Abstract
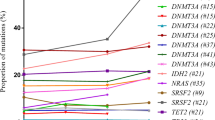
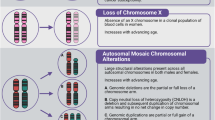
Chronic myelomonocytic leukemia (CMML) is a hematologic malignancy nearly confined to the elderly. Previous studies to determine incidence and prognostic significance of somatic mutations in CMML have relied on candidate gene sequencing, although an unbiased mutational search has not been conducted. As many of the genes commonly mutated in CMML were recently associated with age-related clonal hematopoiesis (ARCH) and aged hematopoiesis is characterized by a myelomonocytic differentiation bias, we hypothesized that CMML and aged hematopoiesis may be closely related. We initially established the somatic mutation landscape of CMML by whole exome sequencing followed by gene-targeted validation. Genes mutated in ⩾10% of patients were SRSF2, TET2, ASXL1, RUNX1, SETBP1, KRAS, EZH2, CBL and NRAS, as well as the novel CMML genes FAT4, ARIH1, DNAH2 and CSMD1. Most CMML patients (71%) had mutations in ⩾2 ARCH genes and 52% had ⩾7 mutations overall. Higher mutation burden was associated with shorter survival. Age-adjusted population incidence and reported ARCH mutation rates are consistent with a model in which clinical CMML ensues when a sufficient number of stochastically acquired age-related mutations has accumulated, suggesting that CMML represents the leukemic conversion of the myelomonocytic-lineage-biased aged hematopoietic system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Patnaik MM, Padron E, LaBorde RR, Lasho TL, Finke CM, Hanson CA et al. Mayo prognostic model for WHO-defined chronic myelomonocytic leukemia: ASXL1 and spliceosome component mutations and outcomes. Leukemia 2013; 27: 1504–1510.
Williamson PJ, Kruger AR, Reynolds PJ, Hamblin TJ, Oscier DG . Establishing the incidence of myelodysplastic syndrome. Br J Haematol 1994; 87: 743–745.
Aul C, Gattermann N, Schneider W . Age-related incidence and other epidemiological aspects of myelodysplastic syndromes. Br J Haematol 1992; 82: 358–367.
Tyner JW, Loriaux MM, Erickson H, Eide CA, Deininger J, MacPartlin M et al. High-throughput mutational screen of the tyrosine kinome in chronic myelomonocytic leukemia. Leukemia 2009; 23: 406–409.
Gelsi-Boyer V, Trouplin V, Roquain J, Adélaïde J, Carbuccia N, Esterni B et al. ASXL1 mutation is associated with poor prognosis and acute transformation in chronic myelomonocytic leukaemia. Br J Haematol 2010; 151: 365–375.
Smith AE, Mohamedali AM, Kulasekararaj A, Lim Z, Gäken J, Lea NC et al. Next-generation sequencing of the TET2 gene in 355 MDS and CMML patients reveals low-abundance mutant clones with early origins, but indicates no definite prognostic value. Blood 2010; 116: 3923–3932.
Jankowska AM, Makishima H, Tiu RV, Szpurka H, Huang Y, Traina F et al. Mutational spectrum analysis of chronic myelomonocytic leukemia includes genes associated with epigenetic regulation: UTX, EZH2, and DNMT3A. Blood 2011; 118: 3932–3941.
Itzykson R, Kosmider O, Renneville A, Gelsi-Boyer V, Meggendorfer M, Morabito M et al. Prognostic score including gene mutations in chronic myelomonocytic leukemia. J Clin Oncol 2013; 31: 2428–2436.
Patnaik MM, Itzykson R, Lasho TL, Kosmider O, Finke CM, Hanson CA et al. ASXL1 and SETBP1 mutations and their prognostic contribution in chronic myelomonocytic leukemia: a two-center study of 466 patients. Leukemia 2014; 28: 2206–2212.
Meggendorfer M, Roller A, Haferlach T, Eder C, Dicker F, Grossmann V et al. SRSF2 mutations in 275 cases with chronic myelomonocytic leukemia (CMML). Blood 2012; 120: 3080–3088.
Laborde RR, Patnaik MM, Lasho TL, Finke CM, Hanson CA, Knudson RA et al. SETBP1 mutations in 415 patients with primary myelofibrosis or chronic myelomonocytic leukemia: independent prognostic impact in CMML. Leukemia 2013; 27: 2100–2102.
Grossmann V, Kohlmann A, Eder C, Haferlach C, Kern W, Cross NC et al. Molecular profiling of chronic myelomonocytic leukemia reveals diverse mutations in >80% of patients with TET2 and EZH2 being of high prognostic relevance. Leukemia 2011; 25: 877–879.
Kuo MC, Liang DC, Huang CF, Shih YS, Wu JH, Lin TL et al. RUNX1 mutations are frequent in chronic myelomonocytic leukemia and mutations at the C-terminal region might predict acute myeloid leukemia transformation. Leukemia 2009; 23: 1426–1431.
Kosmider O, Gelsi-Boyer V, Ciudad M, Racoeur C, Jooste V, Vey N et al. TET2 gene mutation is a frequent and adverse event in chronic myelomonocytic leukemia. Haematologica 2009; 94: 1676–1681.
Kohlmann A, Grossmann V, Klein HU, Schindela S, Weiss T, Kazak B et al. Next-generation sequencing technology reveals a characteristic pattern of molecular mutations in 72.8% of chronic myelomonocytic leukemia by detecting frequent alterations in TET2, CBL, RAS, and RUNX1. J Clin Oncol 2010; 28: 3858–3865.
Xie M, Lu C, Wang J, McLellan MD, Johnson KJ, Wendl MC et al. Age-related mutations associated with clonal hematopoietic expansion and malignancies. Nat Med 2014; 20: 1472–1478.
McKerrell T, Park N, Moreno T, Grove CS, Ponstingl H, Stephens J et al. Leukemia-associated somatic mutations drive distinct patterns of age-related clonal hemopoiesis. Cell Rep 2015; 10: 1239–1245.
Genovese G, Kahler AK, Handsaker RE, Lindberg J, Rose SA, Bakhoum SF et al. Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N Engl J Med 2014; 371: 2477–2487.
Jaiswal S, Fontanillas P, Flannick J, Manning A, Grauman PV, Mar BG et al. Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med 2014; 371: 2488–2498.
Pang WW, Price EA, Sahoo D, Beerman I, Maloney WJ, Rossi DJ et al. Human bone marrow hematopoietic stem cells are increased in frequency and myeloid-biased with age. Proc Natl Acad Sci USA 2011; 108: 20012–20017.
Gondek LP, Tiu R, O'Keefe CL, Sekeres MA, Theil KS, Maciejewski JP . Chromosomal lesions and uniparental disomy detected by SNP arrays in MDS, MDS/MPD, and MDS-derived AML. Blood 2008; 111: 1534–1542.
Shlush LI, Zandi S, Mitchell A, Chen WC, Brandwein JM, Gupta V et al. Identification of pre-leukaemic haematopoietic stem cells in acute leukaemia. Nature 2014; 506: 328–333.
McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res 2010; 20: 1297–1303.
Koboldt DC, Zhang Q, Larson DE, Shen D, McLellan MD, Lin L et al. VarScan 2: somatic mutation and copy number alteration discovery in cancer by exome sequencing. Genome Res 2012; 22: 568–576.
Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, Homer N et al. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009; 25: 2078–2079.
Wang K, Li M, Hakonarson H . ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 2010; 38: e164.
Yandell M, Huff C, Hu H, Singleton M, Moore B, Xing J et al. A probabilistic disease-gene finder for personal genomes. Genome Res 2011; 21: 1529–1542.
Kohane IS, Hsing M, Kong SW . Taxonomizing, sizing, and overcoming the incidentalome. Genet Med 2012; 14: 399–404.
Bamford S, Dawson E, Forbes S, Clements J, Pettett R, Dogan A et al. The COSMIC (Catalogue of Somatic Mutations in Cancer) database and website. Br J Cancer 2004; 91: 355–358.
Tiwari RC, Clegg LX, Zou Z . Efficient interval estimation for age-adjusted cancer rates. Stat Methods Med Res 2006; 15: 547–569.
Onida F, Kantarjian HM, Smith TL, Ball G, Keating MJ, Estey EH et al. Prognostic factors and scoring systems in chronic myelomonocytic leukemia: a retrospective analysis of 213 patients. Blood 2002; 99: 840–849.
Germing U, Kundgen A, Gattermann N . Risk assessment in chronic myelomonocytic leukemia (CMML). Leuk Lymphoma 2004; 45: 1311–1318.
Allen LJS . An Introduction to Stochastic Processes with Applications to Biology. Pearson Education, Inc: Upper Saddle River, NJ, 2003.
Abdel-Wahab O, Adli M, LaFave LM, Gao J, Hricik T, Shih AH et al. ASXL1 mutations promote myeloid transformation through loss of PRC2-mediated gene repression. Cancer Cell 2012; 22: 180–193.
Ernst T, Chase AJ, Score J, Hidalgo-Curtis CE, Bryant C, Jones AV et al. Inactivating mutations of the histone methyltransferase gene EZH2 in myeloid disorders. Nat Genet 2010; 42: 722–726.
Inoue D, Kitaura J, Matsui H, Hou HA, Chou WC, Nagamachi A et al. SETBP1 mutations drive leukemic transformation in ASXL1-mutated MDS. Leukemia 2015; 29: 847–857.
Walter MJ, Shen D, Ding L, Shao J, Koboldt DC, Chen K et al. Clonal architecture of Secondary Acute Myeloid Leukemia. N Engl J Med 2012; 366: 1090–1098.
Durinck S, Ho C, Wanj NJ, Liao W, Jakkula LR, Collisson EA et al. Temporal dissection of tumorigenesis in primary cancers. Cancer Discov 2011; 1: 137–143.
Youn A, Simon R . Estimating the order of mutations during tumorigenesis from tumor genome sequencing data. Bioinformatics 2012; 28: 1555–1561.
Challen GA, Boles NC, Chambers SM, Goodell MA . Distinct hematopoietic stem cell subtypes are differentially regulated by TGF-beta1. Cell Stem Cell 2010; 6: 265–278.
Cho RH, Sieburg HB, Muller-Sieburg CE . A new mechanism for the aging of hematopoietic stem cells: aging changes the clonal composition of the stem cell compartment but not individual stem cells. Blood 2008; 111: 5553–5561.
Beerman I, Bhattacharya D, Zandi S, Sigvardsson M, Weissman IL, Bryder D et al. Functionally distinct hematopoietic stem cells modulate hematopoietic lineage potential during aging by a mechanism of clonal expansion. Proc Natl Acad Sci USA 2010; 107: 5465–5470.
Chambers SM, Shaw CA, Gatza C, Fisk CJ, Donehower LA, Goodell MA . Aging hematopoietic stem cells decline in function and exhibit epigenetic dysregulation. PLoS Biol 2007; 5: e201.
King KY, Goodell MA . Inflammatory modulation of HSCs: viewing the HSC as a foundation for the immune response. Nat Rev Immunol 2011; 11: 685–692.
Acknowledgements
We thank Jonathan Schumacher (ARUP Laboratories) for assisting with the pyrosequencing confirmation, Anthony J Iovino for help with experiments, Jenny Ottley for administrative assistance and James Marvin (University of Utah) for help with fluorescence-activated cell sorting. We are grateful to Dr Nicholas Cross (Southampton, UK) and Dr Tim Ley (Washington University, Saint Louis) for helpful discussions. We also thank Agilent Technologies, Inc. for providing Sure Select XT2 targeted primers. This study was supported by grants from V Foundation for Cancer Research (JWT), The Leukemia & Lymphoma Society (MWD, JWT, BJD), Gabrielle’s Angel Foundation for Cancer Research (JWT), Charles and Ann Johnson Foundation (JG), the National Institutes of Health (5R00CA151457-04, 1R01CA183974-01, CA04963920, 1R01CA178397-01, P01CA049639 and P30 CA042014), the PPHC Human DNA Sequencing Grant, and the Utah Genome Project (MWD). The support and resources from the Center for High Performance Computing at the University of Utah are also gratefully acknowledged. JSK and AME are Fellows of the Leukemia & Lymphoma Society and SKT is a recipient of a fellowship award of the American Society of Hematology.
Author contributions
CCM and JSK conceived and designed the study, collected and assembled the data, analyzed and interpreted the data, and wrote the manuscript. SKT, MSZ, DY, ADP, KRR, AME, BKD collected and assembled the data. TWK, JWT, K-HD, BJD, JG provided study material. ZK, MY analyzed and interpreted the data. RLS collected and assembled the data, and analyzed and interpreted the data. TO collected and assembled the data, and wrote the manuscript. MWD conceived and designed the study, provided study material, analyzed and interpreted the data, and wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
CCM and MWD report a potential related conflict of interest of research funding from Agilent Technologies, Inc. MWD also reports a potential related conflict of interest of research funding from Celgene, Inc. All other authors declare no conflict of interests.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Mason, C., Khorashad, J., Tantravahi, S. et al. Age-related mutations and chronic myelomonocytic leukemia. Leukemia 30, 906–913 (2016). https://doi.org/10.1038/leu.2015.337
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2015.337
This article is cited by
-
Identification of a novel de novo mutation of SETBP1 and new findings of SETBP1 in tumorgenesis
Orphanet Journal of Rare Diseases (2023)
-
Proposals for Clinical Trials in Chronic Myelomonocytic Leukemia
Current Treatment Options in Oncology (2023)
-
Differential prognostic impact of IDH1 and IDH2 mutations in chronic myelomonocytic leukemia
Leukemia (2022)
-
Bone marrow clonal hematopoiesis is highly prevalent in blastic plasmacytoid dendritic cell neoplasm and frequently sharing a clonal origin in elderly patients
Leukemia (2022)
-
The RING finger protein family in health and disease
Signal Transduction and Targeted Therapy (2022)