Abstract
Objective:
Obesity is associated with an increase in various pro-inflammatory and anti-inflammatory cytokines, but the interplay of these cytokines is incompletely understood. We conducted experiments to test a broader hypothesis that a dynamic interplay of pro-inflammatory and anti-inflammatory cytokines controls lipid storage in adipocytes.
Design:
Three experiments were designed to test the overall hypothesis that proinflammatory cytokine (for example, tumor necrosis factor-α (TNF-α) inhibits anti-inflammatory cytokine (for example, adiponectin) activity in an attempt to limit excess lipid accumulation in adipocytes.
Results:
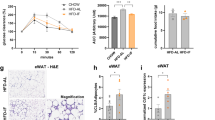
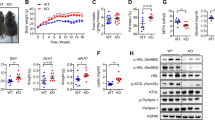
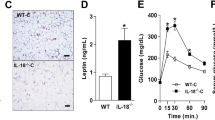
Experiment one showed that in pro-inflammatory animal models (ap2-P65, ob/ob and high-fat diet-induced obese mice), the increase in TNF-α expression was associated with a decrease in adiponectin expression. Experiment two showed that in 3T3-L1 adipocytes, TNF-α significantly reduced lipid accumulation and glucose uptake induced by adiponectin, and increased lipolysis. Experiment three showed that in 3T3-L1 adipocytes, TNF-α reduced mRNA and protein expression of adiponectin. Adiponectin gene transcription and mRNA stability were both reduced by TNF-α. The expression of peroxisome proliferator-activated receptor gamma, an activator of adiponectin gene promoter, was reduced by TNF-α. The inhibitory activity of TNF-α was blocked by the chemical inhibitors of NF-κB and super suppressor IκBα.
Conclusions:
TNF-α opposes the action of adiponectin in the regulation of lipid metabolism, and inhibits adiponectin expression at transcriptional and post-transcriptional levels. The results suggest that pro-inflammatory cytokine inhibit anti-inflammatory cytokine in adipocytes to reduce lipid storage. This suggests a potential role of anti-inflammatory cytokines in the control of adipose tissue expansion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Chavarro JE, Ehrlich S, Colaci DS, Wright DL, Toth TL, Petrozza JC et al. Body mass index and short-term weight change in relation to treatment outcomes in women undergoing assisted reproduction. Fertil Steril 2012; 98: 109–116.
Frisch RE . Population, food intake, and fertility. There is historical evidence for a direct effect of nutrition on reproductive ability. Science 1978; 199: 22–30.
Scott EC, Johnston FE . Critical fat, menarche, and the maintenance of menstrual cycles: a critical review. J Adolesc Health Care 1982; 2: 249–260.
Jensen TK, Andersson AM, Jorgensen N, Andersen AG, Carlsen E, Petersen JH et al. Body mass index in relation to semen quality and reproductive hormones among 1,558 Danish men. Fertil Steril 2004; 82: 863–870.
Shayeb AG, Harrild K, Mathers E, Bhattacharya S . An exploration of the association between male body mass index and semen quality. Reprod Biomed Online 2011; 23: 717–723.
Veleva Z, Tiitinen A, Vilska S, Hyden-Granskog C, Tomas C, Martikainen H et al. High and low BMI increase the risk of miscarriage after IVF/ICSI and FET. Human Reprod 2008; 23: 878–884.
Tilg H, Moschen AR . Adipocytokines: mediators linking adipose tissue, inflammation and immunity. Nat Rev Immunol 2006; 6: 772–783.
Moschos S, Chan JL, Mantzoros CS . Leptin and reproduction: a review. Fertil Sterility 2002; 77: 433–444.
Matsubara M, Maruoka S, Katayose S . Inverse relationship between plasma adiponectin and leptin concentrations in normal-weight and obese women. Eur J Endocrinol 2002; 147: 173–180.
Stryjecki C, Mutch DM . Fatty acid-gene interactions, adipokines and obesity. Eur J Clin Nutr 2011; 65: 285–297.
Hotamisligil GS . Inflammation and metabolic disorders. Nature 2006; 444: 860–867.
Bastard JP, Maachi M, Lagathu C, Kim MJ, Caron M, Vidal H et al. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw 2006; 17: 4–12.
Berg AH, Combs TP, Scherer PE . ACRP30/adiponectin: an adipokine regulating glucose and lipid metabolism. Trends Endocrinol Metab 2002; 13: 84–89.
Qiao L, Kinney B, Schaack J, Shao J . Adiponectin inhibits lipolysis in mouse adipocytes. Diabetes 2011; 60: 1519–1527.
Kadowaki T, Yamauchi T . Adiponectin and adiponectin receptors. Endocr Rev 2005; 26: 439–451.
Mauer J, Chaurasia B, Plum L, Quast T, Hampel B, Bluher M et al. Myeloid cell-restricted insulin receptor deficiency protects against obesity-induced inflammation and systemic insulin resistance. PLoS genetics 2010; 6: e1000938.
Hotamisligil GS, Shargill NS, Spiegelman BM . Adipose expression of tumor necrosis factor-alpha: direct role in obesity-linked insulin resistance. Science 1993; 259: 87–91.
Gao Z, He Q, Peng B, Chiao PJ, Ye J . Regulation of nuclear translocation of hdac3 by ikba is required for tumor necrosis factor inhibition of peroxisome proliferator-activated receptor {gamma} function. J Biol Chem 2006; 281: 4540–4547.
Gao Z, Hwang D, Bataille F, Lefevre M, York D, Quon MJ et al. Serine phosphorylation of insulin receptor substrate 1 by inhibitor KappaB kinase complex. J Biol Chem 2002; 277: 48115–48121.
Tang T, Zhang J, Yin J, Staszkiewicz J, Gawronska-Kozak B, Mynatt R et al. Uncoupling of Inflammation and Insulin Resistance by NF-kB in Transgenic Mice through Induction of Energy Expenditure. J Biol Chem 2010; 285: 4637–4644.
Ye J . Regulation of PPARg function by TNF-a. Biochem Biophys Res Commun 2008; 374: 405–408.
Baker RG, Hayden MS, NF-kappaB GhoshS . Inflammation, and metabolic disease. Cell Metab 2011; 13: 11–22.
Ye J, Gao Z, Yin J, He H . Hypoxia is a potential risk factor for chronic inflammation and adiponectin reduction in adipose tissue of ob/ob and dietary obese mice. Am J Physiol Endocrinol Metab 2007; 293: E1118–E1128.
Rogers PM, Mashtalir N, Rathod MA, Dubuisson O, Wang Z, Dasuri K et al. Metabolically favorable remodeling of human adipose tissue by human adenovirus type 36. Diabetes 2008; 57: 2321–2331.
Wang ZQ, Cefalu WT, Zhang XH, Yu Y, Qin J, Son L et al. Human adenovirus type 36 enhances glucose uptake in diabetic and nondiabetic human skeletal muscle cells independent of insulin signaling. Diabetes 2008; 57: 1805–1813.
Rajala MW, Scherer PE . Minireview: The adipocyte—at the crossroads of energy homeostasis, inflammation, and atherosclerosis. Endocrinology 2003; 144: 3765–3773.
Cawthorn WP, Heyd F, Hegyi K, Sethi JK . Tumour necrosis factor-alpha inhibits adipogenesis via a beta-catenin/TCF4(TCF7L2)-dependent pathway. Cell Death Differ 2007; 14: 1361–1373.
Hector J, Schwarzloh B, Goehring J, Strate TG, Hess UF, Deuretzbacher G et al. TNF-alpha alters visfatin and adiponectin levels in human fat. Horm Metab Res 2007; 39: 250–255.
Zappala G, Rechler MM . IGFBP-3, hypoxia and TNF-alpha inhibit adiponectin transcription. Biochem Biophys Res Commun 2009; 382: 785–789.
Maeda N, Takahashi M, Funahashi T, Kihara S, Nishizawa H, Kishida K et al. PPARgamma ligands increase expression and plasma concentrations of adiponectin, an adipose-derived protein. Diabetes 2001; 50: 2094–2099.
Bruun JM, Lihn AS, Verdich C, Pedersen SB, Toubro S, Astrup A et al. Regulation of adiponectin by adipose tissue-derived cytokines: in vivo and in vitro investigations in humans. Am J Physiol Endocrinol Metab 2003; 285: E527–E533.
Iwaki M, Matsuda M, Maeda N, Funahashi T, Matsuzawa Y, Makishima M et al. Induction of adiponectin, a fat-derived antidiabetic and antiatherogenic factor, by nuclear receptors. Diabetes 2003; 52: 1655–1663.
Rosen ED, Walkey CJ, Puigserver P, Spiegelman BM . Transcriptional regulation of adipogenesis. Genes Dev 2000; 14: 1293–1307.
Zhang J, Henagan TM, Gao Z, Ye J . Inhibition of glyceroneogenesis by histone deacetylase 3 contributes to lipodystrophy in mice with adipose tissue inflammation. Endocrinology 2011; 152: 1829–1838.
Yamauchi T, Kamon J, Minokoshi Y, Ito Y, Waki H, Uchida S et al. Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med 2002; 8: 1288–1295.
Cheng SW, Fryer LG, Carling D, Shepherd PR . Thr2446 is a novel mammalian target of rapamycin (mTOR) phosphorylation site regulated by nutrient status. J Biol Chem 2004; 279: 15719–15722.
Zhang J, Gao Z, Yin J, Quon MJ, Ye J . S6K directly phosphorylates IRS-1 on Ser-270 to promote insulin resistance in response to TNF-α signaling through IKK2. J Biol Chem 2008; 283: 35375–35382.
Ouchi N, Kihara S, Arita Y, Okamoto Y, Maeda K, Kuriyama H et al. Adiponectin, an adipocyte-derived plasma protein, inhibits endothelial NF-kappaB signaling through a cAMP-dependent pathway. Circulation 2000; 102: 1296–1301.
Kim JY, van de Wall E, Laplante M, Azzara A, Trujillo ME, Hofmann SM et al. Obesity-associated improvements in metabolic profile through expansion of adipose tissue. J Clin Invest 2007; 117: 2621–2637.
Ye J . Emerging role of adipose tissue hypoxia in obesity and insulin resistance. Int J Obes 2009; 33: 54–66.
MohanKumar SM, Smith CL, MohanKumar PS . Central adaptation to chronic administration of interleukin-1beta (IL-1beta) in rats. Brain Res Bull 2003; 62: 71–76.
Matsuki T, Horai R, Sudo K, Iwakura Y . IL-1 plays an important role in lipid metabolism by regulating insulin levels under physiological conditions. J. Exp. Med 2003; 198: 877–888.
Wallenius V, Wallenius K, Ahren B, Rudling M, Carlsten H, Dickson SL et al. Interleukin-6-deficient mice develop mature-onset obesity. Nat Med 2002; 8: 75–79.
Netea MG, Joosten LA, Lewis E, Jensen DR, Voshol PJ, Kullberg BJ et al. Deficiency of interleukin-18 in mice leads to hyperphagia, obesity and insulin resistance. Nat Med 2006; 12: 650–656.
Jiao P, Feng B, Ma J, Nie Y, Paul E, Li Y et al. Constitutive activation of IKKβ in adipose tissue prevents diet-induced obesity in mice. Endocrinology 2012; 153: 154–165.
Ye J, Keller J . Regulation of energy metabolism by inflammation: A feedback response in obesity and calorie restriction. Aging 2010; 2: 361–368.
Donath MY, Shoelson SE . Type 2 diabetes as an inflammatory disease. Nat Rev Immunol 2011; 11: 98–107.
Ye J, McGuinness OP . Inflammation during obesity is not all bad: Evidence from animal and human studies. Am J Physiol Endocrinol Metab e-pub ahead of print 26 December 2012; PMID:23269411.
Acknowledgements
We appreciate the technical support from Ms Xin Ye. Quantitative real-time PCR assay was conducted in the genomics core of the Pennington Biomedical Research Center. The core is supported by the NIH grants (P30DK072476 and P20 GM103528). This work was supported by the National Institute of Health research projects (DK085495; DK068036) to Ye, J.
Author contributions
YW, HW, VH and OD, performed the experiments. VH, NVD, GZ and JY analyzed the data and wrote the manuscript. JY is fully responsible for this article. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Wang, Y., Wang, H., Hegde, V. et al. Interplay of pro- and anti-inflammatory cytokines to determine lipid accretion in adipocytes. Int J Obes 37, 1490–1498 (2013). https://doi.org/10.1038/ijo.2013.9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2013.9
Keywords
This article is cited by
-
An association between adropin hormone and total testosterone in obese men: a case-control study
BMC Endocrine Disorders (2022)
-
Cytokines and inflammation in adipogenesis: an updated review
Frontiers of Medicine (2019)
-
Intracellular ATP in balance of pro- and anti-inflammatory cytokines in adipose tissue with and without tissue expansion
International Journal of Obesity (2017)