Abstract
Microbial communities have a central role in ecosystem processes by driving the Earth’s biogeochemical cycles. However, the importance of microbial diversity for ecosystem functioning is still debated. Here, we experimentally manipulated the soil microbial community using a dilution approach to analyze the functional consequences of diversity loss. A trait-centered approach was embraced using the denitrifiers as model guild due to their role in nitrogen cycling, a major ecosystem service. How various diversity metrics related to richness, eveness and phylogenetic diversity of the soil denitrifier community were affected by the removal experiment was assessed by 454 sequencing. As expected, the diversity metrics indicated a decrease in diversity in the 1/103 and 1/105 dilution treatments compared with the undiluted one. However, the extent of dilution and the corresponding reduction in diversity were not commensurate, as a dilution of five orders of magnitude resulted in a 75% decrease in estimated richness. This reduction in denitrifier diversity resulted in a significantly lower potential denitrification activity in soil of up to 4–5 folds. Addition of wheat residues significantly increased differences in potential denitrification between diversity levels, indicating that the resource level can influence the shape of the microbial diversity–functioning relationship. This study shows that microbial diversity loss can alter terrestrial ecosystem processes, which suggests that the importance of functional redundancy in soil microbial communities has been overstated.
Similar content being viewed by others
Introduction
The consequences of decline in biodiversity for ecosystem processes and functioning have long been of considerable interest (Chapin et al., 2000; Loreau et al., 2001; Wardle et al., 2011). Current understanding indicates that in general higher levels of biodiversity correspond to increased ecosystem functioning; however, the magnitude of this effect varies among ecosystem properties and the studied communities (Hooper et al., 2005). The most influential studies have focused on the relationship between plant diversity and primary production in grassland ecosystems, yet despite being central in driving the Earth’s biogeochemical cycles, the role of microbial diversity has been neglected until the last decade.
The significance of biodiversity loss is challenged by the concept of functional redundancy. Since different species can have the same function in ecosystems, functional redundancy predicts that the loss of species does not necessarily alter ecosystem functioning because of their replacement by other species for maintaining processes (Loreau, 2004). In terrestrial ecosystems where microbial diversity is orders of magnitude higher than in aquatic environments (Torsvik et al., 2002), a high functional redundancy is expected (Nannipieri et al., 2003). Schimel (1995) proposed that functional redundancy is greater for processes carried out by a large group of diverse soil microorganisms (defined as broad processes), such as respiration or mineralization, than for processes performed by only a few specific microorganisms (defined as narrow processes). However, experimental decrease of microbial diversity did not alter either ‘broad’ or ‘narrow’ processes such as thymidine and leucine incorporation, carbon mineralization, denitrification and nitrification (Griffiths et al., 2001; Wertz et al., 2006). Similarly, minor changes in carbon mineralization despite important shifts in bacterial and fungal growth also suggest functional redundancy (Rousk et al., 2009). In contrast, comparison of litter decomposition rates in soil microcosms inoculated with different microbial communities showed that differences in community composition lead to functional dissimilarities (Strickland et al., 2009). Functional redundancy is also challenged by the findings that rare soil microbes are not redundant but may play a role in ecosystem functioning by enhancing plant defense against herbivores (Hol et al., 2011). These studies highlight our limited knowledge of the extent of functional redundancy in microbial communities, yet determining that its role in ecosystem functioning is essential for deciphering the value of microbial diversity.
Here, we address the question of the importance of functional redundancy in microbial communities by investigating whether the loss of species can be compensated for by others with the same functional role for maintaining ecosystem processes. For this purpose, we used a trait-based approach with the denitrifier community as model functional guild. Denitrifiers are taxonomically diverse microorganisms capable of reducing soluble nitrogen oxides into the gases N2O and N2 (Philippot et al., 2007). It is the main biological process responsible for the return of fixed nitrogen to the atmosphere, thus completing the N-cycle. Denitrifiers are also responsible for greenhouse gas emission and nitrogen losses (Conrad, 1996). Microbial diversity was manipulated through a removal experiment (Diaz et al., 2003) by inoculating sterile soil microcosms with serial dilutions of a soil microbial suspension. The impact of the removal experiment on the denitrifier genetic diversity was evaluated using high-throughput sequencing. If functional redundancy is supported, denitrification rates would be similar despite differences in diversity levels. We further tested whether the availability of resources influences the relation between microbial diversity and process rates by incubating the soil microcosms with and without plant residues.
Materials and methods
Microcosm and experimental design
The soil was collected from the top 10 cm of a Cambisol near Lusignan (South-West of France: 46°25′12.91″ N; 0°07′29.35″ E). The grassland soil had a texture of 15% sand, 67% silt and 18% clay, a pH of 6.6 and contained 13.2 mg C g−1 soil and 1.5 mg N g−1 soil. The soil was sieved to <2 mm and divided into 81 microcosms of 40 g before sterilization by γ-radiation (35 kGy; Conservatome, Dagneux, France). The soil microcosms were then inoculated with suspensions of the same soil that was not sterilized. An initial soil suspension was prepared by mixing 100 g soil (equivalent dry mass) with 300 ml sterile distilled water using a Waring blender. After blending for 5 min at maximum speed, the soil suspension was serial diluted. Three levels of dilution of the soil suspension were used as inocula to create a gradient of diversity, that is, undiluted (100; D1), 1/103 dilution (D2) and 1/105 dilution (D3) and triplicate microcosms were inoculated for each treatment. Approaches to experimentally manipulate microbial diversity have inherent bias and, for example, dilution approaches neglect the role of cell aggregates or biofilms that occur in soils, potentially biasing the proper diluting out of microbial species. However, in contrast to artificial community assembly approaches, members of the resulting communities following dilution originate from a naturally assembled community, and thereby provide a more realistic scenario for testing the effects of diversity loss. Since serial dilution results in differing inoculum biomass, microcosms were pre-incubated at 20 °C to allow microbial colonization. After 6 weeks pre-incubation, 5 mg g−1 of ground oven-dried wheat shoot residues (65 °C for 48 h) were added in half of the microcosms. At days 0 (T0; addition of the residues), 3 (T3), 7 (T7), 14 (T14) and 29 (T29), three microcosms from each treatment were used for activity and molecular analyses. PCR amplifications of 16S and 18S rRNA genes from wheat residues were negative, indicating that they were not a source of additional denitrifiers.
Denitrification activity measurements
Potential denitrification activity was measured using the acetylene inhibition technique. For each of the five dates, 1 g of soil from the microcosms (3 replicates × 3 dilution × 2 resource levels) was placed in 10-ml flasks and 0.6 ml of a solution containing KNO3 (240 mg l−1), glucose (180 mg l−1) and chloramphenicol (8.33 g l−1) was added. The flasks were then sealed and purged several times by evacuating the ambient air and filling with N2. Acetylene was then added (10% v/v of flask headspace) and the flasks were incubated at 20 °C. The gas atmosphere of these flasks was sampled after 30 min and 60 min of incubation and analyzed on a GC (Varian 3400 Cx) fitted with an ECD detector. Denitrification rates were calculated from the N2O production during incubation.
DNA extraction
For each replicate microcosm from each dilution treatment, DNA was extracted using a procedure developed by the GenoSol platform for application in large-scale soil surveys (Terrat et al., 2012). Briefly, in a 15-ml Falcon tube, 2 g of each soil sample was mixed with 4 ml of a solution containing 100 mM Tris (pH 8.0), 100 mM EDTA (pH 8.0), 100 mM NaCl, and 2% (wt/vol) sodium dodecyl sulfate. In all, 2 g of 100-μm diameter silica beads, 2.5 g of 1.4-mm diameter ceramic beads and 4 glass beads of 4-mm diameter were added to the mixture. The samples were then homogenized for 3 × 30 s at 4 m s−1 in a FastPrep-24 (MP-Biomedicals, Santa Ana, CA, USA) and incubated for 30 min at 70 °C before centrifugation at 7.000 × g for 5 min at 20 °C. For the deproteination, 1 ml of the collected supernatants was incubated for 10 min on ice with 1/10 volume of 3 M potassium acetate (pH 5.5) and centrifuged at 14.000 × g for 5 min. Finally, after precipitation with one volume of ice-cold isopropanol, the nucleic acids were washed with 70% ethanol. Total DNA concentration in each sample was fluorometrically quantified with the Quant-iT PicoGreen dsDNA Assay Kit (Invitrogen, Cergy-Pontoise, France) following the manufacturer’s instructions.
Quantification of the denitrifier community abundance
Evaluation of the denitrifier abundances by quantitative PCR was performed for all sampling times according to Henry et al. (2006) using the nosZ gene as a molecular marker as previously performed (Scala and Kerkhof, 1999; Horn et al., 2006; Kjellin et al., 2007). The nosZ gene encodes the nitrous oxide reductase catalyzing the reduction of the greenhouse gas N2O into N2, which is the last step of the denitrification process. Reactions were carried out in an ABI prism 7900 Sequence Detection System (Applied Biosystems, Grand Island, NY, USA). Quantification was based on the increasing fluorescence intensity of the SYBR Green dye during amplification. The quantitative PCR assay was carried out in a 15-μl reaction volume containing the SYBR green PCR Master Mix (Absolute QPCR SYBR Green Rox ABgene; Thermo Fisher Scientific, Courtaboeuf, France), 1 μM of each primer, 250 ng of T4 gene 32 (QBiogene; MP Biomedicals, Strasbourg, France) and 2 ng of DNA. Standard curves were obtained using serial dilutions of linearized plasmid containing the nosZ gene from Bradyrhizobium japonicum USDA110. PCR efficiency for the different assays was about 93%. Two to three no-template controls were run for each quantitative PCR assay and no-template controls gave null or negligible values. The presence of PCR inhibitors in DNA extracted from soil was estimated by (i) diluting the soil DNA extract and (ii) mixing a known amount of standard DNA with soil DNA extract prior to qPCR. No inhibition was detected in either case.
Barcoded pyrosequencing analyses of the denitrifier community
The denitrifier community composition was assessed just before the addition of wheat shoot residues, that is, 6 weeks after inoculation to enable the soil colonization by the inoculated microorganisms. DNA extracts from the triplicate microcosms inoculated with the different dilutions were amplified using a two-step PCR procedure as recommended by Berry et al. ( 2012) for barcoded pyrosequencing. Briefly, 20 cycles of amplification were performed with the nosZ1 (5′-WCSYTGTTCMTCGACAGCCAG-3′) and nosZ2 (5′-ACSSKSACCTTSTTGCCGTYGC-3′) primers previously described (Henry et al., 2006). In the second step, 1 μl of the PCR products of the first reaction were amplified in a 15-cycle PCR using the nosZ forward primers preceded by 10 basepair-long barcodes, the sequencing key and the sequencing primer (Supplementary Table S1). The PCR products from two PCR were agarose gel verified, pooled and purified using the QIAEX II kit (Qiagen, Courtaboeuf, France). Pyrosequencing on a Roche’s 454 FLX Genome Sequencer was performed by Genoscreen (Lille, France) using the Titanium Chemistry following the manufacturer’s instructions. Due to technical problem, one replicate from the d1 treatment was discarded and not used in subsequent analyses.
Processing of pyrosequencing data and phylogenetic analysis
Sequence data were initially screened and de-multiplexed using the QIIME sequence analysis platform (Caporaso et al., 2010), with default minimum sequence quality (25) and size range (200–1000) parameters. The split sequence libraries were then screened using the HMMFRAME algorithm (Zhang and Sun, 2011) to reduce the occurrence of frame-shift errors within the data set, using a hidden markov model (HMM) based on a reference alignment of full-length NosZ amino-acid sequences obtained from genome sequencing projects. The corrected sequences were then processed using the OTUpipe method within the QIIME, and operational taxonomic units (OTUs) were defined at 97% nucleotide similarity. Representative sequences for each OTU were then translated and compared with the NosZ HMM using HMMER (Eddy, 1998), whereupon sequences that did not produce significant matches (E<0.001) to the model were excluded from both the alignment and the final table of OTU abundance. The final set of amino-acid sequences were appended to the reference alignment using HMMER, and nucleotides were then mapped to the corresponding amino-acid positions. The final nucleotide alignment was then inspected manually using the ARB software (Ludwig et al., 2004). Maximum likelihood phylogeny was calculated using the RAxML algorithm (Stamatakis, 2006), with the GTR+Γ model for nucleotide substitution and empirical base frequencies (Rodriguez et al., 1990). Columns in the final alignment were weighted based on alignment ambiguity using the ZORRO algorithm (Wu et al., 2012), and 50 replicate tree searches were performed to find the optimal tree topology. Node confidence was determined using the rapid bootstrap option in RAxML, with 500 bootstrap replicates. Tree plotting with additional OTU abundance data was performed using the iTOL webserver (Letunic and Bork, 2007). The average abundance of each OTU was calculated among replicates for each dilution level, and values were plotted as a proportion of the maximum OTU abundance detected within each dilution.
Several α-diversity indices, as well as indices depicting the population structure, were calculated based either on the rarefied OTU table at a depth of 7500 sequences per sample (Species richness, Chao1, Shannon and Simpson indices and Pielou’s evenness), or both the rarefied OTU table and the phylogenetic tree generated from the previous analysis (Faith’s phylogenetic diversity (PD) and the Net-Relatedness Index (NRI)). Note that we chose to calculate the NRI rather than the NTI (Nearest Taxon Index) since NTI is generated from tip-level relationships and therefore less suited for a large data set with a terminally unresolved phylogeny (Cooper et al., 2008). All the calculations were performed either with the QIIME pipeline (Caporaso et al., 2010) or with the R software (R Foundation for Statistical Computing, Vienna, Austria) using the vegan (Oksanen et al., 2011) and picante (Kembel et al., 2010) packages. To compare between-sample variations in the composition of the total microbial community, unweighted and weighted UniFrac distances (Lozupone and Knight, 2005) were calculated with the QIIME pipeline (Caporaso et al., 2010). PCoA (Principal Coordinate Analysis) was performed on both distance matrices and coordinates were used to draw 3D graphical outputs.
Deposited 454 read accession numbers
Sequences were submitted to the SRA (Sequence Read Archive) at NCBI under the accession number SRA056092.
Statistical analyses
Repeated-measures ANOVA were performed on the abundance of the nosZ gene copy number per gram of dry soil and on the denitrification rates measured over time at each dilution rate (undiluted, 1/103 and 1/105) in the amended (A) and control (C) treatments. Factors accounting for variation of both response variables were the dilution, soil amendment and sampling time, the latter being the repeated factor. nosZ gene copy number and the denitrification rates were respectively log-transformed and square root-transformed in order to have an approximated Gaussian and homoskedastic residual distribution. Pairwise differences between dilutions for each treatment and at each time point were assessed using a t-test. Bonferroni corrections were used to take into account multiple comparisons.
Results
Quantification of the denitrifier abundance
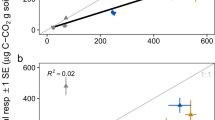
After a period of 6 weeks to allow for colonization of the sterilized soil by the inoculated microorganisms, quantification of the denitrifier community showed globally similar abundances independent of dilution level (Figure 1). Thus, the nosZ denitrification gene copy number per gram of dry soil, which was used as a proxy for the denitrifier abundance, was ∼2 × 107 in all microcosms (Figure 1). However, a significant difference between dilutions was observed at T7 (F2,12=37.079, P<0.001) where the abundance of the nosZ gene is slightly higher in the 1/105 dilution (P< 0.05) (Figure 1). After addition of the wheat residues in half of the microcosms, the denitrifier abundance were generally not affected, with the exception of T7 (F1,12=39.313, P<0.001) where the overall abundance of the nosZ gene is about 5% higher in the amended microcosms (P<0.05). The dilution treatment therefore had little to no significant effect on the abundance of denitrifiers in both the amended and the non-amended microcosms.
Quantification of the denitrifier community in microcosms inoculated with soil at different dilution levels without (a) and with (b) wheat residue amendment (mean±s.d., n=3). For each treatment, the same letters above the bars indicate microcosms without significant differences (P<0.05). Quantification was performed at days 0 (T0; addition of the residues), 3 (T3), 7 (T7), 14 (T14) and 29 (T29).
Denitrifier community diversity and composition
In total, 113 528 high-quality raw sequences ranging from 200 to 550 bp were obtained following de-multiplexing and removal of low-quality reads. After sequence processing with HMMFRAME and removal non-specific amplification products and chimeras from the data set, the remaining 102 265 sequences (out of which 90% were >350 bp) were grouped into 374 OTUs at a 97% nucleotide sequence identity threshold. A rarefaction analysis indicates that at a depth of 7500 sequences per sample, the nosZ PD plateaued even in the undiluted treatment (Figure 2). The decrease in richness and diversity between dilution treatments was supported by various diversity metrics (Table 1). As expected, the undiluted soil harbored the higher average species richness (235.85 [224.55–247.15]), followed by the 1/103 dilution (177.77 [165.19–190.34]) and the 1/105 dilution (57.33 [53.67–60.98]), corresponding to a 24% and 75% reduction in the captured diversity, within the diluted treatments. The Chao1 estimator, which reflects the average species richness given adequate sequencing coverage (Figure 1), the Shannon index and Faith’s PD estimator also showed the same trend. Interestingly, both Pielou’s evenness index and the Gini-Simpson index showed no significant differences between the undiluted treatment and the 1/103 dilution, while these indices were significantly lower at the 1/105 dilution. The NRI, which quantifies the overall clustering of taxa on a tree, was negative in the undiluted treatment, indicating a phylogenetic overdispersion of the taxa on the tree. By contrast, NRI was positive in the 1/103 and 1/105 dilution treatments, indicating a clustering of species that are phylogenetically close. The analysis of the unweighted UniFrac distances showed striking differences in community composition between dilution treatments (Figure 3a) and good between-replicate reproducibility with increasing variability in response to increasing levels of inoculum dilution (Figure 3b). The phylogenetic tree based on the alignment of the obtained sequences together with nosZ sequences from known strains showed sequences clustering with nosZ from the α, β and γ-Proteobacteria. However, most sequences were distantly related to nosZ from the cultured organisms (Figure 4). The Venn diagram revealed 95, 73 and 5 unique OTUs in the undiluted, 1/103 and 1/105 dilutions, respectively (Figure 4). Note that about 20% (78 out of 374) of the OTUs are shared by all three dilutions while about 50% of them (186 out of 374) are shared by the undiluted and the 1/103 dilution.
UniFrac analysis of the denitrifier community composition. (a) The three first axes from a PCoA of the unweighted UniFrac distance matrix are drawn. The percent of variance explained by each axis is given. The Undiluted, 1/103 and 1/105 dilutions bacterial communities are, respectively, represented by green, blue and red plain circles. (b) Between and within-dilution unweighted UniFrac distances. Within-dilution distances are represented with plain bars (green, blue and red, respectively, for undiluted, 1/103 and 1/105 dilutions) and between-dilution distances are represented with open bars.
Phylogenetic placement and distribution of nosZ OTUs in different dilution levels. The maximum likelihood phylogeny (GTR+Γ+F) consists of representative nucleotide sequences for OTUs detected in all dilutions, as well as reference nosZ sequences obtained from genome sequencing projects. Symbols at different tips in the tree denote reference sequences, with color and shape indicating major taxonomic affiliation. Node confidence (n=500 bootstrap replicates) between 50–100% is shown (●), where symbol size is scaled to reflect support levels. The phylogeny is rooted at midpoint, and clades consisting only of reference sequences are collapsed. Barplots indicate the average relative abundance of each OTUs, averaged across replicates and normalized to the maximum average value detected within the respective dilutions. The number of shared and unique OTUs in each dilution treatment is shown as a Venn diagram within the tree, where circle size is proportional to the number of OTUs detected.
Denitrification process rates
The dilution treatment affected potential denitrification consistently at every time point (F1,12=24.909, P<0.0001), with rates decreasing as dilution increased (Figure 5). Significant differences were observed between the undiluted and the 1/105 dilution at all dates except T3 in the non-amended microcosms, with an average decrease in the potential denitrification activity of about 82% in the 1/105 dilution treatment compared with the undiluted one. The 1/103 dilution also resulted in a decrease of the activity of about 60% compared with the undiluted treatment; however, differences were significant only at T29 (Figure 5). Addition of wheat residues significantly stimulated the potential denitrification activity in all dilution treatments at every time points (F1,12=181.853, P<0.0001) with twofold to fourfold increases compared with the non-amended microcosms. Similar to the non-amended microcosms, the same trend of decreasing potential denitrification rates with increasing dilution level was observed, except at T14 where pairwise differences were not significant. However, the absolute differences between dilutions were significantly greater in the amended microcosms (dilution × treatment effect, F2,12=5.890, P=0.016; Supplementary Figure S1). On average, the potential denitrification was about four times higher in the more diverse microcosms amended with residues than in the non-amended microcosms while stimulation by residues was by three and twofold in the 1/103 and 1/105 dilution treatments, respectively.
Potential denitrification in microcosms inoculated with soil at different dilution levels without (a) and with (b) wheat residue amendment (mean±s.d., n=3). For each treatment, the same letters above the bars indicate microcosms without significant differences (P<0.05). Measurements were performed 3, 7, 14 and 29 days after addition of the residues (T3, T7, T14 and T29). Note that the y-axis differ between (a, b).
Discussion
Since the dilution series of the soil suspension creates differences in inoculum biomass that could affect process rates, the microcosms were pre-incubated for 6 weeks to allow the establishment of the inoculated microorganisms. Quantification of the denitrifier abundance at the end of the pre-incubation period (T0) indicates that the inoculated communities had grown and reached a new equilibrium without significant differences among dilutions (Figure 1). The addition of wheat residues had very limited effect on the denitrifier abundance monitored during 1 month. Accordingly, Miller et al. (2008) showed that the number of the denitrifiers was not modified by the short-term addition of wheat, rape or alfafa residues and several studies suggest that carbon is not a strong driver of the denitrifier abundance in soil (Henry et al., 2008; Bru et al., 2011). Overall, serial dilution of the soil did not lead to a reduction in denitrifier abundances in the amended or non-amended microcosms after the pre-incubation period. On the contrary, an increase in community size was observed in the 1/105 dilution treatment at day 7, which is likely due to decreased interspecific competition as predicted by ecological theory (Franklin et al., 2001). Therefore, we can exclude that any decrease in denitrification rates with dilution would be due to a lower denitrifier biomass.
To assess how the denitrifier community has been affected by manipulation of its diversity by the removal experiment, denitrification gene amplicon pyrosequencing was performed at the end of the pre-incubation period (T0). Therefore, any change in the denitrifier diversity might not only be due to the effect of the dilution itself but also to species interactions during the re-colonization process. Previous studies have used fingerprinting methods for estimating the effect of removal experiments on microbial diversity (Franklin et al., 2001; Wertz et al., 2006; Peter et al., 2011), which has inherent limitations (Bent et al., 2007). To our knowledge, this is the first time that a high-throughput sequencing approach is performed to more reliably evaluate this effect using various diversity indices. All measured diversity parameters indicated that the inoculation with a serial dilution of the soil suspension resulted in microcosms with different levels of denitrifier diversity (Table 1). As expected, dilution caused an overall drop in diversity. However, despite the high dilution levels of 103 and 105 fold, only 25% and 75% of the total OTUs were removed in the D2 and D3 treatments, respectively. Similarly, Griffith et al. (2001) reported that the overall biodiversity (calculated from the morphotype of cultivable bacteria, number of DGGE bands, fungal morphotype and protozoa) decreased by about 50% in a sterile soil inoculated with a 106 dilution of a soil suspension compared with the least diluted soil (100). Since the rarefaction curves show a good coverage of the diversity, our results indicate that most OTUs in the denitrifier community are found in high abundance in re-colonized soil and that 24% of the OTUs, which were removed in the 103 dilution treatment, can be considered as ‘rare.’ This is further supported by the high Gini-Simpson index and Pielou’s evenness values for the undiluted and the 1/103 dilution, indicating that the relative abundance of the different species composing the community is relatively similar. At the 1/105 dilution, the additional loss of species may create a disequilibrium that resulted in only few species becoming dominant during establishment of communities in the microcosms, thus explaining the differences of both the Gini-Simpson and Pielou’s indices. In contrast, it was reported for the total bacterial community that taxa with abundance values higher than 105 cells per gram of soil accounted only for 0.1% of the diversity (Gans et al., 2005). Such differences can be explained by the high diversity level of 8 × 106 bacterial species in 10 g estimated by Gans et al. (2005) compared with 374 denitrifier OTUs present in our soil. This is in line with the results of Palmer et al. (2011) where many OTUs from the targeted denitrifiers were related to nosZ from Proteobacteria, commonly described as one of the dominant bacterium phylum in soil (Janssen, 2006; Nemergut et al., 2011). At the same time, organisms from other phyla are also capable of denitrification and might possess nosZ genes that are not targeted with our primers (Jones et al., 2013), while some denitrifiers completely lack the nosZ gene. Therefore, we cannot rule out the possibility that representation of some denitrifying groups, which were not captured in this study, has been underestimated. However, this would only influence the evaluation of the extent of the reduction in diversity along the dilution gradient but not the diversity loss itself. Thus, it is expected that the very act of diluting the entire microbial community led to a significant loss in the diversity of all microbial groups, and therefore the patterns observed likely apply to the denitrifier community as a whole.
Dilution simulation predicts that the greatest differences in community structure among replicated samples will occur at the highest dilution levels (Franklin et al., 2001). Accordingly, we found that the denitrifier community composition between replicates was more similar for the low than for the high dilution treatments (Figure 3). While this prediction did not hold true in the original work of Franklin et al. (2001), other studies using a similar dilution approach also reported greater differences between fingerprints obtained from the samples inoculated with the highest dilution (Griffiths et al., 2001; Wertz et al., 2006). This higher variability could be attributed to the stochastic removal of some species, lower interspecific competition or to priority effects with the establishment of some OTU influencing subsequent colonization by others, all leading to increased stochastic reassembly in the most diluted treatment during soil re-colonization (Lekberg et al., 2012).
When examining potential denitrification, we found a negative response to species removal with a decrease in activity in the diluted treatments (Figure 5). Despite a 105 dilution of the microbial community between D1 and D3, the decrease in potential denitrification was only about four- to fivefold. However, if we consider the measured diversity rather than the dilution level, both decreases in richness and process rates are in the same order of magnitude. Thus, removal of 75% of the total OTUs in the 1/105 dilution treatment led to a decrease in potential activity of about 48 to 88%. Our findings indicating a negative impact of diversity loss on soil functioning are in accordance with a previous experiment showing increased respiration with species richness by manipulation of the bacterial diversity by assembling communities containing up to 72 species (Bell et al., 2005). The observed decrease in potential denitrification might have also been due to changes in the nitrate concentration, resulting from reduction of the diversity of other microbial communities involved in nitrogen cycling by dilution. In all cases, our results suggest that a decrease in microbial diversity can affect nitrogen cycling in soil. At almost all sampling dates, the decrease in potential denitrification observed in the 1/103 dilution treatments was not significant (Figure 5). Accordingly, in this dilution treatment neither the Pielou’s evenness index nor the Gini-Simpson index were significantly different from the undiluted treatment and only 25% of the OTUs have been removed. Griffiths et al. (2001) observed that the decreases in potential nitrification rates were also significant only in the most diluted treatment (106). On the other hand, Wertz et al. (2006) reported that neither carbon mineralization nor denitrification were affected by experimental manipulation of the microbial diversity using a removal approach. These divergent observations could be due to differences in the original microbial communities across soils since communities differing in their structure and species composition may show different patterns of ecosystem response to diversity loss with some being more affected than others. Indeed, previous works emphasized the interrelated roles of species identity, community composition and richness in determining ecosystem processes (Tilman, 1997; Peter et al., 2011). This is summarized by the idiosyncratic response hypothesis, that is, both species identity and biodiversity simultaneously affect ecosystem functions but species-specific effects depend on environmental conditions and are therefore difficult to predict (Tilman et al., 1997; Emmerson et al., 2001). More recently, Wittebolle et al. (2009) showed that evenness, which is seldom taken into account in artificial community assembly studies, is also important for functionality. Accordingly, our results also showed that the decrease of activity between the undiluted and the 1/105 dilution treatments is correlated with a decrease in evenness. Since the abundance of the inoculated communities reached the soil carrying capacity following the 6-week pre-incubation, as demonstrated by the fact that no modification of the denitrifier abundance was observed between T0 and T29, it is very likely that the communities had reached an equilibrium in diversity by time T0.
In order to test whether the resource level could also be of importance in determining the shape of the microbial diversity–functioning relationship, the soil microcosms with different levels of denitrifier diversity were incubated for 1 month with and without wheat residues. Stimulation of potential denitrification in all treatments indicated that the added substrates were available despite the fact that no significant increase in denitrifier biomass was observed (Figures 1 and 5). We also found that the addition of substrates significantly increased absolute differences in potential denitrification among dilutions (P=0.016; Supplementary Figure S1). The greater positive response of denitrification activity to the addition of plant residues in the microcosms with the more diverse communities could be due to a higher functional complementarity leading to a better use of the overall resources. Thus, assemblages of microbial communities showed that the effect of diversity on functioning results from the interplay between the genetic structure of the community and resource complexity, with a direct interaction between genotypic richness and resource complexity (Jousset et al., 2011). In contrast, Langenheder et al. (2010) found that both species diversity and substrate richness enhanced functioning, but no direct interaction was observed. Another explanation for the positive effect of residue addition is that enhanced individual interactions with higher diversity increased the access to the added resources through facilitation. Thus, we verified our hypothesis that addition of resources modified the effect of species loss on ecosystem functioning, which could also explain the apparent discrepancies observed between studies investigating the role of microbial diversity using different soil types (Griffiths et al., 2001; Wertz et al., 2006).
In conclusion, analysis of the impact of the removal experiment on the denitrifier diversity using high-throughput sequencing revealed that despite dilutions of up to five orders of magnitude, the diversity of this functional community did not drop drastically with a decrease of about 75% of the measured OTU richness. However, this decrease in the denitrifier diversity or in any other microbial guild was sufficient to result in a significant decrease of 4–5 folds in the denitrification activity, indicating a limited functional redundancy of the soil microbial community. We also found that the soil resource level had a significant impact on the shape of the denitrifier diversity–activity relationship. Altogether, our results showed that biogeochemical cycling in soil can be affected by diversity loss, which highlights the functional consequences of this major threat to which soils are confronted (Commission of the European Community, 2006). More studies are now required to evaluate the extent of the impact of microbial diversity loss on other soil functions and services in relation to community composition and species identity.
Accession codes
References
Bell T, Newman J, Silverman B, Turner S, Lilley A . (2005). The contribution of species richness and composition to bacterial services. Nature 436: 1157–1160.
Bent J, Pierson J, Forney L . (2007). Measuring species richness based on microbial community fingerprints: the emporor has no clothes. Appl Environ Microbiol 73: 2399–2401.
Berry D, Ben Mahfoudh K, Wagner M, Loy A . (2012). Barcoded primers used in multiplex amplicon pyrosequencing bias amplification. Appl Environ Microbiol 77: 7846–7849.
Bru D, Ramette A, Saby NPA, Dequiedt S, Ranjard L, Jolivet C et al. (2011). Determinants of the distribution of nitrogen-cycling microbial communities at the landscape scale. ISME J 5: 532–542.
Caporaso J, Kuczynski J, Stombaugh J, Bittinger K, Bushman F, Costello E . (2010). QIIME allows analysis of high-throughput community sequencing data. Nature Meth 7: 335–336.
Chapin FS, Zavaleta ES, Eviner VT, Naylor RL, Vitousek PM, Reynolds HL et al. (2000). Consequences of changing biodiversity. Nature 405: 234–242.
Commission of the European Community (2006), Proposal for a directive of the European parlament and of the council establishing a framework for the protection of soil and amending Directive 2004/35/EC.
Conrad R . (1996). Soil microorganisms as controllers of atmospheric trace gases (H2, CO, CH4, OCS, N2O, and NO). Microbiol Rev 60: 609–640.
Cooper N, Rodriguez J, Purvis A . (2008). A common tendency for phylogenetic overdispersion in mammalian assemblages. Proc R Soc B 275: 2031–2037.
Diaz S, Symstad AJ, Chapin FS, Wardle DA, Huenneke LF . (2003). Functional diversity revealed by removal experiments. Trends Ecol Evol 18: 140–146.
Eddy S . (1998). Profile hidden Markov models. Bioinformatics 14: 755–763.
Emmerson MC, Solan M, Emes C, Paterson DM, Raffaelli D . (2001). Consistent patterns and the idiosyncratic effects of biodiversity in marine ecosystems. Nature 411: 73–77.
Franklin R, Garland J, Bolster C, Mills A . (2001). Impact od dilution on microbial community structure and functional potential: comparison of numerical simulation and batch culture experiments. Appl Environ Microbiol 67: 702–712.
Gans J, Wolinsky M, Dunbar J . (2005). Computational improvements reveal great microbial diversity and high metal toxicity in soil. Science 309: 1387–1390.
Griffiths B, Ritz K, Wheatley R . (2001). An examination of the biodiversity-ecosystem function relationship in arable soil microbial communities. Soil Biol Biochem 33: 1713–1722.
Henry S, Bru D, Stres B, Hallet S, Philippot L . (2006). Quantitative detection of the nosZ gene, encoding nitrous oxide reductase, and comparison of the abundance of 16S rRNA, narG, nirK, and nosZ genes in soils. Appl Environ Microbiol 72: 5181–5189.
Henry S, Texier S, Hallet S, Bru D, Dambreville C, Chèneby D et al. (2008). Disentangling the rhizosphere effect on nitrate reducers and denitrifiers: insight into the role of root exudates. Environ Microbiol 10: 3082–3092.
Hol WHG, de Boer W, Termorshuizen AJ, Meyer KM, Schneider JH, van Dam NM et al. (2011). Reduction of rare soil microbes modifies plant-herbivore interactions. Ecol Lett 13: 292–301.
Hooper DU, Chapin FS, Ewel JJ . (2005). Effects of biodiversity on ecosystem functioning: a consensus of current knowledge. Ecol Monogr 75: 3–35.
Horn MA, Drake HL, Schramm A . (2006). Nitrous oxide reductase genes (nosZ) of denitrifying microbial population in soil and the earthworm gut are phylogenetically similar. Appl Environ Microbiol 72: 1019–1026.
Janssen P . (2006). Identifying the dominant soil bacterial taxa in libraries of 16S rRNA and 16S rRNA genes. Appl Environ Microbiol 72: 1719–1728.
Jones CM, Graf D, Bru D, Philippot L, Hallin S . (2013). The unaccounted yet abundant nitrous oxide reducing microbial community—a potential nitrous oxide sink. ISME J 7: 417–426.
Jousset A, Schmid B, Scheu S, Eisenhauer N . (2011). Genotypic richness and dissimilarity opposingly affect ecosystem functioning. Ecol Lett 14: 537–545.
Kembel S, Cowan P, Helmus MR, Cornwell WK, Morlon H, Ackerly DD et al. (2010). Picante: R tools for integrating phylogenies and ecology. Bioinformatics 26: 1463–1464.
Kjellin J, Hallin S, Worman A . (2007). Spatial variations in denitrification activity in wetland sediments explained by hydrology and denitrifying community structure. Water Res 41: 4710–4720.
Langenheder S, Bulling MT, Solan M, Prosser JI . (2010). Bacterial biodiversity-ecosystem functioning relations are modified by environmental complexity. PLoS ONE 5: e10834.
Lekberg Y, Schnoor T, Kjøller R . (2012). 454-sequencing reveals stochastic local reassembly and high disturbance tolerance within arbuscular mycorrhizal fungal communities. J Ecol 100: 151–160.
Letunic I, Bork P . (2007). Interactive Tree of Life (iTOL): an online tool for phylogenetic tree display and anootation. Bioinformatics 23: 127–128.
Loreau M . (2004). Does functional redundancy exists? Oikos 103: 606–611.
Loreau M, Naeem S, Inchausti P, Bengtsson J, Grime JP, Hector A et al. (2001). Ecology—biodiversity and ecosystem functioning: current knowledge and future challenges. Science 294: 804–808.
Lozupone C, Knight R . (2005). UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 71: 8228–8235.
Ludwig W, Strunk O, Westram R, Richter L, Meier H, Buchner A et al. (2004). ARB: a software environment for sequence data. Nucleic Acids Res 32: 1363–1371.
Miller MN, Zebarth BJ, Dandie CE, Burton DL, Goyer C, Trevors JT . (2008). Crop residue influence on denitrification, N2O emissions and denitrifier community abundance in soil. Soil Biol Biochem 40: 2553–2562.
Nannipieri P, Ascher J, Ceccherini MT, Landi L, Pietramellara G, Renella G . (2003). Microbial diversity and soil functions. Eur J Soil Sci 54: 655–670.
Nemergut D, Costello E, Hamady M, Lozupone C, Jiang L, Schmidt SK et al. (2011). Global patterns in the biogeography of bacterial taxa. Environ Microbiol 13: 135–144.
Oksanen J, Blanchet FG, Kindt R . (2011). Vegan: Community Ecology Package. R package version 2.0-0.http://CRAN.R-project.org/package=vegan.
Palmer K, Biasi C, Horn M . (2011). Contrasting denitrifier communities relate to contrasting N2O emission patterns from acidic peat soils in arctic tundra. ISME J 6: 1058–1077.
Peter H, Beier S, Bertilsson S, Lindström E, Langenheder S, Tranvik L . (2011). Function-specic response to depletion of microbial diversity. ISME J 5: 351–361.
Philippot L, Hallin S, Schloter M . (2007). Ecology of denitrifying prokaryotes in agricultural soil. Adv Agronomy 96: 135–190.
Rodriguez F, Oliver J, Marin A, Medina J . (1990). The general stochastic-model of nucleotide substitution. J Theo Biol 142: 485–501.
Rousk J, Brookes P, Baath E . (2009). Contrasting soil pH effects on fungal and bacterial growth suggest functional redundancy in carbon mineralization. Appl Environ Microbiol 75: 1589–1596.
Scala D, Kerkhof L . (1999). Diversity of nitrous oxide reductase (nosZ) genes in continental shelf sediments. Appl Environ Microbiol 65: 1681–1687.
Schimel J . (1995). Ecosystem consequences of microbial diversity and community structure Chapin F, Koerner C, (eds.) Arctic and Alpine Biodiversity: Patterns, Causes, and Ecosystem Consequences. Springer Verlag: New York, NY, USA, pp 239–254.
Stamatakis A . (2006). RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed model. Bioinformatics 22: 485–501.
Strickland M, Lauber C, Fierer N, Bradford M . (2009). Testing the functional significance of microbial community composition. Ecology 90: 441–451.
Terrat S, Christen R, Dequiedt S, Lelièvre M, Nowak V, Regnier T et al. (2012). Molecular biomass and MetaTaxogenomic assessment of soil microbial communities as influenced by soil DNA extraction procedure. Microb Biotechnol 5: 135–141.
Tilman D . (1997). Distinguishing between the effects of species diversity and species composition. Oikos 80: 185–185.
Tilman D, Lehman C, Thomson K . (1997). Plant-diversity and ecosystem productivity: theoretical considerations. Proc Natl Acad Sci USA 94: 1857–1861.
Torsvik V, Ovreas L, Thingstad TF . (2002). Prokaryotic diversity—magnitude, dynamics, and controlling factors. Science 296: 1064–1066.
Wardle DA, Bardgett RD, Callaway RM, Van der Putten WH . (2011). Terrestrial ecosystem responses to species gains and losses. Science 332: 1273–1277.
Wertz S, Degrange V, Prosser JI, Poly F, Commeaux C, Freitag T et al. (2006). Maintenance of soil functioning following erosion of microbial diversity. Environ Microbiol 8: 2162–2169.
Wittebolle L, Marzorati M, Clement L, Balloi A, Daffonchio D, Heylen K et al. (2009). Initial community evenness favours functionality under selective stress. Nature 458: 623–626.
Wu M, Chatterji S, Eisen J . (2012). Accounting for alignment uncertainty in phylogenomics. PLoS ONE 7: e30288.
Zhang Y, Sun Y . (2011). HMM-FRAME: accurate protein domain classification for metagenomic sequences containing frameshift errors. BMC Bioinformatics 12: 198.
Acknowledgements
We gratefully acknowledge Abad Chabbi and Aurore Kaisermann for assistance and the anonymous reviewers for their helpful and constructive comments. This work was supported by the Agence Nationale de Recherche (ANR) under the framework of the ANR Systerra project DIMIMOS, and by the European Commission within EcoFINDERS project (FP7-264465).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies this paper on The ISME Journal website
Supplementary information
Rights and permissions
About this article
Cite this article
Philippot, L., Spor, A., Hénault, C. et al. Loss in microbial diversity affects nitrogen cycling in soil. ISME J 7, 1609–1619 (2013). https://doi.org/10.1038/ismej.2013.34
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ismej.2013.34
Keywords
This article is cited by
-
Erosion and deposition significantly affect the microbial diversity, co-occurrence network, and multifunctionality in agricultural soils of Northeast China
Journal of Soils and Sediments (2024)
-
Metagenomic analysis reveals diverse microbial community and potential functional roles in Baner rivulet, India
Journal of Genetic Engineering and Biotechnology (2023)
-
Leveraging microbiome rediversification for the ecological rescue of soil function
Environmental Microbiome (2023)
-
Functional gene-guided enrichment plus in situ microsphere cultivation enables isolation of new crucial ureolytic bacteria from the rumen of cattle
Microbiome (2023)
-
The rhizosphere Microbiome of Malus sieversii (Ldb.) Roem. in the geographic and environmental gradients of China's Xinjiang
BMC Microbiology (2023)