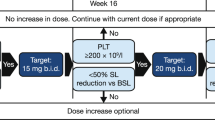
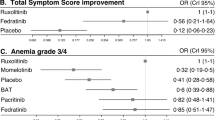
Abstract
Ruxolitinib is an oral Janus-activated kinase 1 (JAK1)/JAK2 inhibitor approved for the treatment of patients with myelofibrosis based on the results of two randomized clinical trials. However, discordant indications were provided by regulatory agencies and scientific societies for selecting the most appropriate candidates to this drug. The European LeukemiaNet and the Italian Society of Hematology shared the aim of building evidence-based recommendations for the use of ruxolitinib according to the GRADE methodology. Eighteen patient-intervention-comparator-outcome profiles were listed, each of them comparing ruxolitinib to other therapies with the aim of improving one of the three clinical outcomes: (a) splenomegaly, (b) disease-related symptoms, and (c) survival. Ruxolitinib was strongly recommended for improving symptomatic or severe (>15 cm below the costal margin) splenomegaly in patients with an International Prognostic Scoring System (IPSS)/dynamic IPSS risk intermediate 2 or high. Ruxolitinib was also strongly recommended for improving systemic symptoms in patients with an MPN10 score >44, refractory severe itching, unintended weight loss not attributable to other causes or unexplained fever. Because of weak evidence, the panel does not recommend ruxolitinib therapy for improving survival. Also, the recommendations given above do not necessarily apply to patients who are candidates for allogeneic stem cell transplant.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sant M, Minicozzi P, Mounier M, Anderson LA, Brenner H, Holleczek B et al. EUROCARE-5 Working Group. Survival for haematological malignancies in Europe between 1997 and 2008 by region and age: results of EUROCARE-5, a population-based study. Lancet Oncol 2014; 15: 931–942.
Lichtenberg FR . The impact of pharmaceutical innovation on premature cancer mortality in Switzerland, 1995-2012. Eur J Health Econ 2016; 17: 833–854.
Lichtenberg FR . The impact of pharmaceutical innovation on premature cancer mortality in Canada, 2000-2011. Int J Health Econ Manag 2016; 5: 339–359.
Harrison C, Kiladjian JJ, Al-Ali HK, Gisslinger H, Waltzman R, Stalbovskaya V et al. JAK inhibition with ruxolitinib versus best available therapy for myelofibrosis. N Engl J Med 2012; 366: 787–798.
Verstovsek S, Mesa RA, Gotlib J, Levy RS, Gupta V, DiPersio JF et al. A double-blind, placebo-controlled trial of ruxolitinib for myelofibrosis. N Engl J Med 2012; 366: 799–807.
Martí-Carvajal AJ, Anand V, Solà I . Janus kinase-1 and Janus kinase-2 inhibitors for treating myelofibrosis. Cochrane Database Syst Rev 2015; 4: CD010298.
Reilly JT, McMullin MF, Beer PA, Butt N, Conneally E, Duncombe AS et al. Use of JAK inhibitors in the management of myelofibrosis: a revision of the British Committee for Standards in Haematology Guidelines for Investigation and Management of Myelofibrosis 2012. Br J Haematol 2014; 167: 418–420.
Vannucchi AM, Barbui T, Cervantes F, Harrison C, Kiladjian JJ, Kroger N et al. Philadelphia chromosome-negative chronic myeloproliferative neoplasms: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2015, v85–v99.
Ho PJ, Marlton P, Tam C, Stevenson W, Ritchie D, Bird R et al. Practical management of myelofibrosis with ruxolitinib. Intern Med J 2015; 45: 1221–1230.
Atkins D, Best D, Briss PA, Eccels M, Falck-Ytter Y, Flottrop S et al. Grading quality of evidence and strength of recommendations. BMJ 2004; 328: 1490–1498.
NICE appraisal 2016: (accessed on 25 April 2016) https://www.nice.org.uk/guidance/TA386/documents/final-appraisal-determination-document.
William PL, Webb C . The Delphi technique: a methodological discussion. J Adv Nurs 1994; 19: 180–186.
Harrison CN, Vannucchi AM, Kiladjian JJ, Al-Ali HK, Gisslinger H, Knoops L et al. Long-term findings from COMFORT-III, a phase 3 study of ruxolitinib vs best available therapy for myelofibrosis. Leukemia 2016; 30: 1701–1707.
Mesa RA, Kiladjian JJ, Verstovsek S, Al-Ali HK, Gotlib J, Gisslinger H et al. Comparison of placebo and best available therapy for the treatment of myelofibrosis in the phase 3 COMFORT studies. Haematologica 2014; 99: 292–298.
Mead AJ, Milojkovic D, Knapper S, Garg M, Chacko J, Farquharson M et al. Response to ruxolitinib in patients with intermediate-1-, intermediate-2-, and high-risk myelofibrosis: results of the UK ROBUST Trial. Br J Haematol 2015; 170: 29–39.
Kathrin Al-Ali Haifa, Griesshammer M, le Coutre P, Waller CF, Liberati AM, Schafausen P et al. Safety and efficacy of ruxolitinib in an open-label, multicenter, single-arm phase 3b expanded-access study in patients with myelofibrosis: a snapshot of 1144 patients in the JUMP trial. Haematologica 2016; 101: 1065–1073.
Kiladjian JJ, Giraudier S, Cassinat B . Interferon-alpha for the therapy of myeloproliferative neoplasms: targeting the malignant clone. Leukemia 2016; 30: 776–781.
Ianotto JC1 Boyer-Perrard F, Gyan E, Laribi K, Cony-Makhoul P, Demory JL et al. Efficacy and safety of pegylated-interferon a-2a in myelofibrosis: a study by the FIM and GEM French cooperative groups. Br J Haematol 2013; 162: 783–791.
Silver RT, Vandris K, Goldman JJ . Recombinant interferon-a may retard progression of early primary myelofibrosis: a preliminary report. Blood 2011; 117: 6669–6672.
Gowin K, Thapaliya P, Samuelsson J, Harrison C, Radia D, Andreasson B et al. Experience with pegylated Interferon alpha-2a in advanced myeloproliferative neoplasms in an international cohort of 118 patients. Haematologica 2012; 97: 1570–1573.
Scherber R, Duecka A, Geyer H, Kosiorek H, Kiladjian JJ, Slots S et al. Symptoms, Risk Classification, and Spleen Size in JAK2 Inhibitor-Naive Myelofibrosis: Implications for JAK2 Inhibitor Treatment. E1345. 21st EHA Meeting, 9-12 June 2016 Copenhagen, Denmark, Available at http://learningcenter.ehaweb.org/eha/2016/21st/137125/eha.annual.congress.eha21.abstract.book.html?f=p5m6l12006.
Vannucchi AM, Kantarjian HM, Kiladjian JJ, Gotllib J, Cervantes F, Mesa RA et al. A pooled analysis of overall survival in COMFORT-I and COMFORT-II, 2 randomized phase III trials of ruxolitinib for the treatment of myelofibrosis. Haematologica 2015; 100: 1139–1145.
Verstovsek S, Kantarjian HM, Estrov Z, Cortes JA, Thoams DA, Kadia T et al. Long-term outcomes of 107 patients with myelofibrosis receiving JAK1/JAK2 inhibitor ruxolitinib: survival advantage in comparison to matched historical controls. Blood 2012; 120: 1202–1209.
Passamonti F, Maffioli M, Cervantes F, Vannucchi AM, Morra E, Barbui T et al. Impact of ruxolitinib on the natural history of primary myelofibrosis: a comparison of the DIPSS and the COMFORT-2 cohorts. Blood 2014; 123: 1833–1835.
Talpaz M, Paquette R, Afrin L, Hamburg SI, Prchal JT, Jamieson K et al. Interim analysis of safety and efficacy of ruxolitinib in patients with myelofibrosis and low platelet counts. J Hematol Oncol 2013; 6: 81.
Griesshammer M, Vannucci AM, Le Coutre P, Tavares RS, Al-Ali HK, Raani P et al Safety and efficacy of ruxolitinib in patients with low platelets enrolled in a phase 3b expanded-access study in myelofibrosis (MF) ASH 2014. Abstract 1859.
Vannucchi AM, Gisslinger H, Harrison CN, Al-Ali HK, Pungolino E, Kiladjiean J-J et al EXPAND: a phase 1b, open-label, dose-finding study of ruxolitinib in patients with myelofibrosis (MF) and low platelet counts (50 × 109/L to 99 × 109/L) at baseline. ASH 2015. Abstract 2817.
Galli S, McLornan D, Harrison C . Safety evaluation of ruxolitinib for treating myelofibrosis. Expert Opin Drug Saf 2014; 13: 967–976.
Harrison C, Mesa R, Ross D, Mead A, Keohane C, Gotlib J et al. Practical management of patients with myelofibrosis receiving ruxolitinib. Expert Rev Hematol 2013; 6: 511–523.
Pieri L, Paoli C, Arena U, Marra F, Mori F, Zucchini M et al Long-term follow-up of a phase 2 study of ruxolitinib in patients with splanchnic vein thrombosis associated with myeloproliferative neoplasms, ASH 2015: abstract 2803.
Colaizzo D, Amitrano L, Guardascione MA, Tiscia GL, D'Andrea G, Longo VA et al. Outcome of patients with splanchnic venous thrombosis presenting without overt MPN: a role for the JAK2 V617F mutation re-evaluation. Thromb Res 2013; 132: e99–e104.
Ageno W, Riva N, Schulman S, Beyer-Westendorf J, Bang SM, Senzolo M et al. Long-term clinical outcomes of splanchnic vein thrombosis: results of an International Registry. JAMA Intern Med 2015; 175: 1474–1480.
Benjamini O, Jain P, Estrov Z, Kantarjian HM, Verstovsek S . Therapeutic effects of ruxolitinib in patients with myelofibrosis without clinically significant splenomegaly. Blood 2012; 120: 2768–2769.
Verstovsek S, Kantanrjian H, Mesa RA, Pardanani AD, Cortes-Franco J, Thomas DA et al. Safety and efficacy of INCB18424, a JAK1 and JAK2 inhibitor, in myelofibrosis. N Engl J Med 2010; 363: 1117–1127.
Isfort S, Kaifie A, Bennemann K, Jost E, Panse J, Bruemmerndorf TH et al. Comorbidity scales in patients with myeloproliferative neoplasms: which one fits best? Blood 2014; 124: 1828.
EPAR EMA. http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/002464/human_med_001568.jsp&mid=vvv WC0b01ac058001d124. Accessed on 4 July 2016.
FDA: ruxolitinib prescribing information www.accessdata.fda.gov/drugsatfda_docs/label/2011/202192lbl.pdf. Accessed on 4 July 2016.
Tefferi A, Cervantes F, Mesa R, Passamonti F, Verstovsek S, Vannucchi AM et al. Revised response criteria for myelofibrosis: International Working Group-Myeloproliferative neoplasms Research and Treatment (IWG-MRT) and European LeukemiaNet (ELN) consensus report. Blood 2013; 122: 1395–1398.
Pardanani A, Tefferi A . Definition and management of ruxolitinib treatment failure in myelofibrosis. Blood Cancer J 2014; 4: e268.
Kroger NM, Deeg JH, Olavarria E, Niederwieser D, Bacigalupo A, Barbui T et al. Indication and management of allogeneic stem cell transplantation in primary myelofibrosis: a consensus process by EBMT/ELN international working group. Leukemia 2015; 29: 2126–2133.
Mehta J, Wang H, Fryzek JP, Iqbal SU, Mesa R . Health resource utilization and cost associated with myeloproliferative neoplasms in a large United States health plan. Leuk Lymphoma 2014; 55: 2368–2374.
Ouagari K, Knight CJ, Mendelson ET . Cost-effectiveness of ruxolitinib versus best-available therapy for medical treatment of myelofibrosis: Canadian societal perspective. Am Soc Hematol Meeting 2012; abstract 4255.
Loblaw DA, Prestrud AA, Somerfield MR, Oliver TK, Brouwers MC, Nam RK et al. American Society of Clinical Oncology Clinical Practice Guidelines: formal systematic review-based consensus methodology. J Clin Oncol 2012; 30: 3136–3140.
Passamonti F, Cervantes F, Vannucchi AM, Morra E, Rumi E, Pereira A . A dynamic prognostic model to predict survival in primary myelofibrosis: a study by the IWG-MRT (International Working Group for Myeloproliferative Neoplasms Research and Treatment). Blood 2010; 115: 1703–1708.
Cervantes F, Dupriez B, Pereira A, Passamonti F, Reilly JT, Morra E et al. New prognostic scoring system for primary myelofibrosis based on a study of the International Working Group for Myelofibrosis Research and Treatment. Blood 2009; 113: 2895–2901.
Scherber R, Dueck AC, Johansson P, Barbui T, Barosi G, Vannucchi AM et al. The Myeloproliferative Neoplasm Symptom Assessment Form (MPN-SAF): international prospective validation and reliability trial in 402 patients. Blood 2011; 118: 401–408.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Barosi participated in speakers’ bureau for Novartis. Dr Kiladjian’s research was funded by Novartis and AOP Orphan; he also participated in speakers’ bureau for Novartis and AOP Orphan. Dr Passamonti participated in speakers’ bureau for Novartis. Dr Griesshammer received travel reimbursement from Novartis and Shire, participated in speakers’ bureaus for Baxalta, AOP Orphan, Novartis and Shire and received honoraria from Baxalta, AOP Orphan, Novartis and Shire. Dr Vannucchi participated in advisory boards and speakers’ bureaus for Novartis. Dr Kröeger received honorarium and research funding from Novartis. Dr McMullin received speaker fee from Novartis and Shire. Dr Harrison received research support from Novartis and honoraria from Baxalta, Novartis, Sanofi and Shire; she joined speakers’ bureaus for Baxalta, Incyte, CTI, Novartis, Sanofi and Shire. Dr Barbui received research grants and speaker fees from Novartis. Dr Marchetti’s research was funded by Janssen, Shire and Celgene; she received travel reimbursements from Janssen, Baxalta, Celgene and Shire and consultant or speaker fees from Gilead and Celgene. Dr Cervantes was a member of advisory boards for Novartis, Baxalta and AOP; he also joined speakers’ bureaus for Novartis, Baxalta and AOP. Dr Birgegård received research funding and speaker fees from Shire.
Author contributions
BT and MM conceived the project. All the authors contributed to the discussion on the PICOs and approved final recommendations. MM conducted the critical appraisal of the literature, tracked the feedbacks from the panelists and drafted the paper. All the authors revised the manuscript and approved the final version.
Rights and permissions
About this article
Cite this article
Marchetti, M., Barosi, G., Cervantes, F. et al. Which patients with myelofibrosis should receive ruxolitinib therapy? ELN-SIE evidence-based recommendations. Leukemia 31, 882–888 (2017). https://doi.org/10.1038/leu.2016.283
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.283
This article is cited by
-
Neoplastic fibrocytes play an essential role in bone marrow fibrosis in Jak2V617F-induced primary myelofibrosis mice
Leukemia (2021)
-
Low-dose ruxolitinib shows effective in treating myelofibrosis
Annals of Hematology (2021)
-
Tracing the decision-making process for myelofibrosis: diagnosis, stratification, and management of ruxolitinib therapy in real-word practice
Annals of Hematology (2020)
-
Management of Myelofibrosis: from Diagnosis to New Target Therapies
Current Treatment Options in Oncology (2020)
-
Efficacy and safety of ruxolitinib and hydroxyurea combination in patients with hyperproliferative myelofibrosis
Annals of Hematology (2019)