal Physiol 2003; 285: F664–F673.
Zuber AM, Centeno G, Pradervand S, Nikolaeva S, Maquelin L, Cardinaux L, Bonny O, Firsov D . Molecular clock is involved in predictive circadian adjustment of renal function. Proc Natl Acad Sci USA 2009; 106: 16523–16528.
Bridges AB, McLaren M, Scott NA, Pringle TH, McNeill GP, Belch JJ . Circadian variation of tissue plasminogen activator and its inhibitor, von Willebrand factor antigen, and prostacyclin stimulating factor in men with ischaemic heart disease. Br Heart J 1993; 69: 121–124.
Naito Y, Tsujino T, Kawasaki D, Okumura T, Morimoto S, Masai M, Sakoda T, Fujioka Y, Ohyanagi M, Iwasaki T . Circadian gene expression of clock genes and plasminogen activator inhibitor-1 in heart and aorta of spontaneously hypertensive and Wistar-Kyoto rats. J Hypertens 2003; 21: 1107–1115.
Kurnik PB . Circadian variation in the efficacy of tissue-type plasminogen activator. Circulation 1995; 91: 1341–1346.
Bagai K, Muldowney JA 3rd, Song Y, Wang L, Bagai J, Artibee KJ, Vaughan DE, Malow BA . Circadian variability of fibrinolytic markers and endothelial function in patients with obstructive sleep apnea. Sleep 2014; 37: 359–367.
Scheer FA, Shea SA . Human circadian system causes a morning peak in prothrombotic plasminogen activator inhibitor-1 (PAI-1) independent of the sleep/wake cycle. Blood 2014; 123: 590–593.
Schoenhard JA, Smith LH, Painter CA, Eren M, Johnson CH, Vaughan DE . Regulation of the PAI-1 promoter by circadian clock components: differential activation by BMAL1 and BMAL2. J Mol Cell Cardiol 2003; 35: 473–481.
Kobrin I, Oigman W, Kumar A, Ventura HO, Messerli FH, Frohlich ED, Dunn FG . Diurnal variation of blood pressure in elderly patients with essential hypertension. J Am Geriatr Soc 1984; 32: 896–899.
Guney HZ, Hodoglugil U, Uluoglu C, Gorgun CZ, Ercan ZS, Abacioglu N, Zengil H . In vitro susceptibility rhythms. II. Biological-time-dependent differences in effect of beta 1- and beta 2-adrenergic agonists of rat aorta and influence of endothelium. Chronobiol Int 1998; 15: 159–172.
Otto ME, Svatikova A, Barretto RB, Santos S, Hoffmann M, Khandheria B, Somers V . Early morning attenuation of endothelial function in healthy humans. Circulation 2004; 109: 2507–2510.
Fujita M, Franklin D . Diurnal changes in coronary blood flow in conscious dogs. Circulation 1987; 76: 488–491.
Saito T, Hirano M, Ide T, Ichiki T, Koibuchi N, Sunagawa K, Hirano K . Pivotal role of Rho-associated kinase 2 in generating the intrinsic circadian rhythm of vascular contractility. Circulation 2013; 127: 104–114.
Mohri M, Shimokawa H, Hirakawa Y, Masumoto A, Takeshita A . Rho-kinase inhibition with intracoronary fasudil prevents myocardial ischemia in patients with coronary microvascular spasm. J Am Coll Cardiol 2003; 41: 15–19.
Kikuchi Y, Yasuda S, Aizawa K, Tsuburaya R, Ito Y, Takeda M, Nakayama M, Ito K, Takahashi J, Shimokawa H . Enhanced Rho-kinase activity in circulating neutrophils of patients with vasospastic angina: a possible biomarker for diagnosis and disease activity assessment. J Am Coll Cardiol 2011; 58: 1231–1237.
Nihei T, Takahashi J, Tsuburaya R, Ito Y, Shiroto T, Hao K, Takagi Y, Matsumoto Y, Nakayama M, Miyata S, Sakata Y, Ito K, Shimokawa H . Circadian variation of Rho-kinase activity in circulating leukocytes of patients with vasospastic angina. Circ J 2014; 78: 1183–1190.
McNamara P, Seo SB, Rudic RD, Sehgal A, Chakravarti D, FitzGerald GA . Regulation of CLOCK and MOP4 by nuclear hormone receptors in the vasculature: a humoral mechanism to reset a peripheral clock. Cell 2001; 105: 877–889.
Nonaka H, Emoto N, Ikeda K, Fukuya H, Rohman MS, Raharjo SB, Yagita K, Okamura H, Yokoyama M . Angiotensin II induces circadian gene expression of clock genes in cultured vascular smooth muscle cells. Circulation 2001; 104: 1746–1748.
Takeda N, Maemura K, Horie S, Oishi K, Imai Y, Harada T, Saito T, Shiga T, Amiya E, Manabe I, Ishida N, Nagai R . Thrombomodulin is a clock-controlled gene in vascular endothelial cells. J Biol Chem 2007; 282: 32561–32567.
Chalmers JA, Martino TA, Tata N, Ralph MR, Sole MJ, Belsham DD . Vascular circadian rhythms in a mouse vascular smooth muscle cell line (Movas-1). Am J Physiol Regul Integr Comp Physiol 2008; 295: R1529–R1538.
Anea CB, Zhang M, Stepp DW, Simkins GB, Reed G, Fulton DJ, Rudic RD . Vascular disease in mice with a dysfunctional circadian clock. Circulation 2009; 119: 1510–1517.
Denniff M, Turrell HE, Vanezis A, Rodrigo GC . The time-of-day variation in vascular smooth muscle contractility depends on a nitric oxide signalling pathway. J Mol Cell Cardiol 2014; 66: 133–140.
Anea CB, Cheng B, Sharma S, Kumar S, Caldwell RW, Yao L, Ali MI, Merloiu AM, Stepp DW, Black SM, Fulton DJ, Rudic RD . Increased superoxide and endothelial NO synthase uncoupling in blood vessels of Bmal1-knockout mice. Circ Res 2012; 111: 1157–1165.
Kunieda T, Minamino T, Miura K, Katsuno T, Tateno K, Miyauchi H, Kaneko S, Bradfield CA, FitzGerald GA, Komuro I . Reduced nitric oxide causes age-associated impairment of circadian rhythmicity. Circ Res 2008; 102: 607–614.
Al Mheid I, Corrigan F, Shirazi F, Veledar E, Li Q, Alexander WR, Taylor WR, Waller EK, Quyyumi AA . Circadian variation in vascular function and regenerative capacity in healthy humans. J Am Heart Assoc 2014; 3: e000845.
Sun YY, Bai WW, Wang B, Lu XT, Xing YF, Cheng W, Liu XQ, Zhao YX . Period 2 is essential to maintain early endothelial progenitor cell function in vitro and angiogenesis after myocardial infarction in mice. J Cell Mol Med 2014; 18: 907–918.
Jensen LD, Cao Z, Nakamura M, Yang Y, Brautigam L, Andersson P, Zhang Y, Wahlberg E, Lanne T, Hosaka K, Cao Y . Opposing effects of circadian clock genes bmal1 and period2 in regulation of VEGF-dependent angiogenesis in developing zebrafish. Cell Rep 2012; 2: 231–241.
Xie Z, Su W, Liu S, Zhao G, Esser K, Schroder EA, Lefta M, Stauss HM, Guo Z, Gong MC . Smooth-muscle BMAL1 participates in blood pressure circadian rhythm regulation. J Clin Invest 2015; 125: 324–336.
Cheng B, Anea CB, Yao L, Chen F, Patel V, Merloiu A, Pati P, Caldwell RW, Fulton DJ, Rudic RD . Tissue-intrinsic dysfunction of circadian clock confers transplant arteriosclerosis. Proc Natl Acad Sci USA 2011; 108: 17147–17152.
Durgan DJ, Hotze MA, Tomlin TM, Egbejimi O, Graveleau C, Abel ED, Shaw CA, Bray MS, Hardin PE, Young ME . The intrinsic circadian clock within the cardiomyocyte. Am J Physiol Heart Circ Physiol 2005; 289: H1530–H1541.
Storch KF, Lipan O, Leykin I, Viswanathan N, Davis FC, Wong WH, Weitz CJ . Extensive and divergent circadian gene expression in liver and heart. Nature 2002; 417: 78–83.
Yamashita T, Sekiguchi A, Iwasaki YK, Sagara K, Iinuma H, Hatano S, Fu LT, Watanabe H . Circadian variation of cardiac K+ channel gene expression. Circulation 2003; 107: 1917–1922.
Young ME, Razeghi P, Cedars AM, Guthrie PH, Taegtmeyer H . Intrinsic diurnal variations in cardiac metabolism and contractile function. Circ Res 2001; 89: 1199–1208.
Young ME, Razeghi P, Taegtmeyer H . Clock genes in the heart: characterization and attenuation with hypertrophy. Circ Res 2001; 88: 1142–1150.
Durgan DJ, Pulinilkunnil T, Villegas-Montoya C, Garvey ME, Frangogiannis NG, Michael LH, Chow CW, Dyck JR, Young ME . Short communication: ischemia/reperfusion tolerance is time-of-day-dependent: mediation by the cardiomyocyte circadian clock. Circ Res 2010; 106: 546–550.
Reiter R, Swingen C, Moore L, Henry TD, Traverse JH . Circadian dependence of infarct size and left ventricular function after ST elevation myocardial infarction. Circ Res 2012; 110: 105–110.
Kung TA, Egbejimi O, Cui J, Ha NP, Durgan DJ, Essop MF, Bray MS, Shaw CA, Hardin PE, Stanley WC, Young ME . Rapid attenuation of circadian clock gene oscillations in the rat heart following ischemia-reperfusion. J Mol Cell Cardiol 2007; 43: 744–753.
Young ME, Wilson CR, Razeghi P, Guthrie PH, Taegtmeyer H . Alterations of the circadian clock in the heart by streptozotocin-induced diabetes. J Mol Cell Cardiol 2002; 34: 223–231.
Hurd MW, Ralph MR . The significance of circadian organization for longevity in the golden hamster. J Biol Rhythms 1998; 13: 430–436.
Penev PD, Kolker DE, Zee PC, Turek FW . Chronic circadian desynchronization decreases the survival of animals with cardiomyopathic heart disease. Am J Physiol 1998; 275: H2334–H2337.
Alibhai FJ, Tsimakouridze EV, Chinnappareddy N, Wright DC, Billia F, O'Sullivan ML, Pyle WG, Sole MJ, Martino TA . Short-term disruption of diurnal rhythms after murine myocardial infarction adversely affects long-term myocardial structure and function. Circ Res 2014; 114: 1713–1722.
Gill S, Le HD, Melkani GC, Panda S . Time-restricted feeding attenuates age-related cardiac decline in Drosophila. Science 2015; 347: 1265–1269.
Vetter C, Fischer D, Matera JL, Roenneberg T . Aligning work and circadian time in shift workers improves sleep and reduces circadian disruption. Curr Biol 2015; 25: 907–911.
Alvarez JD, Hansen A, Ord T, Bebas P, Chappell PE, Giebultowicz JM, Williams C, Moss S, Sehgal A . The circadian clock protein BMAL1 is necessary for fertility and proper testosterone production in mice. J Biol Rhythms 2008; 23: 26–36.
Andrews JL, Zhang X, McCarthy JJ, McDearmon EL, Hornberger TA, Russell B, Campbell KS, Arbogast S, Reid MB, Walker JR, Hogenesch JB, Takahashi JS, Esser KA . CLOCK and BMAL1 regulate MyoD and are necessary for maintenance of skeletal muscle phenotype and function. Proc Natl Acad Sci USA 2010; 107: 19090–19095.
Lefta M, Campbell KS, Feng HZ, Jin JP, Esser KA . Development of dilated cardiomyopathy in Bmal1-deficient mice. Am J Physiol Heart Circ Physiol 2012; 303: H475–H485.
Durgan DJ, Tsai JY, Grenett MH, Pat BM, Ratcliffe WF, Villegas-Montoya C, Garvey ME, Nagendran J, Dyck JR, Bray MS, Gamble KL, Gimble JM, Young ME . Evidence suggesting that the cardiomyocyte circadian clock modulates responsiveness of the heart to hypertrophic stimuli in mice. Chronobiol Int 2011; 28: 187–203.
Bray MS, Shaw CA, Moore MW, Garcia RA, Zanquetta MM, Durgan DJ, Jeong WJ, Tsai JY, Bugger H, Zhang D, Rohrwasser A, Rennison JH, Dyck JR, Litwin SE, Hardin PE, Chow CW, Chandler MP, Abel ED, Young ME . Disruption of the circadian clock within the cardiomyocyte influences myocardial contractile function, metabolism, and gene expression. Am J Physiol Heart Circ Physiol 2008; 294: H1036–H1047.
Durgan DJ, Trexler NA, Egbejimi O, McElfresh TA, Suk HY, Petterson LE, Shaw CA, Hardin PE, Bray MS, Chandler MP, Chow CW, Young ME . The circadian clock within the cardiomyocyte is essential for responsiveness of the heart to fatty acids. J Biol Chem 2006; 281: 24254–24269.
Young ME, Brewer RA, Peliciari-Garcia RA, Collins HE, He L, Birky TL, Peden BW, Thompson EG, Ammons BJ, Bray MS, Chatham JC, Wende AR, Yang Q, Chow CW, Martino TA, Gamble KL . Cardiomyocyte-specific BMAL1 plays critical roles in metabolism, signaling, and maintenance of contractile function of the heart. J Biol Rhythms 2014; 29: 257–276.
Bexton RS, Vallin HO, Camm AJ . Diurnal variation of the QT interval—influence of the autonomic nervous system. Br Heart J 1986; 55: 253–258.
Cinca J, Moya A, Bardaji A, Rius J, Soler-Soler J . Circadian variations of electrical properties of the heart. Ann NY Acad Sci 1990; 601: 222–233.
Oda E, Aizawa Y, Arai Y, Shibata A . Diurnal variation of QT interval in patients with VVI pacemaker. Tohoku J Exp Med 1985; 145: 419–426.
Schroder EA, Lefta M, Zhang X, Bartos DC, Feng HZ, Zhao Y, Patwardhan A, Jin JP, Esser KA, Delisle BP . The cardiomyocyte molecular clock, regulation of Scn5a, and arrhythmia susceptibility. Am J Physiol Cell Physiol 2013; 304: C954–C965.
Jeyaraj D, Haldar SM, Wan X, McCauley MD, Ripperger JA, Hu K, Lu Y, Eapen BL, Sharma N, Ficker E, Cutler MJ, Gulick J, Sanbe A, Robbins J, Demolombe S, Kondratov RV, Shea SA, Albrecht U, Wehrens XH, Rosenbaum DS, Jain MK . Circadian rhythms govern cardiac repolarization and arrhythmogenesis. Nature 2012; 483: 96–99.
Patton KK, Hellkamp AS, Lee KL, Mark DB, Johnson GW, Anderson J, Bardy GH, Poole JE . Unexpected deviation in circadian variation of ventricular arrhythmias: the SCD-HeFT (Sudden Cardiac Death in Heart Failure Trial). J Am Coll Cardiol 2014; 63: 2702–2708.
Alex A, Li A, Zeng X, Tate RE, McKee ML, Capen DE, Zhang Z, Tanzi RE, Zhou C . A circadian clock gene, cry, affects heart morphogenesis and function in drosophila as revealed by optical coherence microscopy. PLoS ONE 2015; 10: e0137236.
Nguyen KD, Fentress SJ, Qiu Y, Yun K, Cox JS, Chawla A . Circadian gene Bmal1 regulates diurnal oscillations of Ly6C(hi) inflammatory monocytes. Science 2013; 341: 1483–1488.
Sato S, Sakurai T, Ogasawara J, Takahashi M, Izawa T, Imaizumi K, Taniguchi N, Ohno H, Kizaki T . A circadian clock gene, Rev-erbalpha, modulates the inflammatory function of macrophages through the negative regulation of Ccl2 expression. J Immunol 2014; 192: 407–417.
Ma H, Zhong W, Jiang Y, Fontaine C, Li S, Fu J, Olkkonen V M, Staels B, Yan D . Increased atherosclerotic lesions in LDL receptor deficient mice with hematopoietic nuclear receptor Rev-erbalpha knock- down. J Am Heart Assoc 2013; 2: e000235.
Grimaldi B, Bellet MM, Katada S, Astarita G, Hirayama J, Amin RH, Granneman JG, Piomelli D, Leff T, Sassone-Corsi P . PER2 controls lipid metabolism by direct regulation of PPARgamma. Cell Metab 2010; 12: 509–520.
Yamaguchi M, Uemura H, Arisawa K, Katsuura-Kamano S, Hamajima N, Hishida A, Suma S, Oze I, Nakamura K, Takashima N, Suzuki S, Ibusuki R, Mikami H, Ohnaka K, Kuriyama N, Kubo M, Tanaka H . Association between brain-muscle-ARNT-like protein-2 (BMAL2) gene polymorphism and type 2 diabetes mellitus in obese Japanese individuals: a cross-sectional analysis of the Japan Multi-institutional Collaborative Cohort Study. Diabetes Res Clin Pract 2015; 110: 301–308.
Eckle T, Hartmann K, Bonney S, Reithel S, Mittelbronn M, Walker LA, Lowes BD, Han J, Borchers CH, Buttrick PM, Kominsky DJ, Colgan SP, Eltzschig HK . Adora2b-elicited Per2 stabilization promotes a HIF-dependent metabolic switch crucial for myocardial adaptation to ischemia. Nat Med 2012; 18: 774–782.
Egg M, Koblitz L, Hirayama J, Schwerte T, Folterbauer C, Kurz A, Fiechtner B, Most M, Salvenmoser W, Sassone-Corsi P, Pelster B . Linking oxygen to time: the bidirectional interaction between the hypoxic signaling pathway and the circadian clock. Chronobiol Int 2013; 30: 510–529.
Acknowledgements
This study was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science, Japan (to KM and NT, 25461113).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Takeda, N., Maemura, K. Circadian clock and the onset of cardiovascular events. Hypertens Res 39, 383–390 (2016). https://doi.org/10.1038/hr.2016.9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2016.9
Keywords
This article is cited by
-
Neutrophil extracellular traps in systemic autoimmune and autoinflammatory diseases
Nature Reviews Immunology (2023)
-
In-vitro antiplatelet effect of melatonin in healthy individuals and patients with type 2 diabetes mellitus
Journal of Endocrinological Investigation (2023)
-
Serial assessment of thrombogenicity and hemodynamics in patients with type II diabetes in a clinical research unit: Evidence for circadian variations in clot formation
Journal of Thrombosis and Thrombolysis (2022)
-
Methamphetamine-induced changes in myocardial gene transcription are sex-dependent
BMC Genomics (2021)
-
A disturbance beyond the barrier—chronic kidney disease allows angiotensinogen invasion
Hypertension Research (2021)