Abstract
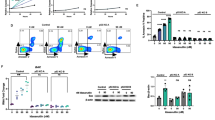
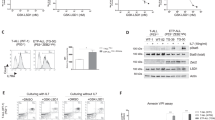
Inhibition of anti-apoptotic BCL-2 (B-cell lymphoma 2) has recently emerged as a promising new therapeutic strategy for the treatment of a variety of human cancers, including leukemia. Here, we used T-cell acute lymphoblastic leukemia (T-ALL) as a model system to identify novel synergistic drug combinations with the BH3 mimetic venetoclax (ABT-199). In vitro drug screening in primary leukemia specimens that were derived from patients with high risk of relapse or relapse and cell lines revealed synergistic activity between venetoclax and the BET (bromodomain and extraterminal) bromodomain inhibitor JQ1. Notably, this drug synergism was confirmed in vivo using T-ALL cell line and patient-derived xenograft models. Moreover, the therapeutic benefit of this drug combination might, at least in part, be mediated by an acute induction of the pro-apoptotic factor BCL2L11 and concomitant reduction of BCL-2 upon BET bromodomain inhibition, ultimately resulting in an enhanced binding of BIM (encoded by BCL2L11) to BCL-2. Altogether, our work provides a rationale to develop a new type of targeted combination therapy for selected subgroups of high-risk leukemia patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Pui C-H, Relling MV, Downing JR . Acute lymphoblastic leukemia. N Engl J Med 2004; 350: 1535–1548.
Pui CH, Pei D, Campana D, Cheng C, Sandlund JT, Bowman WP et al. A revised definition for cure of childhood acute lymphoblastic leukemia. Leukemia 2014; 28: 2336–2343.
Pui CH, Mullighan CG, Evans WE, Relling MV . Pediatric acute lymphoblastic leukemia: where are we going and how do we get there? Blood 2012; 120: 1165–1174.
Bhojwani D, Pui CH . Relapsed childhood acute lymphoblastic leukaemia. Lancet Oncol 2013; 14: e205–e217.
Bassan R, Hoelzer D . Modern therapy of acute lymphoblastic leukemia. J Clin Oncol 2011; 29: 532–543.
Oriol A, Vives S, Hernandez-Rivas JM, Tormo M, Heras I, Rivas C et al. Outcome after relapse of acute lymphoblastic leukemia in adult patients included in four consecutive risk-adapted trials by the PETHEMA Study Group. Haematologica 2010; 95: 589–596.
Faderl S, O'Brien S, Pui CH, Stock W, Wetzler M, Hoelzer D et al. Adult acute lymphoblastic leukemia: concepts and strategies. Cancer 2010; 116: 1165–1176.
Peirs S, Matthijssens F, Goossens S, Van de Walle I, Ruggero K, de Bock CE et al. ABT-199 mediated inhibition of BCL-2 as a novel therapeutic strategy in T-cell acute lymphoblastic leukemia. Blood 2014; 124: 3738–3747.
Anderson NM, Harrold I, Mansour MR, Sanda T, McKeown M, Nagykary N et al. BCL2-specific inhibitor ABT-199 synergizes strongly with cytarabine against the early immature LOUCY cell line but not more-differentiated T-ALL cell lines. Leukemia 2014; 28: 1145–1148.
Chonghaile TN, Roderick JE, Glenfield C, Ryan J, Sallan SE, Silverman LB et al. Maturation stage of T-cell acute lymphoblastic leukemia determines BCL-2 versus BCL-XL dependence and sensitivity to ABT-199. Cancer Discov 2014; 4: 1074–1087.
Fresquet V, Rieger M, Carolis C, Garcia-Barchino MJ, Martinez-Climent JA . Acquired mutations in BCL2 family proteins conferring resistance to the BH3 mimetic ABT-199 in lymphoma. Blood 2014; 123: 4111–4119.
Tahir SK, Smith ML, Hessler P, Roberts-Rapp L, Leverson JD, Lam LT . Abstract B30: Mechanisms of resistance to ABT-199 in leukemia and lymphoma cell lines. Clin Cancer Res 2015; 21: B30.
Choudhary GS, Al-Harbi S, Mazumder S, Hill BT, Smith MR, Bodo J et al. MCL-1 and BCL-xL-dependent resistance to the BCL-2 inhibitor ABT-199 can be overcome by preventing PI3K/AKT/mTOR activation in lymphoid malignancies. Cell Death Dis 2015; 6: e1593.
Roberts AW, Davids MS, Pagel JM, Kahl BS, Puvvada SD, Gerecitano JF et al. Targeting BCL2 with venetoclax in relapsed chronic lymphocytic leukemia. N Engl J Med 2016; 374: 311–322.
Stilgenbauer S, Eichhorst B, Schetelig J, Coutre S, Seymour JF, Munir T et al. Venetoclax in relapsed or refractory chronic lymphocytic leukaemia with 17p deletion: a multicentre, open-label, phase 2 study. Lancet Oncol 2016; 17: 768–778.
Filippakopoulos P, Qi J, Picaud S, Shen Y, Smith WB, Fedorov O et al. Selective inhibition of BET bromodomains. Nature 2010; 468: 1067–1073.
Lovén J, Hoke HA, Lin CY, Lau A, Orlando DA, Vakoc CR et al. Selective inhibition of tumor oncogenes by disruption of super-enhancers. Cell 2013; 153: 320–334.
Hnisz D, Abraham BJ, Lee TI, Lau A, Saint-Andre V, Sigova AA et al. Super-enhancers in the control of cell identity and disease. Cell 2013; 155: 934–947.
Filippakopoulos P, Knapp S . Targeting bromodomains: epigenetic readers of lysine acetylation. Nat Rev Drug Discov 2014; 13: 337–356.
Loosveld M, Castellano R, Gon S, Goubard A, Crouzet T, Pouyet L et al. Therapeutic targeting of c-Myc in T-cell acute lymphoblastic leukemia, T-ALL. Oncotarget 2014; 5: 3168–3172.
Roderick JE, Tesell J, Shultz LD, Brehm MA, Greiner DL, Harris MH et al. c-Myc inhibition prevents leukemia initiation in mice and impairs the growth of relapsed and induction failure pediatric T-ALL cells. Blood 2014; 123: 1040–1050.
King B, Trimarchi T, Reavie L, Xu L, Mullenders J, Ntziachristos P et al. The ubiquitin ligase FBXW7 modulates leukemia-initiating cell activity by regulating MYC stability. Cell 2013; 153: 1552–1566.
Bonapace L, Bornhauser BC, Schmitz M, Cario G, Ziegler U, Niggli FK et al. Induction of autophagy-dependent necroptosis is required for childhood acute lymphoblastic leukemia cells to overcome glucocorticoid resistance. J Clin Invest 2010; 120: 1310–1323.
R Development Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria 2008; ISBN 3-900051-07-0 Available at http://www.R-project.org.http://wwproject.org.
Edgar R, Domrachev M, Lash AE . Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 2002; 30: 207–210.
Yang X, Boehm JS, Yang X, Salehi-Ashtiani K, Hao T, Shen Y et al. A public genome-scale lentiviral expression library of human ORFs. Nat Methods 2011; 8: 659–661.
Meerbrey KL, Hu G, Kessler JD, Roarty K, Li MZ, Fang JE et al. The pINDUCER lentiviral toolkit for inducible RNA interference in vitro and in vivo. Proc Natl Acad Sci USA 2011; 108: 3665–3670.
Fischer U, Forster M, Rinaldi A, Risch T, Sungalee S, Warnatz HJ et al. Genomics and drug profiling of fatal TCF3-HLF-positive acute lymphoblastic leukemia identifies recurrent mutation patterns and therapeutic options. Nat Genet 2015; 47: 1020–1029.
Frismantas V, Dobay MP, Rinaldi A, Tchinda J, Dunn SH, Kunz J et al. Ex vivo drug response profiling detects recurrent sensitivity patterns in drug resistant ALL. Blood 2017; blood-2016-09-738070. Available at: https://doi.org/10.1182/blood-2016-09-738070.
Berthon C, Raffoux E, Thomas X, Vey N, Gomez-Roca C, Yee K et al. Bromodomain inhibitor OTX015 in patients with acute leukaemia: a dose-escalation, phase 1 study. Lancet Haematol 2016; 3: e186–e195.
Amorim S, Stathis A, Gleeson M, Iyengar S, Magarotto V, Leleu X et al. Bromodomain inhibitor OTX015 in patients with lymphoma or multiple myeloma: a dose-escalation, open-label, pharmacokinetic, phase 1 study. Lancet Haematol 2016; 3: e196–e204.
Wallaert A, Durinck K, Van Loocke W, Van de Walle I, Matthijssens F, Volders PJ et al. Long noncoding RNA signatures define oncogenic subtypes in T-cell acute lymphoblastic leukemia. Leukemia 2016; 30: 1927–1930.
Souers AJ, Leverson JD, Boghaert ER, Ackler SL, Catron ND, Chen J et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat Med 2013; 19: 202–208.
Del Gaizo Moore V, Brown JR, Certo M, Love TM, Novina CD, Letai A . Chronic lymphocytic leukemia requires BCL2 to sequester prodeath BIM, explaining sensitivity to BCL2 antagonist ABT-737. J Clin Invest 2007; 117: 112–121.
Merino D, Khaw SL, Glaser SP, Anderson DJ, Belmont LD, Wong C et al. Bcl-2, Bcl-x(L), and Bcl-w are not equivalent targets of ABT-737 and navitoclax (ABT-263) in lymphoid and leukemic cells. Blood 2012; 119: 5807–5816.
Khaw SL, Merino D, Anderson MA, Glaser SP, Bouillet P, Roberts AW et al. Both leukaemic and normal peripheral B lymphoid cells are highly sensitive to the selective pharmacological inhibition of prosurvival Bcl-2 with ABT-199. Leukemia 2014; 28: 1207–1215.
Mogilyansky E, Rigoutsos I . The miR-17/92 cluster: a comprehensive update on its genomics, genetics, functions and increasingly important and numerous roles in health and disease. Cell Death Differ 2013; 20: 1603–1614.
Cang S, Iragavarapu C, Savooji J, Song Y, Liu D . ABT-199 (venetoclax) and BCL-2 inhibitors in clinical development. J Hematol Oncol 2015; 8: 129.
Cervantes-Gomez F, Lamothe B, Woyach JA, Wierda WG, Keating MJ, Balakrishnan K et al. Pharmacological and protein profiling suggests venetoclax (ABT-199) as optimal partner with ibrutinib in chronic lymphocytic leukemia. Clin Cancer Res 2015; 21: 3705–3715.
Chiron D, Dousset C, Brosseau C, Touzeau C, Maiga S, Moreau P et al. Biological rational for sequential targeting of Bruton tyrosine kinase and Bcl-2 to overcome CD40-induced ABT-199 resistance in mantle cell lymphoma. Oncotarget 2015; 6: 8750–8759.
Zhao X, Bodo J, Sun D, Durkin L, Lin J, Smith MR et al. Combination of ibrutinib with ABT-199: synergistic effects on proliferation inhibition and apoptosis in mantle cell lymphoma cells through perturbation of BTK, AKT and BCL2 pathways. Br J Haematol 2015; 168: 765–768.
Ruvolo PP, Ruvolo VR, Benton CB, AlRawi A, Burks JK, Schober W et al. Combination of galectin inhibitor GCS-100 and BH3 mimetics eliminates both p53 wild type and p53 null AML cells. Biochim Biophys Acta 2016; 1863: 562–571.
Ham J, Costa C, Sano R, Lochmann TL, Sennott EM, Patel NU et al. Exploitation of the apoptosis-primed state of MYCN-amplified neuroblastoma to develop a potent and specific targeted therapy combination. Cancer Cell 2016; 29: 159–172.
Phillips DC, Xiao Y, Lam LT, Litvinovich E, Roberts-Rapp L, Souers AJ et al. Loss in MCL-1 function sensitizes non-Hodgkin's lymphoma cell lines to the BCL-2-selective inhibitor venetoclax (ABT-199). Blood Cancer J 2015; 5: e368.
Choudhary GS, Tat TT, Misra S, Hill BT, Smith MR, Almasan A et al. Cyclin E/Cdk2-dependent phosphorylation of Mcl-1 determines its stability and cellular sensitivity to BH3 mimetics. Oncotarget 2015; 6: 16912–16925.
Lee JS, Tang SS, Ortiz V, Vo TT, Fruman DA . MCL-1-independent mechanisms of synergy between dual PI3K/mTOR and BCL-2 inhibition in diffuse large B cell lymphoma. Oncotarget 2015; 6: 35202–35217.
Sun B, Shah B, Fiskus W, Qi J, Rajapakshe K, Coarfa C et al. Synergistic activity of BET protein antagonist-based combinations in mantle cell lymphoma cells sensitive or resistant to ibrutinib. Blood 2015; 126: 1565–1574.
Cinar M, Rosenfelt F, Rokhsar S, Lopategui J, Pillai R, Cervania M et al. Concurrent inhibition of MYC and BCL2 is a potentially effective treatment strategy for double hit and triple hit B-cell lymphomas. Leuk Res 2015; 39: 730–738.
Johnson-Farley N, Veliz J, Bhagavathi S, Bertino JR . ABT-199, a BH3 mimetic that specifically targets Bcl-2, enhances the antitumor activity of chemotherapy, bortezomib and JQ1 in 'double hit' lymphoma cells. Leuk Lymphoma 2015; 56: 2146–2152.
Certo M, Del Gaizo Moore V, Nishino M, Wei G, Korsmeyer S, Armstrong SA et al. Mitochondria primed by death signals determine cellular addiction to antiapoptotic BCL-2 family members. Cancer Cell 2006; 9: 351–365.
Durinck K, Van Loocke W, Van der Meulen J, Van de Walle I, Ongenaert M, Rondou P et al. Characterization of the genome-wide TLX1 binding profile in T-cell acute lymphoblastic leukemia. Leukemia 2015; 29: 2317–2327.
Bhadury J, Nilsson LM, Muralidharan SV, Green LC, Li Z, Gesner EM et al. BET and HDAC inhibitors induce similar genes and biological effects and synergize to kill in Myc-induced murine lymphoma. Proc Natl Acad Sci USA 2014; 111: E2721–E2730.
Hata AN, Engelman JA, Faber AC . The BCL2 family: key mediators of the apoptotic response to targeted anticancer therapeutics. Cancer Discov 2015; 5: 475–487.
Tinsley S, Meja K, Shepherd C, Khwaja A . Synergistic induction of cell death in haematological malignancies by combined phosphoinositide-3-kinase and BET bromodomain inhibition. Br J Haematol 2015; 170: 275–278.
Patel AJ, Liao CP, Chen Z, Liu C, Wang Y, Le LQ . BET bromodomain inhibition triggers apoptosis of NF1-associated malignant peripheral nerve sheath tumors through Bim induction. Cell Rep 2014; 6: 81–92.
Li GQ, Guo WZ, Zhang Y, Seng JJ, Zhang HP, Ma XX et al. Suppression of BRD4 inhibits human hepatocellular carcinoma by repressing MYC and enhancing BIM expression. Oncotarget 2016; 7: 2462–2474.
Xu Z, Sharp PP, Yao Y, Segal D, Ang CH, Khaw SL et al. BET inhibition represses miR17-92 to drive BIM-initiated apoptosis of normal and transformed hematopoietic cells. Leukemia 2016; 30: 1531–1541.
Acknowledgements
We thank the following funding agencies: Fund for Scientific Research Flanders (‘FWO Vlaanderen’ research projects GA00113N, 3G065614, G.0C47.13N and 31500615W to PVV; research projects G.0529.12N and G.0817.13N to GB; doctoral grant to SP; postdoctoral grant to SG; BP is a senior clinical investigator), Children Cancer Fund Ghent, Belgian Foundation Against Cancer (Grants 365W3415W and B/13590) and the Belgian Stand Up To Cancer Foundation (Research Grant 365Y9115W; doctoral grant to SP; postdoctoral grants to TP and FM), agency for Innovation by Science and Technology (‘IWT’, SB Grant 111528 to NV), Geconcerteerde Onderzoeksacties Ghent University (GOA-01GB1013W to GB), Cancer League of the Canton of Zurich, Empiris Foundation, Kinderkrebsforschung Schweiz, Sassella Foundation, Stiftung für Krebsbekämpfung, Swiss National Science Foundation (310030-133108), Fondation Panacée and the clinical research focus program ‘Human Hemato-Lymphatic Diseases’ of the University of Zurich. We also thank Lindy Reunes for excellent technical assistance and the Innovative Flemish in vivo imaging technology (INFINITY) laboratory at Ghent University Hospital. Finally, the computational resources (Stevin Supercomputer Infrastructure) and services used in this work were provided by the VSC (Flemish Supercomputer Center), funded by Ghent University, the Hercules Foundation and the Flemish Government–Department EWI.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Peirs, S., Frismantas, V., Matthijssens, F. et al. Targeting BET proteins improves the therapeutic efficacy of BCL-2 inhibition in T-cell acute lymphoblastic leukemia. Leukemia 31, 2037–2047 (2017). https://doi.org/10.1038/leu.2017.10
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2017.10
This article is cited by
-
Bromodomain and extraterminal (BET) proteins: biological functions, diseases, and targeted therapy
Signal Transduction and Targeted Therapy (2023)
-
Targeted therapy and immunotherapy for T cell acute lymphoblastic leukemia/lymphoma
Annals of Hematology (2023)
-
BRD4 PROTAC degrader ARV-825 inhibits T-cell acute lymphoblastic leukemia by targeting 'Undruggable' Myc-pathway genes
Cancer Cell International (2021)
-
Relapsed/refractory early T-cell precursor acute lymphoblastic leukemia was salvaged by venetoclax plus HAG regimen
Annals of Hematology (2020)
-
Bromodomains: a new target class for drug development
Nature Reviews Drug Discovery (2019)