Abstract
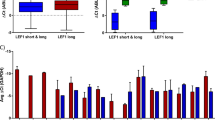
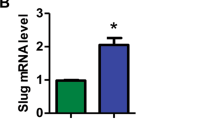
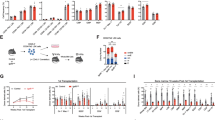
Dysregulation of the Wnt/β-catenin pathway has been observed in various malignancies, including acute myeloid leukemia (AML), where the overexpression of β-catenin is an independent adverse prognostic factor. β-catenin was found upregulated in the vast majority of AML samples and more frequently localized in the nucleus of leukemic stem cells compared with normal bone marrow CD34+ cells. The knockdown of β-catenin, using a short hairpin RNA (shRNA) lentiviral approach, accelerates all-trans retinoic acid-induced differentiation and impairs the proliferation of HL60 leukemic cell line. Using in vivo quantitative tracking of these cells, we observed a reduced engraftment potential after xenotransplantation when β-catenin was silenced. However, when studying primary AML cells, despite effective downregulation of β-catenin we did not observe any impairment of their in vitro long-term maintenance on MS-5 stroma nor of their engraftment potential in vivo. Altogether, these results show that despite a frequent β-catenin upregulation in AML, leukemia-initiating cells might not be ‘addicted’ to this pathway and thus targeted therapy against β-catenin might not be successful in all patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gilliland DG, Jordan CT, Felix CA . The molecular basis of leukemia. Hematol Am Soc Hematol Educ Program 2004; 1: 80–97.
Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 1994; 367: 645–648.
Bonnet D, Dick JE . Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997; 3: 730–737.
Ailles LE, Gerhard B, Kawagoe H, Hogge DE . Growth characteristics of acute myelogenous leukemia progenitors that initiate malignant hematopoiesis in nonobese diabetic/severe combined immunodeficient mice. Blood 1999; 94: 1761–1772.
Ailles LE, Humphries RK, Thomas TE, Hogge DE . Retroviral marking of acute myelogenous leukemia progenitors that initiate long-term culture and growth in immunodeficient mice. Exp Hematol 1999; 27: 1609–1620.
Feuring-Buske M, Gerhard B, Cashman J, Humphries RK, Eaves CJ, Hogge DE . Improved engraftment of human acute myeloid leukemia progenitor cells in ß 2-microglobulin-deficient NOD/SCID mice and in NOD/SCID mice transgenic for human growth factors. Leukemia 2003; 17: 760–763.
Rizo A, Dontje B, Vellenga E, de Haan G, Schuringa JJ . Long-term maintenance of human hematopoietic stem/progenitor cells by expression of BMI1. Blood 2008; 111: 2621–2630.
Sengupta A, Banerjee D, Chandra S, Banerji SK, Ghosh R, Roy R et al. Deregulation and cross talk among Sonic hedgehog, Wnt, Hox and Notch signaling in chronic myeloid leukemia progression. Leukemia 2007; 21: 949–955.
Dash AB, Williams IR, Kutok JL, Tomasson MH, Anastasiadou E, Lindahl K et al. A murine model of CML blast crisis induced by cooperation between BCR/ABL and NUP98/HOXA9. Proc Natl Acad Sci USA 2002; 99: 7622–7627.
Bansal D, Scholl C, Frohling S, McDowell E, Lee BH, Dohner K et al. Cdx4 dysregulates Hox gene expression and generates acute myeloid leukemia alone and in cooperation with Meis1a in a murine model. Proc Natl Acad Sci USA 2006; 103: 16924–16929.
Nusse R . The Wnt Homepage. http://www.stanford.edu/group/nusselab/cgi-bin/wnt/.
Reya T, Duncan AW, Ailles L, Domen J, Scherer DC, Willert K et al. A role for Wnt signaling in self-renewal of haematopoietic stem cells. Nature 2003; 423: 409–414.
Mikesch JH, Steffen B, Berdel WE, Serve H, Muller-Tidow C . The emerging role of Wnt signaling in the pathogenesis of acute myeloid leukemia. Leukemia 2007; 21: 1638–1647.
Willert K, Brown JD, Danenberg E, Duncan AW, Weissman IL, Reya T et al. Wnt proteins are lipid-modified and can act as stem cell growth factors. Nature 2003; 423: 448–452.
Baba Y, Yokota T, Spits H, Garrett KP, Hayashi S, Kincade PW . Constitutively active β-catenin promotes expansion of multipotent hematopoietic progenitors in culture. J Immunol 2006; 177: 2294–2303.
Kirstetter P, Anderson K, Porse BT, Jacobsen SE, Nerlov C . Activation of the canonical Wnt pathway leads to loss of hematopoietic stem cell repopulation and multilineage differentiation block. Nat Immunol 2006; 7: 1048–1056.
Scheller M, Huelsken J, Rosenbauer F, Taketo MM, Birchmeier W, Tenen DG et al. Hematopoietic stem cell and multilineage defects generated by constitutive β-catenin activation. Nat Immunol 2006; 7: 1037–1047.
Zhao C, Blum J, Chen A, Kwon HY, Jung SH, Cook JM et al. Loss of β-catenin impairs the renewal of normal and CML stem cells in vivo. Cancer Cell 2007; 12: 528–541.
Sloma I, Jiang X, Eaves AC, Eaves CJ . Insights into the stem cells of chronic myeloid leukemia. Leukemia 2010; 24: 1823–1833.
Jamieson CH, Ailles LE, Dylla SJ, Muijtjens M, Jones C, Zehnder JL et al. Granulocyte-macrophage progenitors as candidate leukemic stem cells in blast-crisis CML. N Engl J Med 2004; 351: 657–667.
Abrahamsson AE, Geron I, Gotlib J, Dao KH, Barroga CF, Newton IG et al. Glycogen synthase kinase 3ß missplicing contributes to leukemia stem cell generation. Proc Natl Acad Sci USA 2009; 106: 3925–3929.
Brandts CH, Sargin B, Rode M, Biermann C, Lindtner B, Schwable J et al. Constitutive activation of Akt by Flt3 internal tandem duplications is necessary for increased survival, proliferation, and myeloid transformation. Cancer Res 2005; 65: 9643–9650.
Tickenbrock L, Schwable J, Wiedehage M, Steffen B, Sargin B, Choudhary C et al. Flt3 tandem duplication mutations cooperate with Wnt signaling in leukemic signal transduction. Blood 2005; 105: 3699–3706.
Muller-Tidow C, Steffen B, Cauvet T, Tickenbrock L, Ji P, Diederichs S et al. Translocation products in acute myeloid leukemia activate the Wnt signaling pathway in hematopoietic cells. Mol Cell Biol 2004; 24: 2890–2904.
Roman-Gomez J, Cordeu L, Agirre X, Jimenez-Velasco A, San Jose-Eneriz E, Garate L et al. Epigenetic regulation of Wnt-signaling pathway in acute lymphoblastic leukemia. Blood 2007; 109: 3462–3469.
Valencia A, Roman-Gomez J, Cervera J, Such E, Barragan E, Bolufer P et al. Wnt signaling pathway is epigenetically regulated by methylation of Wnt antagonists in acute myeloid leukemia. Leukemia 2009; 23: 1658–1666.
Ysebaert L, Chicanne G, Demur C, De Toni F, Prade-Houdellier N, Ruidavets JB et al. Expression of beta-catenin by acute myeloid leukemia cells predicts enhanced clonogenic capacities and poor prognosis. Leukemia 2006; 20: 1211–1216.
Majeti R, Becker MW, Tian Q, Lee TL, Yan X, Liu R et al. Dysregulated gene expression networks in human acute myelogenous leukemia stem cells. Proc Natl Acad Sci USA 2009; 106: 3396–3401.
Yeung J, Esposito MT, Gandillet A, Zeisig BB, Griessinger E, Bonnet D et al. catenin mediates the establishment and drug resistance of MLL leukemic stem cells. Cancer Cell 2010; 18: 606–618.
Siapati EK, Papadaki M, Kozaou Z, Rouka E, Michali E, Savvidou I et al. Proliferation and bone marrow engraftment of AML blasts is dependent on β-catenin signalling. Br J Haematol 2011; 152: 164–174.
Taussig DC, Miraki-Moud F, Anjos-Afonso F, Pearce DJ, Allen K, Ridler C et al. Anti-CD38 antibody-mediated clearance of human repopulating cells masks the heterogeneity of leukemia-initiating cells. Blood 2008; 112: 568–575.
Taussig DC, Vargaftig J, Miraki-Moud F, Griessinger E, Sharrock K, Luke T et al. Leukemia-initiating cells from some acute myeloid leukemia patients with mutated nucleophosmin reside in the CD34(-) fraction. Blood 2010; 115: 1976–1984.
Ortyn WE, Hall BE, George TC, Frost K, Basiji DA, Perry DJ et al. Sensitivity measurement and compensation in spectral imaging. Cytometry A 2006; 69: 852–862.
Schuringa JJ, Schepers H . Ex vivo assays to study self-renewal and long-term expansion of genetically modified primary human acute myeloid leukemia stem cells. Methods Mol Biol 2009; 538: 287–300.
Matkovic K, Brugnoli F, Bertagnolo V, Banfic H, Visnjic D . The role of the nuclear Akt activation and Akt inhibitors in all-trans-retinoic acid-differentiated HL-60 cells. Leukemia 2006; 20: 941–951.
Wang X, Rosol M, Ge S, Peterson D, McNamara G, Pollack H et al. Dynamic tracking of human hematopoietic stem cell engraftment using in vivo bioluminescence imaging. Blood 2003; 102: 3478–3482.
Luo J, Solimini NL, Elledge SJ . Principles of cancer therapy: oncogene and non-oncogene addiction. Cell 2009; 136: 823–837.
Acknowledgements
We thank all members of the London Research Institute Flow Cytometry and Equipment Park facilities and Christopher Ridler for technical assistance; Stuart Horswell for statistical analysis; Katie Foster and Yasmin Reyal for proof reading of the final paper and David Taussig for helpful discussions. We are grateful to R De Maria for kindly providing us with the TWR Luc−Fluc+RFP plasmid construct and to Fernando Anjos-Afonso for providing us with some of the qPCR primers used here. This work was funded by Cancer Research UK (DB) and by European grant (contract No. 037632) to DB. AG, SP, EG and JV are supported by London Research Institute Cancer Research UK fellowships.
Author Contributions
AG and SP: designed experiments, performed research, analyzed data and wrote the paper; FL and EG: performed some of the experiments; JV: provided clinical data and performed screening for engraftment and phenotype of the LSC used; AF: performed research and analyzed data; TAL: provided AML samples; DB: designed experiments, performed research, analyzed data and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Gandillet, A., Park, S., Lassailly, F. et al. Heterogeneous sensitivity of human acute myeloid leukemia to β-catenin down-modulation. Leukemia 25, 770–780 (2011). https://doi.org/10.1038/leu.2011.17
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.17
Keywords
This article is cited by
-
Regulome analysis in B-acute lymphoblastic leukemia exposes Core Binding Factor addiction as a therapeutic vulnerability
Nature Communications (2022)
-
Non-canonical activation of β-catenin by PRL-3 phosphatase in acute myeloid leukemia
Oncogene (2019)
-
Understanding of leukemic stem cells and their clinical implications
Molecular Cancer (2017)
-
Regulation of normal and leukemic stem cells through cytokine signaling and the microenvironment
International Journal of Hematology (2017)
-
Novel strategies for targeting leukemia stem cells: sounding the death knell for blood cancer
Cellular Oncology (2017)