Abstract
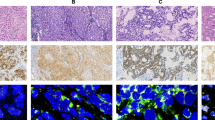
Detection of anaplastic lymphoma kinase (ALK), ROS proto-oncogene 1 (ROS1), and rearranged during transfection (RET) gene rearrangements in lung adenocarcinoma is usually performed by immunohistochemistry (IHC) screening followed by fluorescence in situ hybridization (FISH), which is an expensive and difficult technique. Ligation-dependent reverse transcription polymerase chain reaction (RT-PCR) multiplex technique can detect gene rearrangements using probes specifically hybridized to either side of the break point. PCR products are then sequenced by pyrosequencing or high throughput sequencing in order to identify the two genes involved. The reagent cost is <15 dollars per patient and results are available in 2 days. We have developed a 47-probe LD-RT-PCR kit especially for lung adenocarcinomas. Thirty-nine lung adenocarcinomas were studied: 24 ALK+, 14 ROS1+, and 1 RET+. ALK+ and ROS1+ were IHC+ (D5F3 Ventana for ALK and D4D6 Cell Signaling Technology for ROS1) and all cases were FISH+ (Vysis ALK Breakapart Probe Abbott for ALK, Zytolight SPEC ROS1 Dualcolor Breakapart Probe for ROS1 and Zytolight SPEC RET Dual Color Breakapart for RET); 14 wild type samples were included as negative controls. Using LD-RT-PCR, 15 rearrangements (63%) were detected in the ALK cases (gene partner: EML4 in all cases), 9 rearrangements (64%) in the ROS1 cases (gene partners: CD74 in 8 cases and SLC34A2 in 1 case) and 1 (100%) in the single RET case (gene partner: KIF5B). No rearrangement was found in the 14 negative control cases. Negative cases using LD-RT-PCR could be explained by the fact that some partner genes were not included in our assay and therefore could not be detected. Because it is an affordable, fast, and very simple technique, we propose using LD-RT-PCR when ALK immunostaining is positive. For LD-RT-PCR-negative cases, samples should then be analyzed by FISH.
Similar content being viewed by others
Main
Lung cancer is a major cause of cancer worldwide in terms of incidence and mortality.1, 2 Adenocarcinoma represents an increasing histological type of lung cancer and since 2005 is more frequent than squamous cell carcinoma.2, 3
In recent years, driver mutations have been described in lung adenocarcinoma, involving mainly EGFR, KRAS, BRAF, ERBB2, MET, anaplastic lymphoma kinase (ALK), ROS proto-oncogene 1 (ROS1), and rearranged during transfection (RET) genes.4, 5, 6, 7 For the three latter, the molecular mechanism consists of a gene rearrangement leading to deregulated over expression of the fusion-gene and therefore the fusion-protein. Although ALK alterations are detected in 5% of all lung adenocarcinomas, they represent 14% in never smokers.8 ROS and RET translocations are much rarer, involving, respectively, ~1 and 2% of lung adenocarcinomas.9 It is crucial to detect such abnormalities because they are good predictive biomarkers of response to targeted therapies such as crizotinib for ALK10, 11 and ROS translocations.12 Some targeted therapies seem to have a good anti-tumor effect in case of a RET translocation.13
Detection of these molecular alterations can be challenging, especially when biopsies are very small, which is often the case with lung adenocarcinomas because they tend to be peripheral. In many centers, ALK immunohistochemistry (IHC) is performed first so as to reduce the flow of fluorescence in situ hybridization (FISH).14 If negative, it is admitted that there is no ALK rearrangement and FISH is performed only if the patient is a non-smoker or if there is signet ring morphology. In case of positive IHC, an ALK translocation is confirmed by FISH. For ROS1, a similar two-step procedure can be performed, whereas IHC is not available for RET detection and only FISH is performed when the patient is a never smoker and no mutation or gene rearrangement is retrieved by conventional molecular analysis. FISH is quite expensive (roughly 75 € per slide), time-consuming and can be very difficult to interpret because it is not possible to ensure that the observed cell is neoplastic. It is very sensitive to pre-analytic conditions (fixation, sample storage) and cannot identify the partner gene when using breakapart probes. In addition, specificity is not perfect. However, it allows detection of virtually all translocations (except in very rare cases) regardless of the partner gene. Conversely, fusion probes can detect a partner gene but are impractical routinely because of the multiplicity of potential partner genes. Molecular techniques are used in some centers as an alternative to FISH: conventional real-time retrotranscription polymerase chain reaction (RT-PCR) or high throughput sequencing. Although RT-PCR is unable to detect all different types of fusion transcripts, high-throughput sequencing is of limited routine use for detection of fusion transcripts, mainly because of the complexity of the bioinformatic analysis for alignment of sequences.
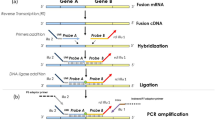
The ligation-dependent reverse transcription polymerase chain reaction (LD-RT-PCR) is a rapid and parsimonious technique inspired by RT-PCR technique and has been developed by the INSERM U918 research group of Henri Becquerel Cancer Center (Rouen, France). This assay is successfully used on a daily basis to classify diffuse large B-cell lymphomas15 and acute leukemia.16 The principle is simple (Figure 1):15, 16 total RNA from tumors are extracted from samples and then converted into complementary DNA (cDNA) using reverse transcription. Next, cDNA are incubated with oligonucleotides probes which are the ends of the exons fused on hybrid messenger RNAs (mRNAs). When a fusion transcript is present, two probes hybridize side-to-side at the aberrant cDNA junction. A DNA ligase is next added to the reaction mixture to create a covalent link between these probes, which allows their amplification by PCR using primers that correspond to their additional tails When no fusion transcript is present, amplification does not happen. The two partners and the break point are next identified using pyrosequencing analysis or high-throughput sequencing. The main advantage of this technique compared to conventional RT-PCR is that it requires only a short RNA sequence (~50 base pairs) on both sides of the break point. Therefore, it is less dependent on RNA alteration and can easily be performed on formalin-fixed paraffin-embedded (FFPE) tissues. In addition, it allows multiplex analysis and >150 probes can be added to the same mixture. Cost is negligible (less than 15 € per patient, all inclusive) and results are rapidly obtained in 2 days on average (regardless of RNA extraction procedure).
Principle of LD-RT-PCR. Total RNA from tumors are extracted from samples and converted into complementary DNA (cDNA) using reverse transcription. Next, cDNA are incubated with oligonucleotide probes which are the ends of the exons fused on hybrid messenger RNAs (mRNAs). When a fusion transcript is present, two probes hybridize side-to-side at the aberrant cDNA junction. A DNA ligase is next added to the reaction mixture to create a covalent link between these probes, which allows their amplification by PCR using primers that correspond to their additional tails. When no fusion transcript is present, amplification does not happen. The two partners and the break point are next identified using pyrosequencing analysis or high throughput sequencing.
The aim of our study was to assess this innovative technique for detection of gene fusion transcripts in lung adenocarcinoma from FFPE samples (biopsies of specimen resections) and to estimate its sensitivity and specificity.
Materials and methods
Ethics
Ethical approval was obtained according to the agreement of the tumor biobank of Rouen University Hospital (tissue sample collection no. DC2008-689) by the institutional review board of Rouen University Hospital and the French Ministry of Scientific Research.
Patients and Tumors
Prospective cases of lung adenocarcinomas harboring ALK, ROS, or RET translocation analyzed between 01 January 2014 and 31 December 2015 in Rouen University Hospital (France) were eligible for inclusion. For ALK and ROS, both IHC and FISH were performed to detect gene rearrangement. For detection of RET translocation, only FISH was used.
When possible, data were retrieved from the patient’s medical records on smoking status, as well as the prescription of a targeted therapy and its efficacy.
As negative controls, we used 14 resection specimens of lung adenocarcinoma analyzed at Rouen University Hospital between the same period of time, without any mutation or rearrangement of EGFR, KRAS, ALK, ROS, or RET retrieved. FFPE cell clots obtained from NCI-H2228 cell line (ATCC) were used as a positive control for ALK translocation.
IHC
Immunoassayed slides were retrieved and re-scored by CG and JSC. For detection of ALK protein, we used the Ventana ALK (D5F3) CDx Assay, whereas ROS1 protein was detected using the ROS1 D4D6 Rabbit monoclonal antibody #3287 (Cell Signaling Technology).
For ALK and ROS detection, a four-category system was used to assess staining, regardless of the percentage of stained tumor cells: 0=no staining, 1+: weak staining, 2+: moderate staining, 3+: strong staining. From an immunohistochemical point of view, a tumor was considered ALK positive from 2+ staining or ROS1 positive from 1+ staining, and therefore was considered for FISH analysis.
As stated above, we did not perform immunostaining for RET protein detection.
FISH
We performed FISH using breakapart probes so as to detect gene rearrangements: using Vysis ALKBreak Apart FISH Probe Kit for ALK and ZytoLight SPEC ROS1 Dual Color Break Apart Probe kit (CliniSciences) for ROS1. FISH for RET was performed using the ZytoLight SPEC RET Dual Color Break Apart Probe kit (CliniSciences). At least 100 tumor nuclei were analyzed and a case was considered as positive when the number of neoplastic nuclei with split signal or with single 3′ signal above at minimum 20% of observed neoplastic nuclei.
Exclusion Criteria
Exclusion criteria are listed below:
-
Less than 15% of tumor cells.
-
Weak IHC staining (1+) for ALK only, according to the manufacturer’s instructions.
-
Weak GAPDH expression.
LD-RT-PCR Assay
We developed a LD-RT-PCR assay dedicated to the detection of ALK, ROS, and RET gene fusion transcripts in lung adenocarcinoma. First, a comprehensive review of the literature was performed from PubMed, allowing the listing of all described fusion transcripts until April 2016. Finally, 43 probes were included in the kit (Figure 2). In addition, two probes complementary to the housekeeping gene GAPDH were added to the kit in order to assess the quality of RNA in samples. The technique was performed according to the procedure described above in the introduction. We also assessed the RNA concentration of the samples using the Qubit RNA HS Assay kit (ThermoFisher Scientific).
In brief, serial sections of FFPE tumors were cut from a paraffin block representative of lesion and placed on glass slides: a 4-mm-thick section was stained with hematoxylin & eosin (H&E) for microscopic examination. The following section was processed for tumor RNA preparation. The microtome razor blade was changed between each FFPE tumor sample, and the paraffin sections were processed individually to avoid cross contamination. H&E preparation enabled tumor area delimitation and visual estimation of tumor cell percentage simultaneously by two pathologists (CG and JCS) using a dual-headed microscope (Leica DM 2000). The tumor area was macro-dissected on a 10-mm-thick section placed on a glass slide using a single-use sterilized scalpel. RNA was extracted from this material with the Maxwell 16 LEV RNA FFPE kit (Promega). The reverse transcription was performed with the kit Superscript VILO cDNA Synthesis kit (ThermoFischer Scientific). Next, the probes were incubated with cDNA at 95 °C for 2 min (denaturation) and at 60 °C for 1 h (hybridization). Ligation was achieved using the Salsa MLPA kit (MRC-Holland). For PCR amplification, 5 μl of the ligation products were transferred to new tubes containing 45 μl of a PCR mix (20 μl Red’y’Star Mix (Eurogentec, Liege, Belgium); 1 μl 10 μM 5′ biotinylated primer U1 (GGGTTCCCTAAGGGTTGGA), 1 μl 10 μM primer U2 (GTGCCAGCAAGATCCAATCTAGA) and 18 μl water). Alternatively, a biotinylated primer U2 was used together with an unmodified primer U1 to allow the characterization of the junction from the other end. The amplification was performed as follow: 6 min at 94 °C; 35 cycles (30 s at 94 °C, 30 s at 58 °C, 30 s at 72 °C); 4 min at 72 °C; and cooled at 16 °C. PCR products (10 μl) were analyzed on 8% acrylamide/bis-acrylamide (40/1) gels. If a PCR product was detected after migration on polyacrylamide gel, it was sequenced to identify the two partner genes using pyrosequencing, high throughput sequencing or both methods. For pyrosequencing, 20 μl were analyzed on a PyroMark Q24 platform (Qiagen, Venlo, the Netherlands) with a 15(ACTG) dispensation order following standard procedures. For high throughput sequencing, PCR products were obtained using the U1 probes with a P1 tail (P1–U1) and the U2 probes with a A tail (A-U2). Products were then purified using the Agencourt AMPure XP kit (Beckman Coulter) and analyzed with an Ion PGM System (Thermo Scientific). Sequencing results were blindly interpreted by PR and VM.
Results
Patients and Tumors
ALK
Thirty-seven tumors from 36 patients were initially selected as ALK-positive cases between 01 January 2014 and 31 December 2015 in our center. Among these 37 samples, 7 were initially excluded because the neoplastic tissue was completely exhausted. Then, a further six cases were excluded because GAPDH mRNA was weak. Finally, 24 cases were analyzed. Among these (Table 1), mean age at resection or biopsy was 58 years (standard deviation=16). The majority of samples were obtained by biopsy (16/24), whereas only eight by specimen resection. Eleven cases (46%) were from metastasis and 13 (54%) from the initial lung lesion. Twenty-three lesions were diagnosed as adenocarcinoma and only one was considered as 'non-small cell lung carcinoma not otherwise specified' according to the 2015 World Health Organization classification. It is important to note that no tissue had undergone decalcification, and that there were no tumor necrosis areas. Information regarding treatment was available in only five patients: four were treated by anti-ALK targeted therapy with reported efficacy in three patients.
The percentage of tumor cells was above 50% in 11 cases (46%). ALK immunostaining was strong (3+) in 16 cases (67%) (Table 2). Another 'driver mutation' was retrieved in two cases, involving KRAS.
ROS1
Eighteen tumors from 17 patients were included in our study as ROS1-positive cases, but 4 were excluded because the neoplastic tissue was exhausted. Among the 14 cases from 13 patients finally analyzed (Table 3), mean age at resection or biopsy was 54 years (standard deviation=13). The majority of samples were obtained by specimen resection (50%) with only 23% of biopsies. One case involved a cytological sample. The large majority involved metastasis (72%, ie, 10 cases) and no tissue underwent decalcification. None of the samples contained tumor necrosis. Histological types were more diverse than for the ALK cases, with 10 adenocarcinomas, 2 'non-small cell lung carcinoma not otherwise specified', but also 1 adenosquamous carcinoma and 1 sarcomatoid carcinoma. We retrieved data on only three patients for whom therapy was available, and only one patient was successfully treated by several lines of ROS1 inhibitors (crizotinib, ceritinib, cabozantinib) and conventional chemotherapy, displaying a dissociated anti-tumor response.
ROS immunostaining was strong (3+) in 7 cases (50%) and weak (1+) in 6 cases, ie, 43% (Table 4). In 10 cases, samples contained >50% of tumor cells. RNA was always considered as well-conserved as GAPDH expression was high in all cases. Contrary to the ALK cohort, we did not detect another 'driver mutation'.
RET
Only one tumor, neither decalcified nor necrotic, harboring a rearrangement of RET was included in our study (Table 5). It was obtained from a bronchial biopsy performed in a 47-year-old woman diagnosed with a lung adenocarcinoma. To our knowledge, this patient was treated by conventional chemotherapy, radiotherapy, and immunotherapy.
Tumor genotyping did not retrieve another 'driver mutation'. The sample contained between 25 and 50% of tumor cells and 80% of tumor nuclei harbored a translocation of RET at FISH examination. GAPDH expression was high and the RNA concentration of the sample was 72 ng/μl.
LD-RT-PCR
All the 14 negative controls were negative using LD-RT-PCR and in all cases, a high expression of GAPDH was detected. Conversely, the cell clot used as a positive control for ALK rearrangement was positive using LD-RT-PCR, with the ALKe20-EML4e6 transcript retrieved. It is noteworthy to mention that the detected rearrangements were mutually exclusive, that is to say that ALK-positive cases were negative for ROS1 and RET, ROS1-positive cases negative for ALK and RET, and the only RET-positive case negative for ALK and ROS1. Similarly, when a gene translocation was observed, it was unique, for example we never observed more than one translocation in positive cases.
ALK
Fifteen cases were positive using LD-RT-PCR (Table 2), meaning that the sensitivity of our technique was calculated at 63% (CI 95% (43–79%)). Conversely, specificity was 100% because all negative cases were negative using LD-RT-PCR. Eleven cases were detected both by pyrosequencing and high-throughput sequencing and for four cases, a fusion transcript was detected by pyrosequencing but this method was unable to give the precise name of the rearrangement, which was easily identified by high throughput sequencing. ALK translocation involved the EML4 gene in all cases and the most frequently retrieved fusion was ALKe20-EML4e13 (seven cases). Only ALK exon 20 was involved. Interestingly, a polysomy of chromosome 2 was observed in one case (patient 22). Four cases were detected by the technique, whereas the RNA concentration was zero according to the Qubit RNA HS Assay kit.
ROS1
LD-RT-PCR allowed detection of a translocation of ROS1 in 64% (nine cases). Hence, the sensitivity of our technique for the detection of ROS1 translocation was 64% (CI 95% (39–84%)), and the specificity was 100%. In eight samples, transcripts involved exon 34 of ROS and exon 6 of the partner gene CD74 (ROSe34-CD74e6 translocation). Similarly to the ALK samples, four cases were detected by the technique, whereas the RNA concentration was zero according to the Qubit RNA HS Assay kit.
RET
LD-RT-PCR allowed us to detect the translocation RETe12-KIF5Be15 in the only case (Table 6).
Discussion
This work enabled us to detect ALK, ROS1, or RET translocations in lung tumors using LD-RT-PCR, with a sensitivity of 64% (25 cases out of 39) and perfect specificity.
The reason why we decided to exclude FISH-positive cases with rearranged nuclei between 15 and 20% was the high risk of a false positive result using this technique,17 as we did not want to test LD-RT-PCR on these unsure 'positive' cases.
The main drawback of LD-RT-PCR is that the number of partner genes and break point is potentially very high and new partner genes are regularly identified, which would imply a regular update of our kit. Theoretically, it could contain up to 150 probes,15 which means that the technique would be outdated quickly. Therefore, the 43-probe assay that we used did not contain all the potential partner genes, which certainly explains the relatively low sensitivity. An alternative explanation, maybe complementary to the first one, is that these rearranged genes are expressed at a very low level, regardless of their 'oncogenic addiction', leading to technical difficulties to detect RNA when using RNA-based methods. This hypothesis is consistent with the fact that, for instance, detection of the ALK rearranged protein in lung carcinoma requires a much more enhanced revelation system that in ALK: for example, the use of an amplification system (such as the Roche OptiView Amplification Kit) is recommended to detect an abnormal ALK expression in lung adenocarcinoma, whereas the ALK1 immunostaining used with a conventional detection system is usually sufficient to detect an abnormal expression in lymphomas.18 However, the fact that LD-RT-PCR enabled us to detect four ALK-positive cases (Table 2) and four ROS-positive cases (Table 4), whereas RNA concentration of these samples were very low according to the Qubit RNA HS Assay kit would suggest that the new technique requires only very few rearranged RNA molecules, and that the first explanation for non-detected cases is more robust. Similarly, it is unlikely that undetected cases were owing to low tumor cell content because some positive cases were paucicellular, whereas some negative cases contained a large proportion of tumor cells (Tables 2 and 4).
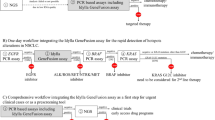
What role could LD-RT-PCR play in routine diagnosis? In 2015, Lantuejoul et al14 recommended performing a second FISH analysis in case of a discrepancy between IHC and FISH results. Because LD-RT-PCR is affordable, simple and a very fast technique, we suggest performing it whenever IHC analysis is positive for ALK or ROS1 even if weak, and directly if a RET translocation is suspected (Figure 3). FISH would be performed only in case of discordance between IHC and LD-RT-PCR results because this assay is not able to detect all translocations and therefore only has significance if positive.
References
IARC. Globocan 2012 [Internet] http://globocan.iarc.fr/Pages/fact_sheets_population.aspx.
Travis WD, Brambilla E, Muller-Hermelink HK et al. WHO Pathology and genetics of tumors of the lung, pleura, thymus and heart. IARC: Lyon, France, 2004.
Travis WD, Brambilla E, Noguchi M et al. International association for the study of lung cancer/american thoracic society/european respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 2011;6:244–285.
Lynch TJ, Bell DW, Sordella R et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med; 350:2129–2139.
Soda M, Choi YL, Enomoto M et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature; 448:561–566.
Takeuchi K, Soda M, Togashi Y et al. RET, ROS1 and ALK fusions in lung cancer. Nat Med 2012;18:378–381.
Kohno T, Ichikawa H, Totoki Y et al. KIF5B-RET fusions in lung adenocarcinoma. Nat Med 2012;18:375–377.
Barlesi F, Mazieres J, Merlio J-P et al. Routine molecular profiling of patients with advanced non-small-cell lung cancer: results of a 1-year nationwide programme of the French Cooperative Thoracic Intergroup (IFCT). Lancet Lond Engl 2016;387:1415–1426.
Gainor JF, Shaw AT . Novel targets in non-small cell lung cancer: ROS1 and RET fusions. Oncologist 2013;18:865–875.
Kwak EL, Bang Y-J, Camidge DR et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med 2010;363:1693–1703.
Camidge DR, Bang Y-J, Kwak EL et al. Activity and safety of crizotinib in patients with ALK-positive non-small-cell lung cancer: updated results from a phase 1 study. Lancet Oncol 2012;13:1011–1019.
Mazières J, Zalcman G, Crinò L et al. Crizotinib therapy for advanced lung adenocarcinoma and a ROS1 rearrangement: results from the EUROS1 cohort. J Clin Oncol 2015;33:992–999.
Kodama T, Tsukaguchi T, Satoh Y et al. Alectinib shows potent antitumor activity against RET-rearranged non-small cell lung cancer. Mol Cancer Ther 2014;13:2910–2918.
Lantuejoul S, Rouquette I, Blons H et al. French multicentric validation of ALK rearrangement diagnostic in 547 lung adenocarcinomas. Eur Respir J 2015;46:207–218.
Mareschal S, Ruminy P, Bagacean C et al. Accurate classification of germinal center B-cell-like/activated B-cell-like diffuse large B-cell lymphoma using a simple and rapid reverse transcriptase-multiplex ligation-dependent probe amplification assay: a CALYM study. J Mol Diagn 2015;17:273–283.
Ruminy P, Marchand V, Buchbinder N et al. Multiplexed targeted sequencing of recurrent fusion genes in acute leukaemia. Leukemia 2015;30:757–760.
von Laffert M, Stenzinger A, Hummel M et al. ALK-FISH borderline cases in non-small cell lung cancer: Implications for diagnostics and clinical decision making. Lung Cancer 2015;90:465–471.
Le Quesne J, Maurya M, Yancheva SG et al. A comparison of immunphistochemical assays and FISH in detecting the ALK translocation in diagnostic histological and cytological lung tumor material. J Thorac Oncol 2014;9:769–774.
Acknowledgements
The authors are indebted to Nikki Sabourin-Gibbs (Rouen University Hospital) for assistance with language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
The authors assessed an innovative, quick and inexpensive RNA-based technique (LD-RT-PCR) to detect ALK, ROS and RET gene translocations in lung adenocarcinomas from formalin-fixed paraffin-embedded samples. Because this method was very specific (100%) but less sensitive (63–64%), we propose using this technique when ALK immunostaining is positive. For LD-RT-PCR-negative cases, samples should then be analyzed by fluorescence in situ hybridization.
Rights and permissions
About this article
Cite this article
Piton, N., Ruminy, P., Gravet, C. et al. Ligation-dependent RT-PCR: a new specific and low-cost technique to detect ALK, ROS, and RET rearrangements in lung adenocarcinoma. Lab Invest 98, 371–379 (2018). https://doi.org/10.1038/labinvest.2017.124
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2017.124
This article is cited by
-
Detection of sarcoma fusions by a next-generation sequencing based–ligation-dependent multiplex RT-PCR assay
Modern Pathology (2022)
-
An improved assay for detection of theranostic gene translocations and MET exon 14 skipping in thoracic oncology
Laboratory Investigation (2021)
-
Simultaneous detection of EGFR amplification and EGFRvIII variant using digital PCR-based method in glioblastoma
Acta Neuropathologica Communications (2020)
-
Rapid detection and genotyping of ALK fusion variants by adapter multiplex PCR and high-resolution melting analysis
Laboratory Investigation (2020)
-
Fluorometric Detection of Oncogenic EML4-ALK Fusion Gene based on a Graphene Oxide System
BioChip Journal (2019)