Abstract
Background/Objectives:
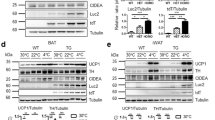
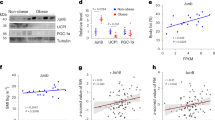
Thermogenic fat is present in humans and emerging evidence indicates that increasing the content and activity of these adipocytes may lead to weight loss and improved metabolic health. Multiple reporter systems have been developed to assay thermogenic fat activity based on the transcriptional and translational activation of Ucp1, the key molecule that mediates nonshivering thermogenesis. Our study aims to develop a much-needed tool to monitor thermogenic fat activity through a mechanism independent of Ucp1 regulation, therefore effectively assaying not only canonical β-adrenergic activation but also various non-UCP1-mediated thermogenic pathways that have been increasingly appreciated.
Methods:
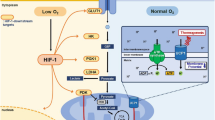
We detected increased luciferase activity upon thermogenic activation in interscapular brown and inguinal subcutaneous fat in ODD-Luc mice, a hypoxia reporter mouse model. We then developed an OLTAM ( O DD- L uc based T hermogenic A ctivity M easurement) system to assay thermogenic fat cell activity.
Results:
In both primary murine and human adipocytes and an immortalized adipose cell line that were transduced with the OLTAM system, luciferase activity can be readily measured and visualized by bioluminescence imaging in response to a variety of stimuli, including UCP1-independent thermogenic signaling. This system can offer a convenient method to assay thermogenic activity for both basic and translational research.
Conclusions:
The OLTAM system offers a convenient way to measure the activation of thermogenic fat and presents opportunities to discover novel signaling pathways and unknown compounds targeting metabolically active adipocytes to counteract human obesity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kajimura S, Spiegelman BM, Seale P . Brown and beige fat: physiological roles beyond heat generation. Cell Metab 2015; 22: 546–559.
Hanssen MJ, Hoeks J, Brans B, van der Lans AA, Schaart G, van den Driessche JJ et al. Short-term cold acclimation improves insulin sensitivity in patients with type 2 diabetes mellitus. Nat med 2015; 21: 863–865.
Sidossis L, Kajimura S . Brown and beige fat in humans: thermogenic adipocytes that control energy and glucose homeostasis. J clin invest 2015; 125: 478–486.
Galmozzi A, Sonne SB, Altshuler-Keylin S, Hasegawa Y, Shinoda K, Luijten IH et al. ThermoMouse: an in vivo model to identify modulators of UCP1 expression in brown adipose tissue. Cell rep 2014; 9: 1584–1593.
Mao L, Nie B, Nie T, Hui X, Gao X, Lin X et al. Visualization and quantification of browning using a Ucp1-2A-luciferase knock-in mouse model. Diabetes 2017; 66: 407–417.
Kazak L, Chouchani ET, Jedrychowski MP, Erickson BK, Shinoda K, Cohen P et al. A creatine-driven substrate cycle enhances energy expenditure and thermogenesis in beige fat. Cell 2015; 163: 643–655.
Shabalina IG, Kramarova TV, Nedergaard J, Cannon B . Carboxyatractyloside effects on brown-fat mitochondria imply that the adenine nucleotide translocator isoforms ANT1 and ANT2 may be responsible for basal and fatty-acid-induced uncoupling respectively. Biochem J 2006; 399: 405–414.
Long JZ, Svensson KJ, Bateman LA, Lin H, Kamenecka T, Lokurkar IA et al. The secreted enzyme PM20D1 regulates lipidated amino acid uncouplers of mitochondria. Cell 2016; 166: 424–435.
Foster DO, Frydman ML . Nonshivering thermogenesis in the rat. II. Measurements of blood flow with microspheres point to brown adipose tissue as the dominant site of the calorigenesis induced by noradrenaline. Can J Physiol Pharmacol 1978; 56: 110–122.
Xue Y, Petrovic N, Cao R, Larsson O, Lim S, Chen S et al. Hypoxia-independent angiogenesis in adipose tissues during cold acclimation. Cell Metab 2009; 9: 99–109.
Safran M, Kim WY, O'Connell F, Flippin L, Gunzler V, Horner JW et al. Mouse model for noninvasive imaging of HIF prolyl hydroxylase activity: assessment of an oral agent that stimulates erythropoietin production. Proc Natl Acad Sci USA 2006; 103: 105–110.
Cannon B, Nedergaard J . Brown adipose tissue: function and physiological significance. Physiol Rev 2004; 84: 277–359.
Wu J, Bostrom P, Sparks LM, Ye L, Choi JH, Giang AH et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012; 150: 366–376.
Handschin C, Spiegelman BM . Peroxisome proliferator-activated receptor gamma coactivator 1 coactivators, energy homeostasis, and metabolism. Endocr Rev 2006; 27: 728–735.
Wilson-Fritch L, Burkart A, Bell G, Mendelson K, Leszyk J, Nicoloro S et al. Mitochondrial biogenesis and remodeling during adipogenesis and in response to the insulin sensitizer rosiglitazone. Mol cell biol 2003; 23: 1085–1094.
Ohno H, Shinoda K, Spiegelman BM, Kajimura S . PPARgamma agonists induce a white-to-brown fat conversion through stabilization of PRDM16 protein. Cell Metab 2012; 15: 395–404.
Kajimura S, Seale P, Kubota K, Lunsford E, Frangioni JV, Gygi SP et al. Initiation of myoblast to brown fat switch by a PRDM16-C/EBP-beta transcriptional complex. Nature 2009; 460: 1154–1158.
Elsen M, Raschke S, Tennagels N, Schwahn U, Jelenik T, Roden M et al. BMP4 and BMP7 induce the white-to-brown transition of primary human adipose stem cells. Am J Physiol Cell Physiol 2014; 306: C431–C440.
Bartesaghi S, Hallen S, Huang L, Svensson PA, Momo RA, Wallin S et al. Thermogenic activity of UCP1 in human white fat-derived beige adipocytes. Mol Endocrinol 2015; 29: 130–139.
Odegaard JI, Lee MW, Sogawa Y, Bertholet AM, Locksley RM, Weinberg DE et al. Perinatal licensing of thermogenesis by IL-33 and ST2. Cell 2016; 166: 841–854.
Takahashi A, Adachi S, Morita M, Tokumasu M, Natsume T, Suzuki T et al. Post-transcriptional stabilization of Ucp1 mRNA protects mice from diet-induced obesity. Cell Rep 2015; 13: 2756–2767.
Chouchani ET, Kazak L, Jedrychowski MP, Lu GZ, Erickson BK, Szpyt J et al. Mitochondrial ROS regulate thermogenic energy expenditure and sulfenylation of UCP1. Nature 2016; 532: 112–116.
Ouellet V, Labbe SM, Blondin DP, Phoenix S, Guerin B, Haman F et al. Brown adipose tissue oxidative metabolism contributes to energy expenditure during acute cold exposure in humans. J Clin Invest 2012; 122: 545–552.
Muzik O, Mangner TJ, Leonard WR, Kumar A, Janisse J, Granneman JG . 15O PET measurement of blood flow and oxygen consumption in cold-activated human brown fat. J Nucl Med 2013; 54: 523–531.
Krohn KA, Link JM, Mason RP . Molecular imaging of hypoxia. J Nucl Med 2008; 49 (Suppl 2): 129S–148S.
Acknowledgements
This research was supported by the Edward Mallinckrodt Jr. Foundation Mallinckrodt grant (to JW), NIH Grant R01 DK107583 (to JW), NIH Grant R01 DK095201 (to YMS), NIH Grant R01 DK47918 (to MBO), NIH Grant F31DK112625 (to MPE), a postdoctoral fellowship from the American Heart Association 17POST33060001 (to D-IK), Clinical and Translational Science Award Grant UL1TR000433 (to M-JP) and NIH grant P30 DK34933 to the University of Michigan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Kim, DI., Liao, J., Emont, M. et al. An OLTAM system for analysis of brown/beige fat thermogenic activity. Int J Obes 42, 939–945 (2018). https://doi.org/10.1038/ijo.2017.308
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.308
This article is cited by
-
Hypoxia-induced signaling in the cardiovascular system: pathogenesis and therapeutic targets
Signal Transduction and Targeted Therapy (2023)